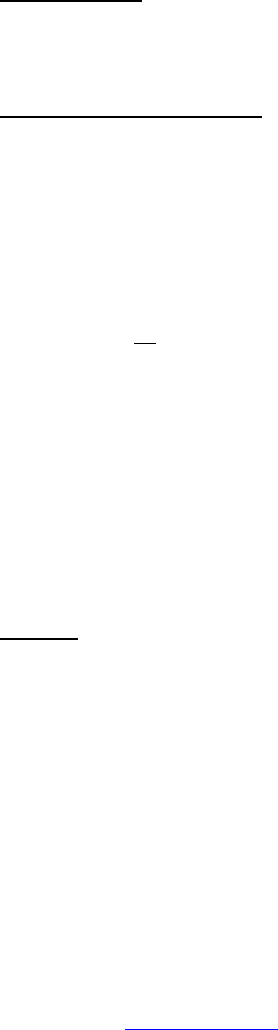
Cancer Therapy Evaluation Program, Common Terminology Criteria for Adverse Events, Version 3.0, DCTD, NCI, NIH, DHHS
March 31, 2003 (http://ctep.cancer.gov
), Publish Date: August 9, 2006
Common Terminology Criteria for Adverse Events v3.0 (CTCAE)
Publish Date: August 9, 2006
Quick Reference
The NCI Common Terminology Criteria for Adverse Events
v3.0 is a descriptive terminology which can be utilized for
Adverse Event (AE) reporting. A grading (severity) scale is
provided for each AE term.
Components and Organization
CATEGORY
A CATEGORY is a broad classification of AEs based on
anatomy and/or pathophysiology. Within each CATEGORY,
AEs are listed accompanied by their descriptions of severity
(Grade).
Adverse Event Terms
An AE is any unfavorable and unintended sign (including an
abnormal laboratory finding), symptom, or disease temporally
associated with the use of a medical treatment or procedure
that may or may not
be considered related to the medical
treatment or procedure. An AE is a term that is a unique
representation of a specific event used for medical
documentation and scientific analyses. Each AE term is
mapped to a MedDRA term and code. AEs are listed
alphabetically within CATEGORIES.
Short AE Name
The ‘SHORT NAME’ column is new and it is used to simplify
documentation of AE names on Case Report Forms.
Supra-ordinate Terms
A supra-ordinate term is located within a CATEGORY and is a
grouping term based on disease process, signs, symptoms,
or diagnosis. A supra-ordinate term is followed by the word
‘Select’ and is accompanied by specific AEs that are all
related to the supra-ordinate term. Supra-ordinate terms
provide clustering and consistent representation of Grade for
related AEs. Supra-ordinate terms are not AEs, are not
mapped to a MedDRA term and code, cannot be graded and
cannot be used for reporting.
REMARK
A ‘REMARK’ is a clarification of an AE.
ALSO CONSIDER
An ‘ALSO CONSIDER’ indicates additional AEs that are to be
graded if they are clinically significant.
NAVIGATION NOTE
A ‘NAVIGATION NOTE’ indicates the location of an AE term
within the CTCAE document. It lists signs/symptoms
alphabetically and the CTCAE term will appear in the same
CATEGORY unless the ‘N
AVIGATION NOTE’ states differently.
Grades
Grade refers to the severity of the AE. The CTCAE v3.0
displays Grades 1 through 5 with unique clinical descriptions
of severity for each AE based on this general guideline:
Grade 1 Mild AE
Grade 2 Moderate AE
Grade 3 Severe AE
Grade 4 Life-threatening or disabling AE
Grade 5 Death related to AE
A Semi-colon indicates ‘or’ within the description of the grade.
An ‘Em dash’ (—) indicates a grade not available.
Not all Grades are appropriate for all AEs. Therefore, some
AEs are listed with fewer than five options for Grade
selection.
Grade 5
Grade 5 (Death) is not appropriate for some AEs and
therefore is not an option.
The DEATH CATEGORY is new. Only one Supra-ordinate
term is listed in this CATEGORY: ‘Death not associated with
CTCAE term – Select’ with 4 AE options: Death NOS;
Disease progression NOS; Multi-organ failure; Sudden death.
Important:
• Grade 5 is the only appropriate Grade
• This AE is to be used in the situation where
a death
1. cannot be reported using a CTCAE v3.0
term associated with Grade 5, or
2. cannot be reported within a CTCAE
CATEGORY as ‘Other (Specify)’
Contents
ALLERGY/IMMUNOLOGY ............................................ 1
AUDITORY/EAR............................................................ 2
BLOOD/BONE MARROW ............................................. 4
CARDIAC ARRHYTHMIA.............................................. 5
CARDIAC GENERAL .................................................... 7
COAGULATION .......................................................... 10
CONSTITUTIONAL SYMPTOMS................................ 11
DEATH ........................................................................ 13
DERMATOLOGY/SKIN ............................................... 14
ENDOCRINE ............................................................... 17
GASTROINTESTINAL................................................. 19
GROWTH AND DEVELOPMENT................................ 29
HEMORRHAGE/BLEEDING ....................................... 30
HEPATOBILIARY/PANCREAS ................................... 34
INFECTION................................................................. 35
LYMPHATICS ............................................................. 38
METABOLIC/LABORATORY ...................................... 40
MUSCULOSKELETAL/SOFT TISSUE........................ 43
NEUROLOGY ............................................................. 47
OCULAR/VISUAL........................................................ 52
PAIN............................................................................ 55
PULMONARY/UPPER RESPIRATORY...................... 56
RENAL/GENITOURINARY.......................................... 60
SECONDARY MALIGNANCY ..................................... 63
SEXUAL/REPRODUCTIVE FUNCTION ..................... 64
SURGERY/INTRA-OPERATIVE INJURY ................... 66
SYNDROMES ............................................................. 68
VASCULAR................................................................. 70

ALLERGY/IMMUNOLOGY Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 1 - March 31, 2003, Publish Date: August 9, 2006
Allergic reaction/
hypersensitivity
(including drug fever)
Allergic reaction Transient flushing or
rash; drug fever <38°C
(<100.4°F)
Rash; flushing; urticaria;
dyspnea; drug fever
≥38°C (≥100.4°F)
Symptomatic
bronchospasm, with or
without urticaria;
parenteral medication(s)
indicated; allergy-related
edema/angioedema;
hypotension
Anaphylaxis Death
REMARK: Urticaria with manifestations of allergic or hypersensitivity reaction is graded as Allergic reaction/hypersensitivity (including drug fever).
A
LSO CONSIDER: Cytokine release syndrome/acute infusion reaction.
Allergic rhinitis
(including sneezing,
nasal stuffiness,
postnasal drip)
Rhinitis Mild, intervention not
indicated
Moderate, intervention
indicated
— — —
REMARK: Rhinitis associated with obstruction or stenosis is graded as Obstruction/stenosis of airway – Select in the PULMONARY/UPPER RESPIRATORY CATEGORY.
Autoimmune reaction Autoimmune reaction Asymptomatic and
serologic or other
evidence of autoimmune
reaction, with normal
organ function and
intervention not indicated
Evidence of autoimmune
reaction involving a non-
essential organ or
function (e.g.,
hypothyroidism)
Reversible autoimmune
reaction involving function
of a major organ or other
adverse event (e.g.,
transient colitis or
anemia)
Autoimmune reaction with
life-threatening
consequences
Death
ALSO CONSIDER: Colitis; Hemoglobin; Hemolysis (e.g., immune hemolytic anemia, drug-related hemolysis); Thyroid function, low (hypothyroidism).
Serum sickness Serum sickness — — Present — Death
NAVIGATION NOTE: Splenic function is graded in the BLOOD/BONE MARROW CATEGORY.
NAVIGATION NOTE: Urticaria as an isolated symptom is graded as Urticaria (hives, welts, wheals) in the DERMATOLOGY/SKIN CATEGORY.
Vasculitis Vasculitis Mild, intervention not
indicated
Symptomatic, non-
steroidal medical
intervention indicated
Steroids indicated Ischemic changes;
amputation indicated
Death
Allergy/Immunology –
Other (Specify, __)
Allergy – Other (Specify) Mild Moderate Severe Life-threatening; disabling Death

AUDITORY/EAR Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 2 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Earache (otalgia) is graded as Pain – Select in the PAIN CATEGORY.
Hearing:
patients with/without
baseline audiogram and
enrolled in a monitoring
program
1
Hearing (monitoring
program)
Threshold shift or loss of
15 – 25 dB relative to
baseline, averaged at 2
or more contiguous test
frequencies in at least
one ear; or subjective
change in the absence of
a Grade 1 threshold shift
Threshold shift or loss of
>25 – 90 dB, averaged at
2 contiguous test
frequencies in at least
one ear
Adult only: Threshold shift
of >25 – 90 dB, averaged
at 3 contiguous test
frequencies in at least
one ear
Pediatric:
Hearing loss sufficient to
indicate therapeutic
intervention, including
hearing aids (e.g., ≥20 dB
bilateral HL in the speech
frequencies; ≥30 dB
unilateral HL; and
requiring additional
speech-language related
services)
Adult only: Profound
bilateral hearing loss
(>90 dB)
Pediatric:
Audiologic indication for
cochlear implant and
requiring additional
speech-language related
services
—
REMARK: Pediatric recommendations are identical to those for adults, unless specified. For children and adolescents (≤18 years of age) without a baseline test, pre-exposure/pre-
treatment hearing should be considered to be <5 dB loss.
Hearing:
patients without baseline
audiogram and not
enrolled in a monitoring
program
1
Hearing (without
monitoring program)
— Hearing loss not requiring
hearing aid or
intervention (i.e., not
interfering with ADL)
Hearing loss requiring
hearing aid or
intervention (i.e.,
interfering with ADL)
Profound bilateral hearing
loss (>90 dB)
—
REMARK: Pediatric recommendations are identical to those for adults, unless specified. For children and adolescents (≤18 years of age) without a baseline test, pre-exposure/pre-
treatment hearing should be considered to be <5 dB loss.
Otitis, external ear
(non-infectious)
Otitis, external External otitis with
erythema or dry
desquamation
External otitis with moist
desquamation, edema,
enhanced cerumen or
discharge; tympanic
membrane perforation;
tympanostomy
External otitis with
mastoiditis; stenosis or
osteomyelitis
Necrosis of soft tissue or
bone
Death
ALSO CONSIDER: Hearing: patients with/without baseline audiogram and enrolled in a monitoring program
1
; Hearing: patients without baseline audiogram and not enrolled in a
monitoring program
1
.
Otitis, middle ear
(non-infectious)
Otitis, middle Serous otitis Serous otitis, medical
intervention indicated
Otitis with discharge;
mastoiditis
Necrosis of the canal soft
tissue or bone
Death

AUDITORY/EAR Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 3 - March 31, 2003, Publish Date: August 9, 2006
Tinnitus Tinnitus — Tinnitus not interfering
with ADL
Tinnitus interfering with
ADL
Disabling —
ALSO CONSIDER: Hearing: patients with/without baseline audiogram and enrolled in a monitoring program
1
; Hearing: patients without baseline audiogram and not enrolled in a
monitoring program
1
.
Auditory/Ear – Other
(Specify, __)
Auditory/Ear – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
1
Drug-induced ototoxicity should be distinguished from age-related threshold decrements or unrelated cochlear insult. When considering whether an adverse event has occurred, it is
first necessary to classify the patient into one of two groups. (1) The patient is under standard treatment/enrolled in a clinical trial <2.5 years, and has a 15 dB or greater threshold
shift averaged across two contiguous frequencies; or (2) The patient is under standard treatment/enrolled in a clinical trial >2.5 years, and the difference between the expected age-
related and the observed threshold shifts is 15 dB or greater averaged across two contiguous frequencies. Consult standard references for appropriate age- and gender-specific
hearing norms, e.g., Morrell, et al. Age- and gender-specific reference ranges for hearing level and longitudinal changes in hearing level. Journal of the Acoustical Society of America
100:1949-1967, 1996; or Shotland, et al. Recommendations for cancer prevention trials using potentially ototoxic test agents. Journal of Clinical Oncology 19:1658-1663, 2001.
In the absence of a baseline prior to initial treatment, subsequent audiograms should be referenced to an appropriate database of normals. ANSI. (1996)
American National Standard: Determination of occupational noise exposure and estimation of noise-induced hearing impairment, ANSI S 3.44-1996. (Standard S 3.44). New York:
American National Standards Institute. The recommended ANSI S3.44 database is Annex B.
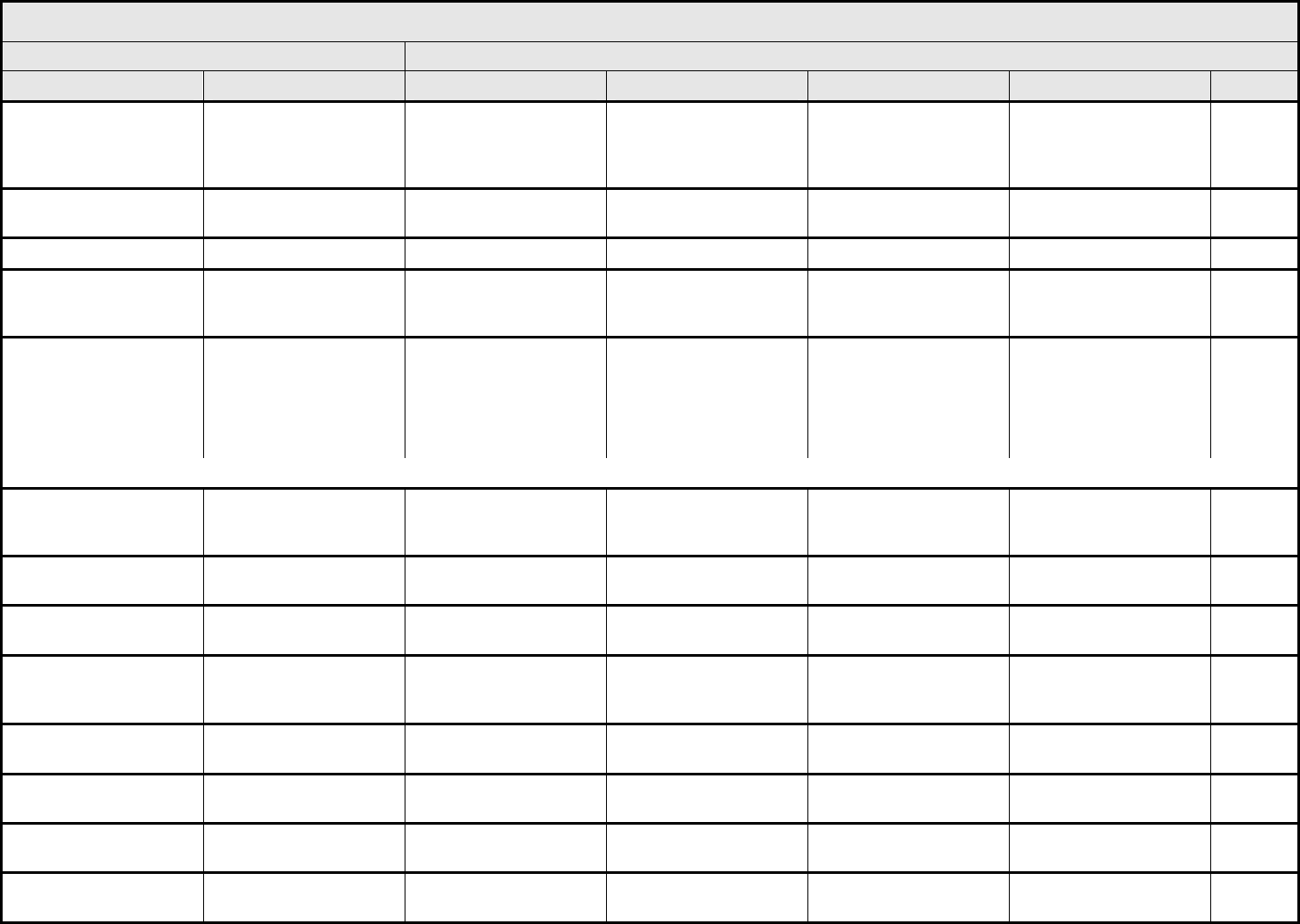
BLOOD/BONE MARROW Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 4 - March 31, 2003, Publish Date: August 9, 2006
Bone marrow cellularity Bone marrow cellularity Mildly hypocellular or
≤25% reduction from
normal cellularity for age
Moderately hypocellular
or >25 – ≤50% reduction
from normal cellularity for
age
Severely hypocellular or
>50 – ≤75% reduction
cellularity from normal for
age
— Death
CD4 count CD4 count <LLN – 500/mm
3
<LLN – 0.5 x 10
9
/L
<500 – 200/mm
3
<0.5 – 0.2 x 10
9
/L
<200 – 50/mm
3
<0.2 x 0.05 – 10
9
/L
<50/mm
3
<0.05 x
10
9
/L
Death
Haptoglobin Haptoglobin <LLN — Absent — Death
Hemoglobin Hemoglobin <LLN – 10.0 g/dL
<LLN – 6.2 mmol/L
<LLN – 100 g/L
<10.0 – 8.0 g/dL
<6.2 – 4.9 mmol/L
<100 – 80g/L
<8.0 – 6.5 g/dL
<4.9 – 4.0 mmol/L
<80 – 65 g/L
<6.5 g/dL
<4.0 mmol/L
<65 g/L
Death
Hemolysis (e.g., immune
hemolytic anemia, drug-
related hemolysis)
Hemolysis Laboratory evidence of
hemolysis only (e.g.,
direct antiglobulin test
[DAT, Coombs’]
schistocytes)
Evidence of red cell
destruction and ≥2 gm
decrease in hemoglobin,
no transfusion
Transfusion or medical
intervention (e.g.,
steroids) indicated
Catastrophic
consequences of
hemolysis (e.g., renal
failure, hypotension,
bronchospasm,
emergency splenectomy)
Death
ALSO CONSIDER: Haptoglobin; Hemoglobin.
Iron overload Iron overload — Asymptomatic iron
overload, intervention not
indicated
Iron overload,
intervention indicated
Organ impairment (e.g.,
endocrinopathy,
cardiopathy)
Death
Leukocytes (total WBC) Leukocytes <LLN – 3000/mm
3
<LLN – 3.0 x 10
9
/L
<3000
–
2000/mm
3
<3.0 – 2.0 x 10
9
/L
<2000
– 1000/mm
3
<2.0 – 1.0 x 10
9
/L
<1000/mm
3
<1.0 x 10
9
/L
Death
Lymphopenia Lymphopenia <LLN – 800/mm
3
<LLN x 0.8 – 10
9
/L
<800
– 500/mm
3
<0.8 – 0.5 x 10
9
/L
<500 – 200 mm
3
<0.5 – 0.2 x 10
9
/L
<200/mm
3
<0.2 x 10
9
/L
Death
Myelodysplasia Myelodysplasia — — Abnormal marrow
cytogenetics (marrow
blasts ≤5%)
RAEB or RAEB-T
(marrow blasts >5%)
Death
Neutrophils/granulocytes
(ANC/AGC)
Neutrophils <LLN – 1500/mm
3
<LLN – 1.5 x 10
9
/L
<1500
– 1000/mm
3
<1.5 – 1.0 x 10
9
/L
<1000 – 500/mm
3
<1.0 – 0.5 x 10
9
/L
<500/mm
3
<0.5 x 10
9
/L
Death
Platelets Platelets <LLN – 75,000/mm
3
<LLN – 75.0 x 10
9
/L
<75,000 – 50,000/mm
3
<75.0 – 50.0 x 10
9
/L
<50,000 – 25,000/mm
3
<50.0 – 25.0 x 10
9
/L
<25,000/mm
3
<25.0 x 10
9
/L
Death
Splenic function Splenic function Incidental findings (e.g.,
Howell-Jolly bodies)
Prophylactic antibiotics
indicated
— Life-threatening
consequences
Death
Blood/Bone Marrow –
Other (Specify, __)
Blood – Other (Specify) Mild Moderate Severe Life-threatening; disabling Death

CARDIAC ARRHYTHMIA Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 5 - March 31, 2003, Publish Date: August 9, 2006
Conduction abnormality/
atrioventricular heart
block
– Select:
Conduction abnormality
– Select
Asymptomatic,
intervention not indicated
– Asystole
– AV Block-First degree
– AV Block-Second degree Mobitz Type I (Wenckebach)
– AV Block-Second degree Mobitz Type II
– AV Block-Third degree (Complete AV block)
– Conduction abnormality NOS
– Sick Sinus Syndrome
– Stokes-Adams Syndrome
– Wolff-Parkinson-White Syndrome
Non-urgent medical
intervention indicated
Incompletely controlled
medically or controlled
with device (e.g.,
pacemaker)
Life-threatening (e.g.,
arrhythmia associated
with CHF, hypotension,
syncope, shock)
Death
Palpitations Palpitations Present Present with associated
symptoms (e.g.,
lightheadedness,
shortness of breath)
— — —
REMARK: Grade palpitations only in the absence of a documented arrhythmia.
Prolonged QTc interval Prolonged QTc QTc >0.45 – 0.47 second QTc >0.47 – 0.50
second; ≥0.06 second
above baseline
QTc >0.50 second QTc >0.50 second; life-
threatening signs or
symptoms (e.g.,
arrhythmia, CHF,
hypotension, shock
syncope); Torsade de
pointes
Death
Supraventricular and
nodal arrhythmia
– Select:
Supraventricular
arrhythmia – Select
Asymptomatic,
intervention not indicated
Non-urgent medical
intervention indicated
– Atrial fibrillation
– Atrial flutter
– Atrial tachycardia/Paroxysmal Atrial Tachycardia
– Nodal/Junctional
– Sinus arrhythmia
– Sinus bradycardia
– Sinus tachycardia
– Supraventricular arrhythmia NOS
– Supraventricular extrasystoles (Premature Atrial Contractions; Premature Nodal/Junctional Contractions)
– Supraventricular tachycardia
Symptomatic and
incompletely controlled
medically, or controlled
with device (e.g.,
pacemaker)
Life-threatening (e.g.,
arrhythmia associated
with CHF, hypotension,
syncope, shock)
Death
NAVIGATION NOTE: Syncope is graded as Syncope (fainting) in the NEUROLOGY CATEGORY.
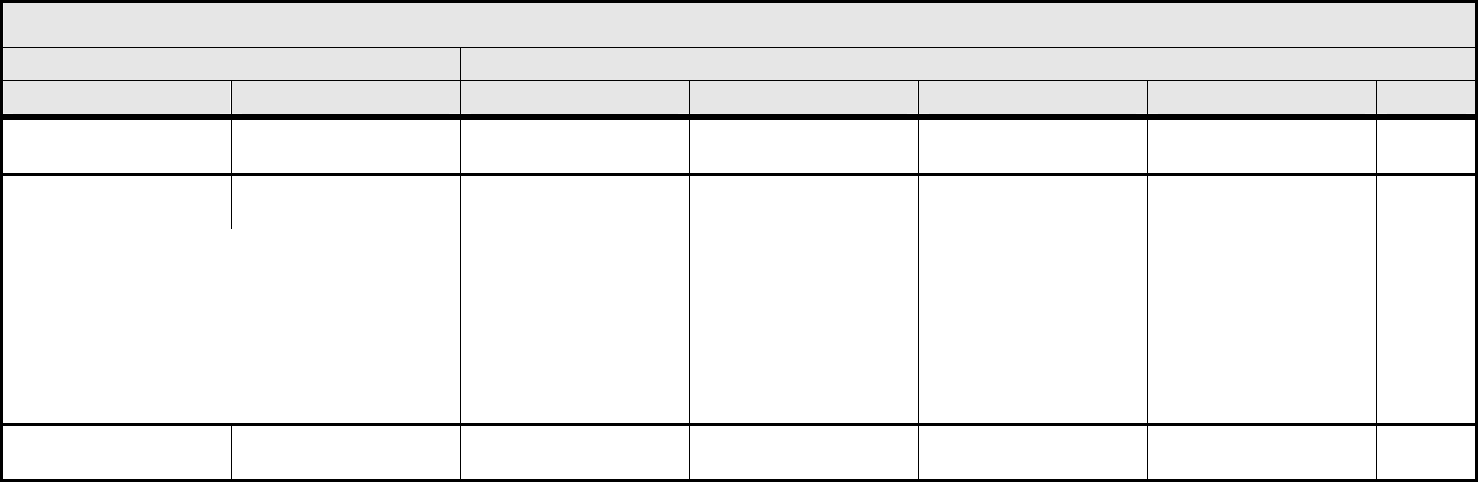
CARDIAC ARRHYTHMIA Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 6 - March 31, 2003, Publish Date: August 9, 2006
Vasovagal episode Vasovagal episode — Present without loss of
consciousness
Present with loss of
consciousness
Life-threatening
consequences
Death
Ventricular arrhythmia
– Select:
Ventricular arrhythmia
– Select
– Bigeminy
– Idioventricular rhythm
– PVCs
– Torsade de pointes
– Trigeminy
– Ventricular arrhythmia NOS
– Ventricular fibrillation
– Ventricular flutter
– Ventricular tachycardia
Asymptomatic, no
intervention indicated
Non-urgent medical
intervention indicated
Symptomatic and
incompletely controlled
medically or controlled
with device (e.g.,
defibrillator)
Life-threatening (e.g.,
arrhythmia associated
with CHF, hypotension,
syncope, shock)
Death
Cardiac Arrhythmia
– Other (Specify, __)
Cardiac Arrhythmia –
Other (Specify)
Mild Moderate Severe Life-threatening;
disabling
Death
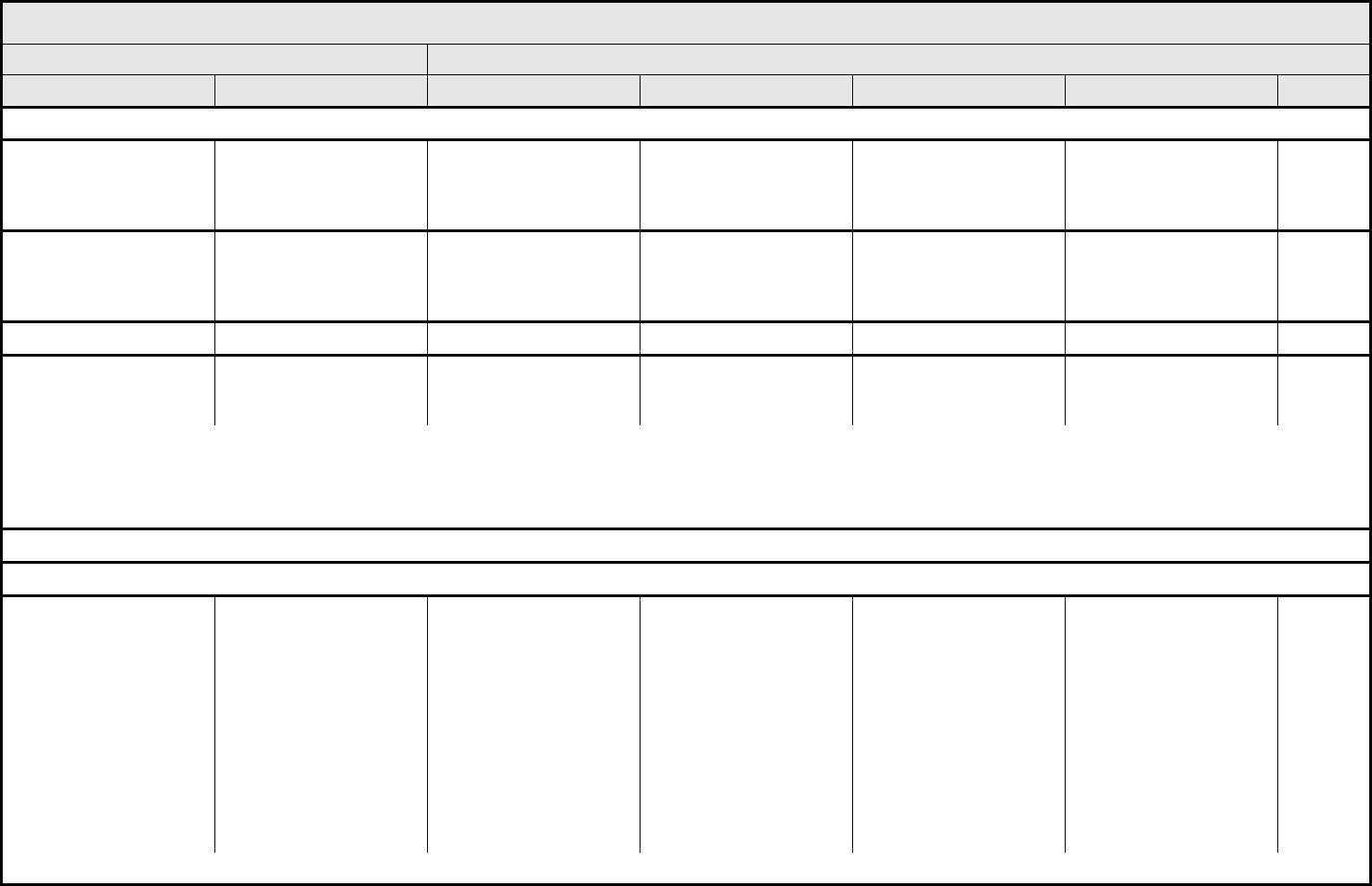
CARDIAC GENERAL Page 1 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 7 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Angina is graded as Cardiac ischemia/infarction in the CARDIAC GENERAL CATEGORY.
Cardiac
ischemia/infarction
Cardiac
ischemia/infarction
Asymptomatic arterial
narrowing without
ischemia
Asymptomatic and testing
suggesting ischemia;
stable angina
Symptomatic and testing
consistent with ischemia;
unstable angina;
intervention indicated
Acute myocardial
infarction
Death
Cardiac troponin I (cTnI) cTnI — — Levels consistent with
unstable angina as
defined by the
manufacturer
Levels consistent with
myocardial infarction as
defined by the
manufacturer
Death
Cardiac troponin T (cTnT) cTnT 0.03 – <0.05 ng/mL 0.05 – <0.1 ng/mL 0.1 – <0.2 ng/mL 0.2 ng/mL Death
Cardiopulmonary arrest,
cause unknown
(non-fatal)
Cardiopulmonary arrest — — — Life-threatening —
REMARK: Grade 4 (non-fatal) is the only appropriate grade. CTCAE provides three alternatives for reporting Death:
1. A CTCAE term associated with Grade 5.
2. A CTCAE 'Other (Specify, __)’ within any CATEGORY.
3. Death not associated with CTCAE term – Select in the DEATH CATEGORY.
NAVIGATION NOTE: Chest pain (non-cardiac and non-pleuritic) is graded as Pain – Select in the PAIN CATEGORY.
NAVIGATION NOTE: CNS ischemia is graded as CNS cerebrovascular ischemia in the NEUROLOGY CATEGORY.
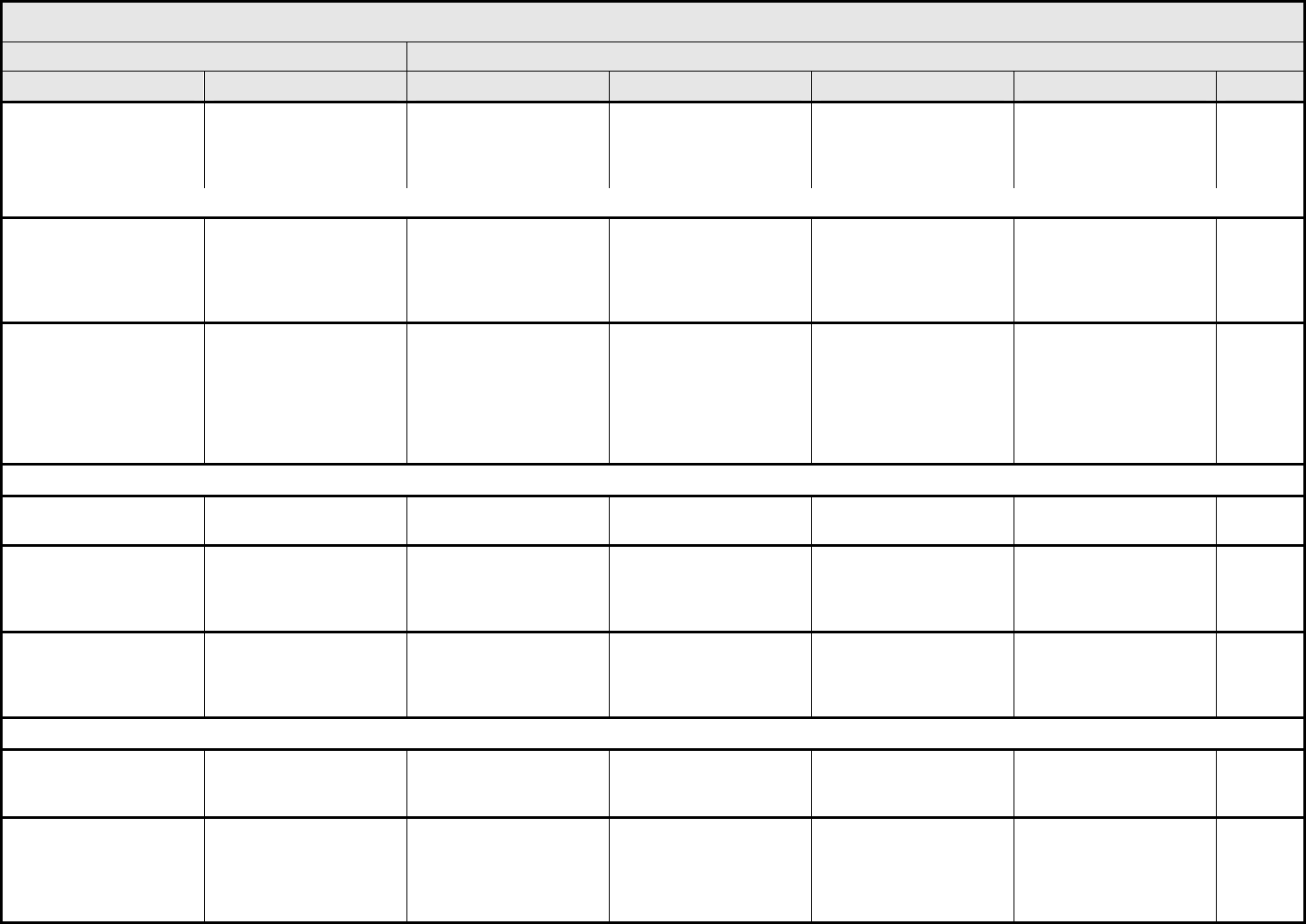
Hypertension Hypertension Asymptomatic, transient
(<24 hrs) increase by >20
mmHg (diastolic) or to
>150/100 if previously
WNL; intervention not
indicated
Pediatric:
Asymptomatic, transient
(<24 hrs) BP increase
>ULN; intervention not
indicated
Recurrent or persistent
(≥24 hrs) or symptomatic
increase by >20 mmHg
(diastolic) or to >150/100
if previously WNL;
monotherapy may be
indicated
Pediatric:
Recurrent or persistent
(≥24 hrs) BP >ULN;
monotherapy may be
indicated
Requiring more than one
drug or more intensive
therapy than previously
Pediatric:
Same as adult
Life-threatening
consequences (e.g.,
hypertensive crisis)
Pediatric:
Same as adult
Death
REMARK: Use age and gender-appropriate normal values >95
th
percentile ULN for pediatric patients.
CARDIAC GENERAL Page 2 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 8 - March 31, 2003, Publish Date: August 9, 2006
Hypotension Hypotension Changes, intervention not
indicated
Brief (<24 hrs) fluid
replacement or other
therapy; no physiologic
consequences
Sustained (≥24 hrs)
therapy, resolves without
persisting physiologic
consequences
Shock (e.g., acidemia;
impairment of vital organ
function)
Death
ALSO CONSIDER: Syncope (fainting).
Left ventricular diastolic
dysfunction
Left ventricular diastolic
dysfunction
Asymptomatic diagnostic
finding; intervention not
indicated
Asymptomatic,
intervention indicated
Symptomatic CHF
responsive to intervention
Refractory CHF, poorly
controlled; intervention
such as ventricular assist
device or heart transplant
indicated
Death
Left ventricular systolic
dysfunction
Left ventricular systolic
dysfunction
Asymptomatic, resting
ejection fraction (EF)
<60 – 50%; shortening
fraction (SF) <30 – 24%
Asymptomatic, resting
EF <50 – 40%;
SF <24 – 15%
Symptomatic CHF
responsive to
intervention;
EF <40 – 20%
SF <15%
Refractory CHF or poorly
controlled; EF <20%;
intervention such as
ventricular assist device,
ventricular reduction
surgery, or heart
transplant indicated
Death
NAVIGATION NOTE: Myocardial infarction is graded as Cardiac ischemia/infarction in the CARDIAC GENERAL CATEGORY.
Myocarditis Myocarditis — — CHF responsive to
intervention
Severe or refractory CHF Death
Pericardial effusion
(non-malignant)
Pericardial effusion Asymptomatic effusion — Effusion with physiologic
consequences
Life-threatening
consequences (e.g.,
tamponade); emergency
intervention indicated
Death
Pericarditis Pericarditis Asymptomatic, ECG or
physical exam (rub)
changes consistent with
pericarditis
Symptomatic pericarditis
(e.g., chest pain)
Pericarditis with
physiologic
consequences (e.g.,
pericardial constriction)
Life-threatening
consequences;
emergency intervention
indicated
Death
NAVIGATION NOTE: Pleuritic pain is graded as Pain – Select in the PAIN CATEGORY.
Pulmonary hypertension Pulmonary hypertension Asymptomatic without
therapy
Asymptomatic, therapy
indicated
Symptomatic
hypertension, responsive
to therapy
Symptomatic
hypertension, poorly
controlled
Death
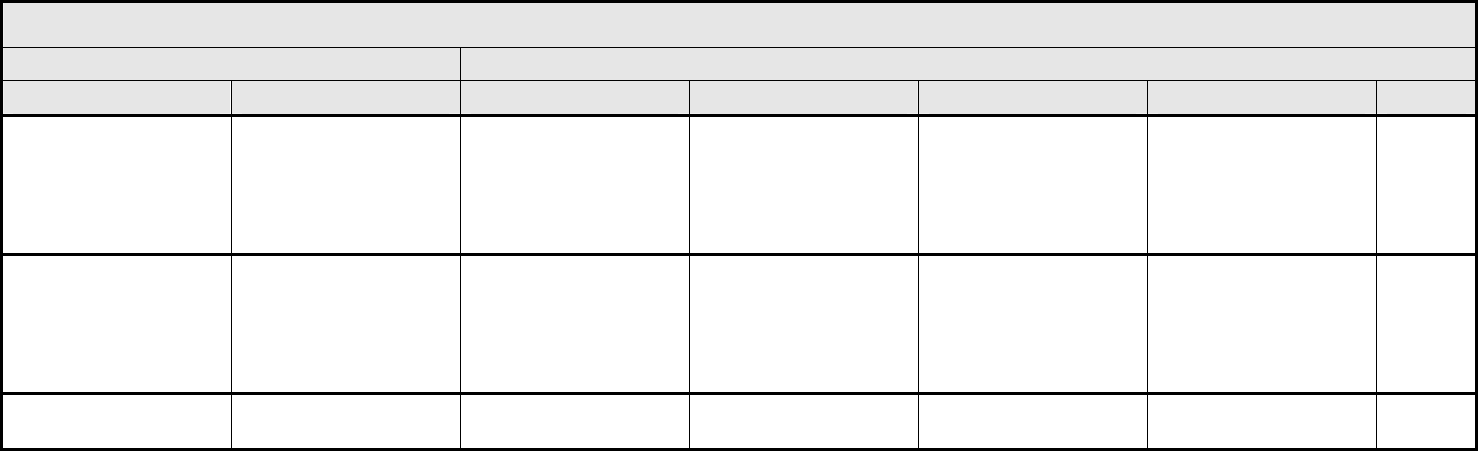
Restrictive
cardiomyopathy
Restrictive
cardiomyopathy
Asymptomatic, therapy
not indicated
Asymptomatic, therapy
indicated
Symptomatic CHF
responsive to intervention
Refractory CHF, poorly
controlled; intervention
such as ventricular assist
device, or heart
transplant indicated
Death
CARDIAC GENERAL Page 3 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 9 - March 31, 2003, Publish Date: August 9, 2006
Right ventricular
dysfunction
(cor pulmonale)
Right ventricular
dysfunction
Asymptomatic without
therapy
Asymptomatic, therapy
indicated
Symptomatic cor
pulmonale, responsive to
intervention
Symptomatic cor
pulmonale poorly
controlled; intervention
such as ventricular assist
device, or heart
transplant indicated
Death
Valvular heart disease Valvular heart disease Asymptomatic valvular
thickening with or without
mild valvular regurgitation
or stenosis; treatment
other than endocarditis
prophylaxis not indicated
Asymptomatic; moderate
regurgitation or stenosis
by imaging
Symptomatic; severe
regurgitation or stenosis;
symptoms controlled with
medical therapy
Life-threatening;
disabling; intervention
(e.g., valve replacement,
valvuloplasty) indicated
Death
Cardiac General – Other
(Specify, __)
Cardiac General – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
COAGULATION Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 10 - March 31, 2003, Publish Date: August 9, 2006
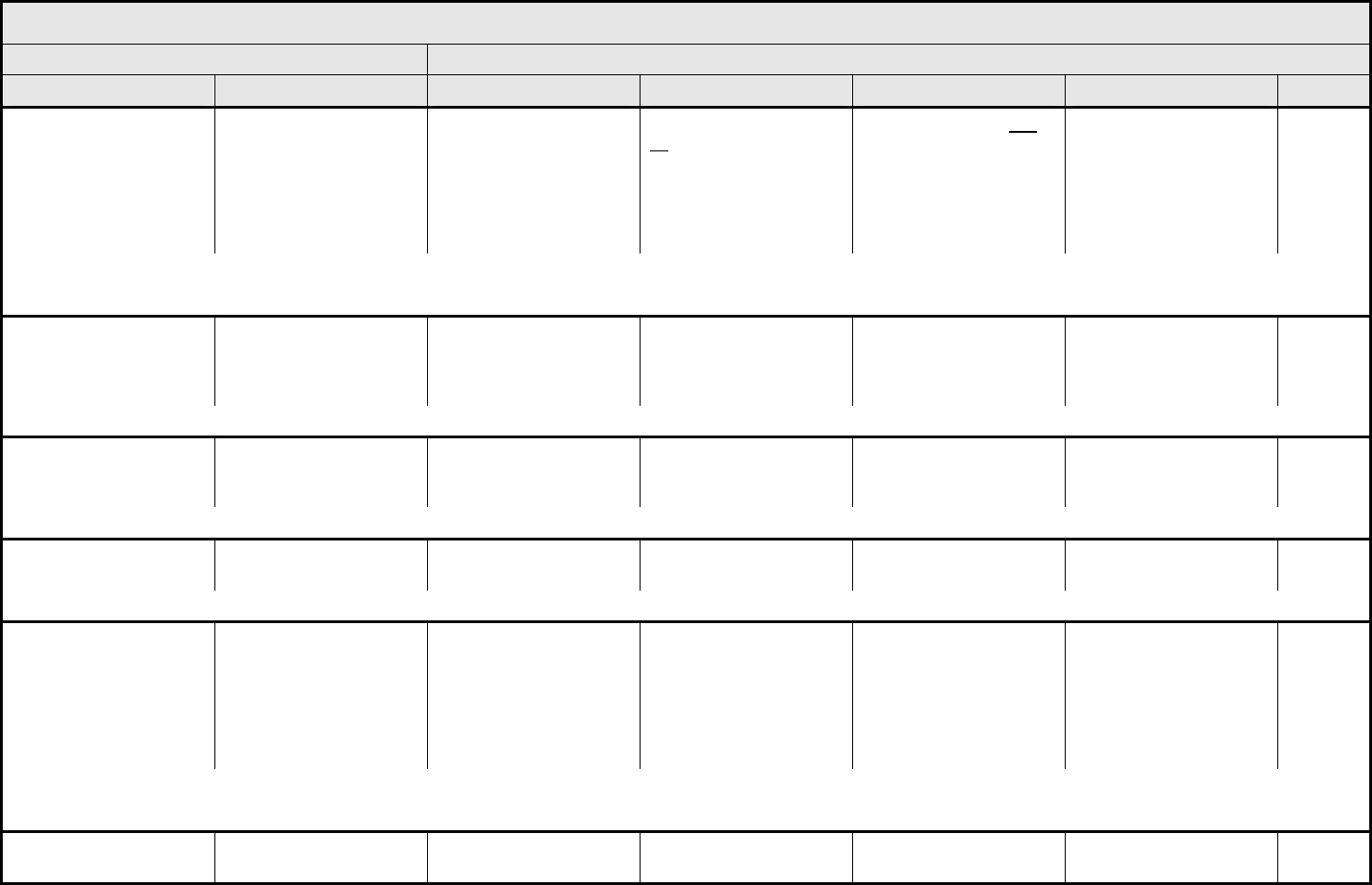
DIC (disseminated
intravascular coagulation)
DIC — Laboratory findings with
no
bleeding
Laboratory findings and
bleeding
Laboratory findings, life-
threatening or disabling
consequences (e.g., CNS
hemorrhage, organ
damage, or
hemodynamically
significant blood loss)
Death
REMARK: DIC (disseminated intravascular coagulation) must have increased fibrin split products or D-dimer.
A
LSO CONSIDER: Platelets.
Fibrinogen Fibrinogen <1.0 – 0.75 x LLN
or <25% decrease from
baseline
<0.75 – 0.5 x LLN
or 25 – <50% decrease
from baseline
<0.5 – 0.25 x LLN
or 50 – <75% decrease
from baseline
<0.25 x LLN
or 75% decrease from
baseline or absolute
value <50 mg/dL
Death
REMARK: Use % decrease only when baseline is <LLN (local laboratory value).
INR (International
Normalized Ratio of
prothrombin time)
INR
>1 – 1.5 x ULN >1.5 – 2 x ULN >2 x ULN — —
ALSO CONSIDER: Hemorrhage, CNS; Hemorrhage, GI – Select; Hemorrhage, GU – Select; Hemorrhage, pulmonary/upper respiratory – Select.
PTT (Partial
Thromboplastin Time)
PTT >1 – 1.5 x ULN >1.5 – 2 x ULN >2 x ULN — —
ALSO CONSIDER: Hemorrhage, CNS; Hemorrhage, GI – Select; Hemorrhage, GU – Select; Hemorrhage, pulmonary/upper respiratory – Select.
Thrombotic
microangiopathy (e.g.,
thrombotic
thrombocytopenic
purpura [TTP] or
hemolytic uremic
syndrome [HUS])
Thrombotic
microangiopathy
Evidence of RBC
destruction
(schistocytosis) without
clinical consequences
— Laboratory findings
present with clinical
consequences (e.g., renal
insufficiency, petechiae)
Laboratory findings and
life-threatening or
disabling consequences,
(e.g., CNS hemorrhage/
bleeding or thrombosis/
embolism or renal failure)
Death
REMARK: Must have microangiopathic changes on blood smear (e.g., schistocytes, helmet cells, red cell fragments).
ALSO CONSIDER: Creatinine; Hemoglobin; Platelets.
Coagulation – Other
(Specify, __)
Coagulation – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
CONSTITUTIONAL SYMPTOMS Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 11 - March 31, 2003, Publish Date: August 9, 2006
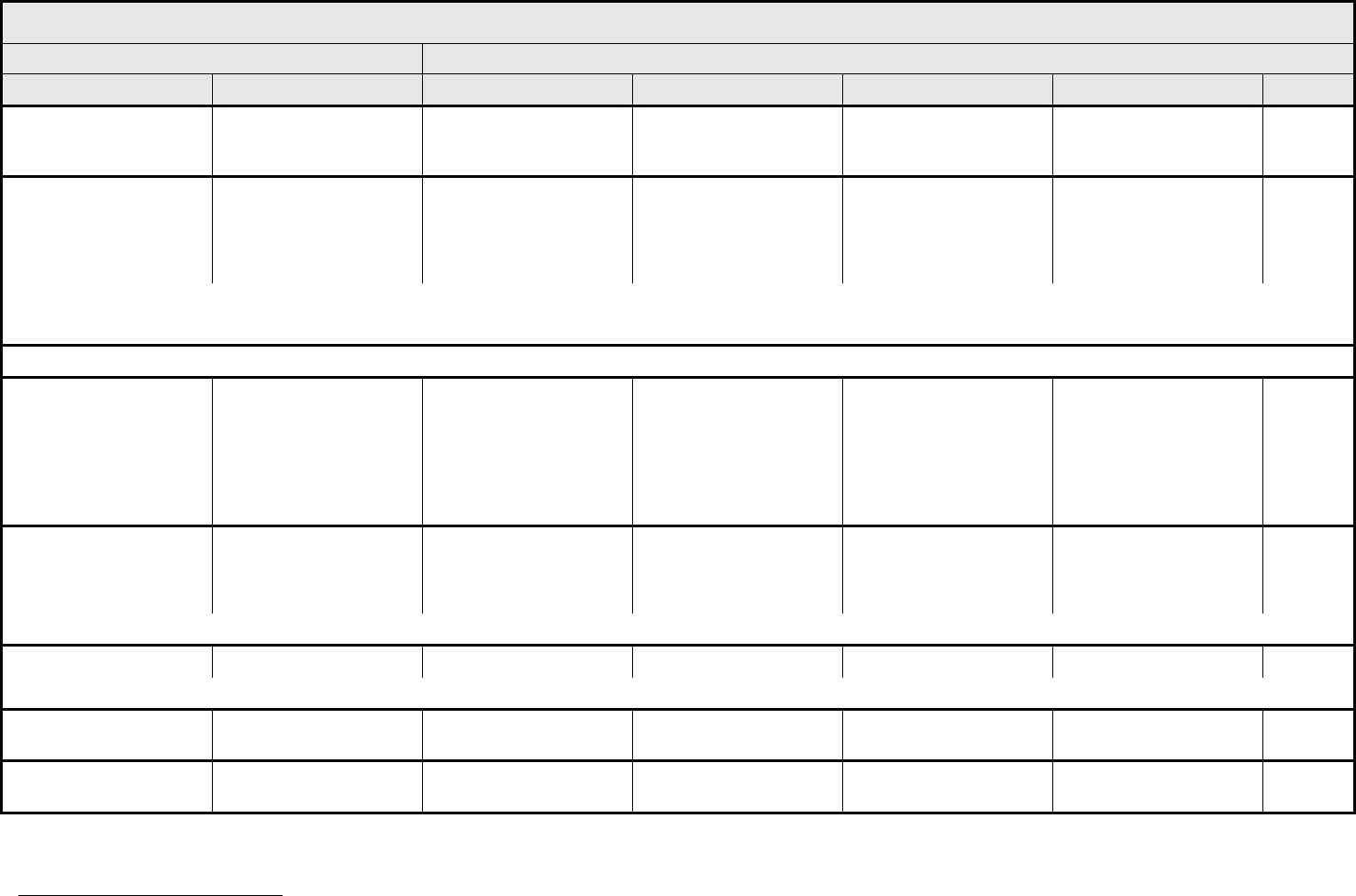
Fatigue
(asthenia, lethargy,
malaise)
Fatigue Mild fatigue over baseline Moderate or causing
difficulty performing some
ADL
Severe fatigue interfering
with ADL
Disabling —
Fever
(in the absence of
neutropenia, where
neutropenia is defined as
ANC <1.0 x 10
9
/L)
Fever 38.0 – 39.0°C
(100.4 – 102.2°F)
>39.0 – 40.0°C
(102.3 – 104.0°F)
>40.0°C
(>104.0°F) for ≤24 hrs
>40.0°C
(>104.0°F) for >24 hrs
Death
REMARK: The temperature measurements listed are oral or tympanic.
ALSO CONSIDER: Allergic reaction/hypersensitivity (including drug fever).
NAVIGATION NOTE: Hot flashes are graded as Hot flashes/flushes in the ENDOCRINE CATEGORY.
Hypothermia Hypothermia — 35 – >32°C
95 – >89.6°F
32 – >28°C
89.6 – >82.4° F
≤28 °C
82.4°F or life-threatening
consequences (e.g.,
coma, hypotension,
pulmonary edema,
acidemia, ventricular
fibrillation)
Death
Insomnia Insomnia Occasional difficulty
sleeping, not interfering
with function
Difficulty sleeping,
interfering with function
but not interfering with
ADL
Frequent difficulty
sleeping, interfering with
ADL
Disabling —
REMARK: If pain or other symptoms interfere with sleep, do NOT grade as insomnia. Grade primary event(s) causing insomnia.
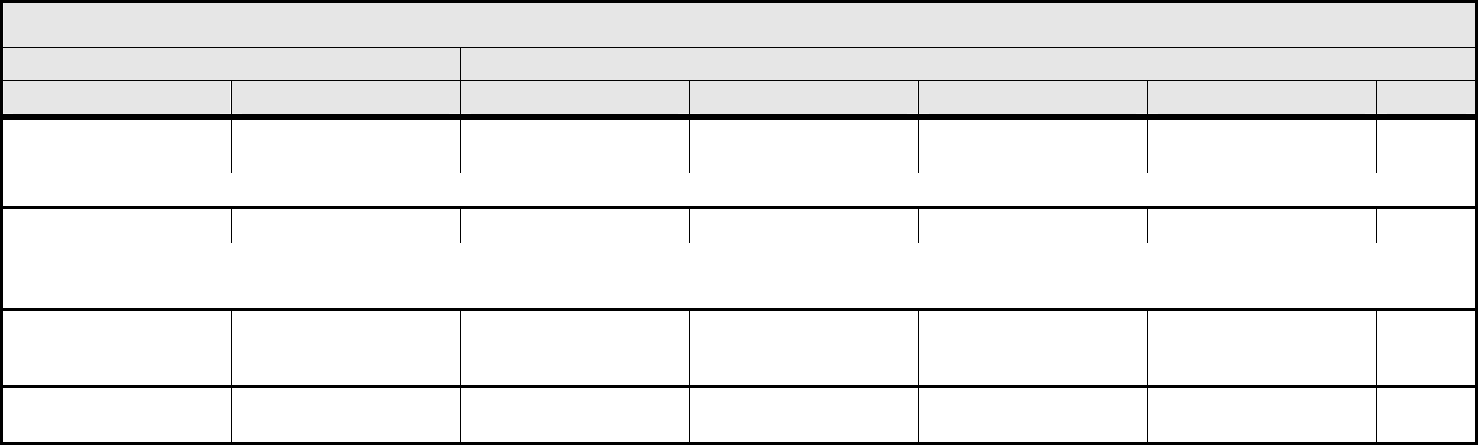
Obesity
2
Obesity — BMI 25 – 29.9 kg/m
2
BMI 30 – 39.99 kg/m
2
BMI ≥40 kg/m
2
—
REMARK: BMI = (weight [kg]) / (height [m])
2
Odor
(patient odor)
Patient odor Mild odor Pronounced odor — — —
Rigors/chills Rigors/chills Mild Moderate, narcotics
indicated
Severe or prolonged, not
responsive to narcotics
— —
2
NHLBI Obesity Task Force. "Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults," The Evidence Report, Obes Res 6:51S-
209S, 1998.
CONSTITUTIONAL SYMPTOMS Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 12 - March 31, 2003, Publish Date: August 9, 2006
Sweating
(diaphoresis)
Sweating Mild and occasional Frequent or drenching — — —
ALSO CONSIDER: Hot flashes/flushes.
Weight gain Weight gain 5 – <10% of baseline 10 – <20% of baseline
≥20% of baseline
— —
REMARK: Edema, depending on etiology, is graded in the CARDIAC GENERAL or LYMPHATICS CATEGORIES.
A
LSO CONSIDER: Ascites (non-malignant); Pleural effusion (non-malignant).
Weight loss Weight loss 5 to <10% from baseline;
intervention not indicated
10 – <20% from baseline;
nutritional support
indicated
≥20% from baseline; tube
feeding or TPN indicated
— —
Constitutional Symptoms
– Other (Specify, __)
Constitutional Symptoms
– Other (Specify)
Mild Moderate Severe Life-threatening; disabling Death
DEATH Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 13 - March 31, 2003, Publish Date: August 9, 2006
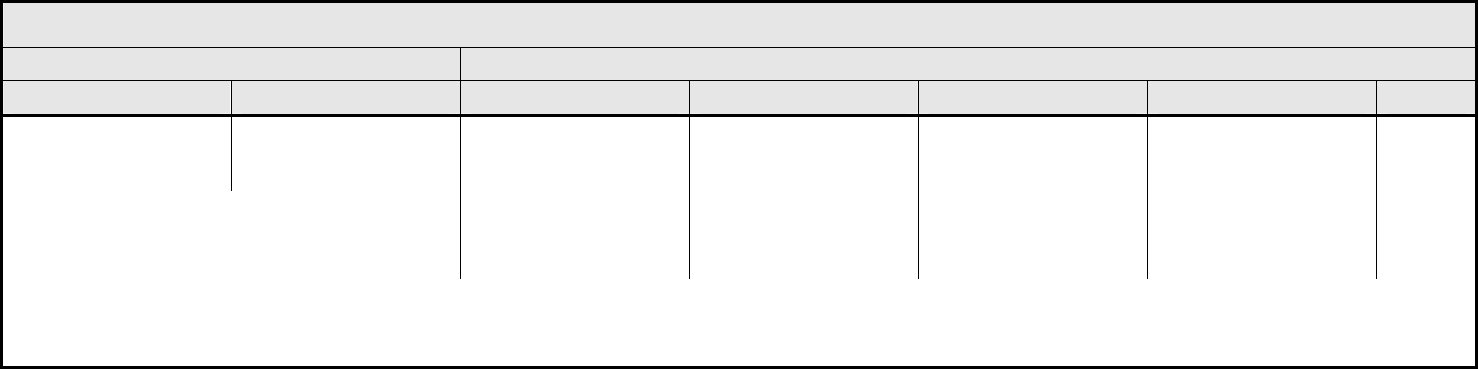
Death not associated with
CTCAE term
– Select:
Death not associated with
CTCAE term – Select
– Death NOS
– Disease progression NOS
– Multi-organ failure
– Sudden death
— — — — Death
REMARK: Grade 5 is the only appropriate grade. 'Death not associated with CTCAE term – Select' is to be used where a death:
1. Cannot be attributed to a CTCAE term associated with Grade 5.
2. Cannot be reported within any CATEGORY using a CTCAE 'Other (Specify, __)’.
DERMATOLOGY/SKIN Page 1 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 14 - March 31, 2003, Publish Date: August 9, 2006
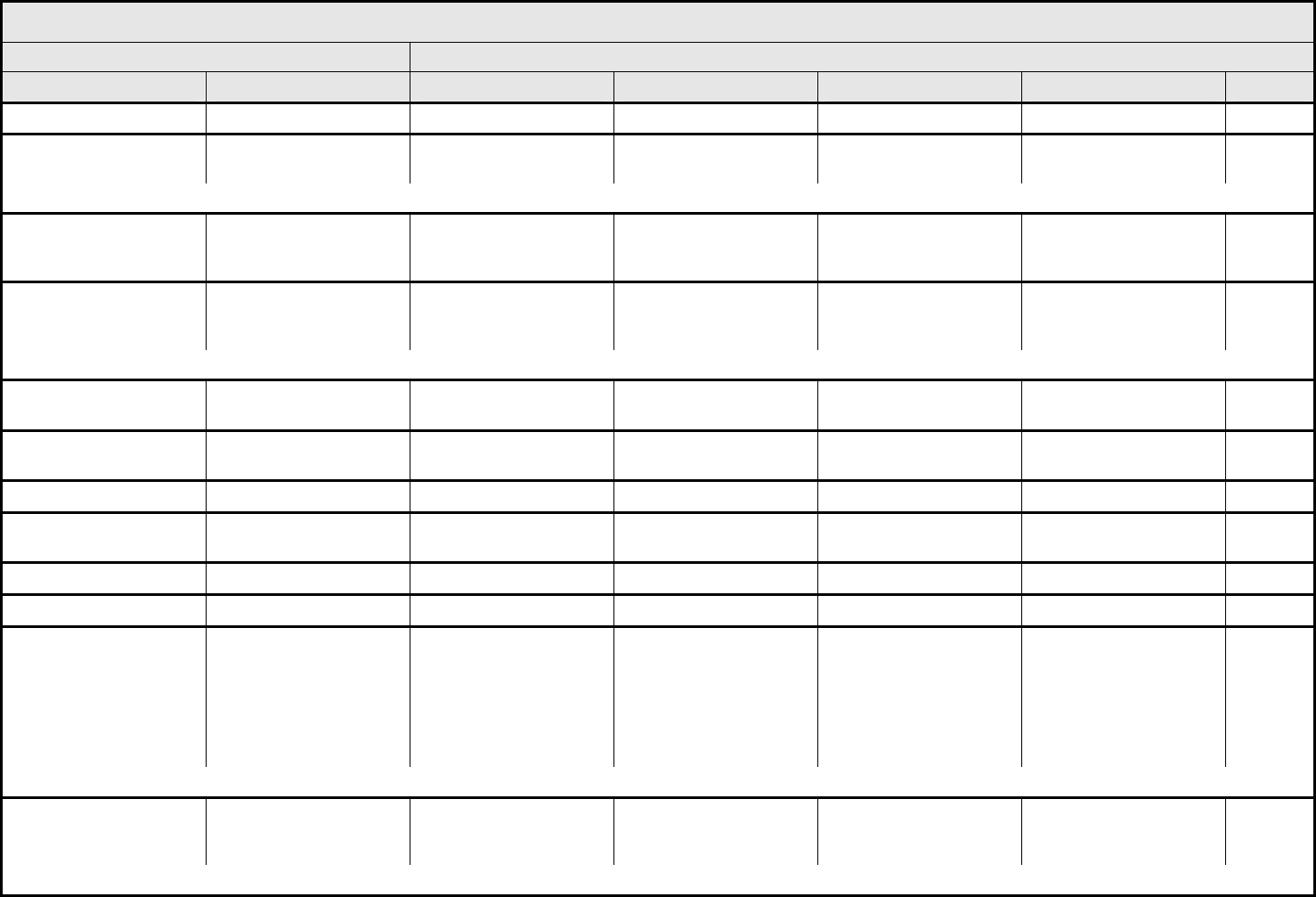
Atrophy, skin Atrophy, skin Detectable Marked — — —
Atrophy, subcutaneous
fat
Atrophy, subcutaneous
fat
Detectable Marked — — —
ALSO CONSIDER: Induration/fibrosis (skin and subcutaneous tissue).
Bruising
(in absence of Grade 3 or
4 thrombocytopenia)
Bruising Localized or in a
dependent area
Generalized — — —
Burn Burn Minimal symptoms;
intervention not indicated
Medical intervention;
minimal debridement
indicated
Moderate to major
debridement or
reconstruction indicated
Life-threatening
consequences
Death
REMARK: Burn refers to all burns including radiation, chemical, etc.
Cheilitis Cheilitis Asymptomatic Symptomatic, not
interfering with ADL
Symptomatic, interfering
with ADL
— —
Dry skin Dry skin Asymptomatic Symptomatic, not
interfering with ADL
Interfering with ADL — —
Flushing Flushing Asymptomatic Symptomatic — — —
Hair loss/alopecia
(scalp or body)
Alopecia Thinning or patchy Complete — — —
Hyperpigmentation Hyperpigmentation Slight or localized Marked or generalized — — —
Hypopigmentation Hypopigmentation Slight or localized Marked or generalized — — —
Induration/fibrosis
(skin and subcutaneous
tissue)
Induration Increased density on
palpation
Moderate impairment of
function not interfering
with ADL; marked
increase in density and
firmness on palpation
with or without minimal
retraction
Dysfunction interfering
with ADL; very marked
density, retraction or
fixation
— —
ALSO CONSIDER: Fibrosis-cosmesis; Fibrosis-deep connective tissue.
Injection site reaction/
extravasation changes
Injection site reaction Pain; itching; erythema Pain or swelling, with
inflammation or phlebitis
Ulceration or necrosis
that is severe; operative
intervention indicated
— —
ALSO CONSIDER: Allergic reaction/hypersensitivity (including drug fever); Ulceration.
DERMATOLOGY/SKIN Page 2 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 15 - March 31, 2003, Publish Date: August 9, 2006
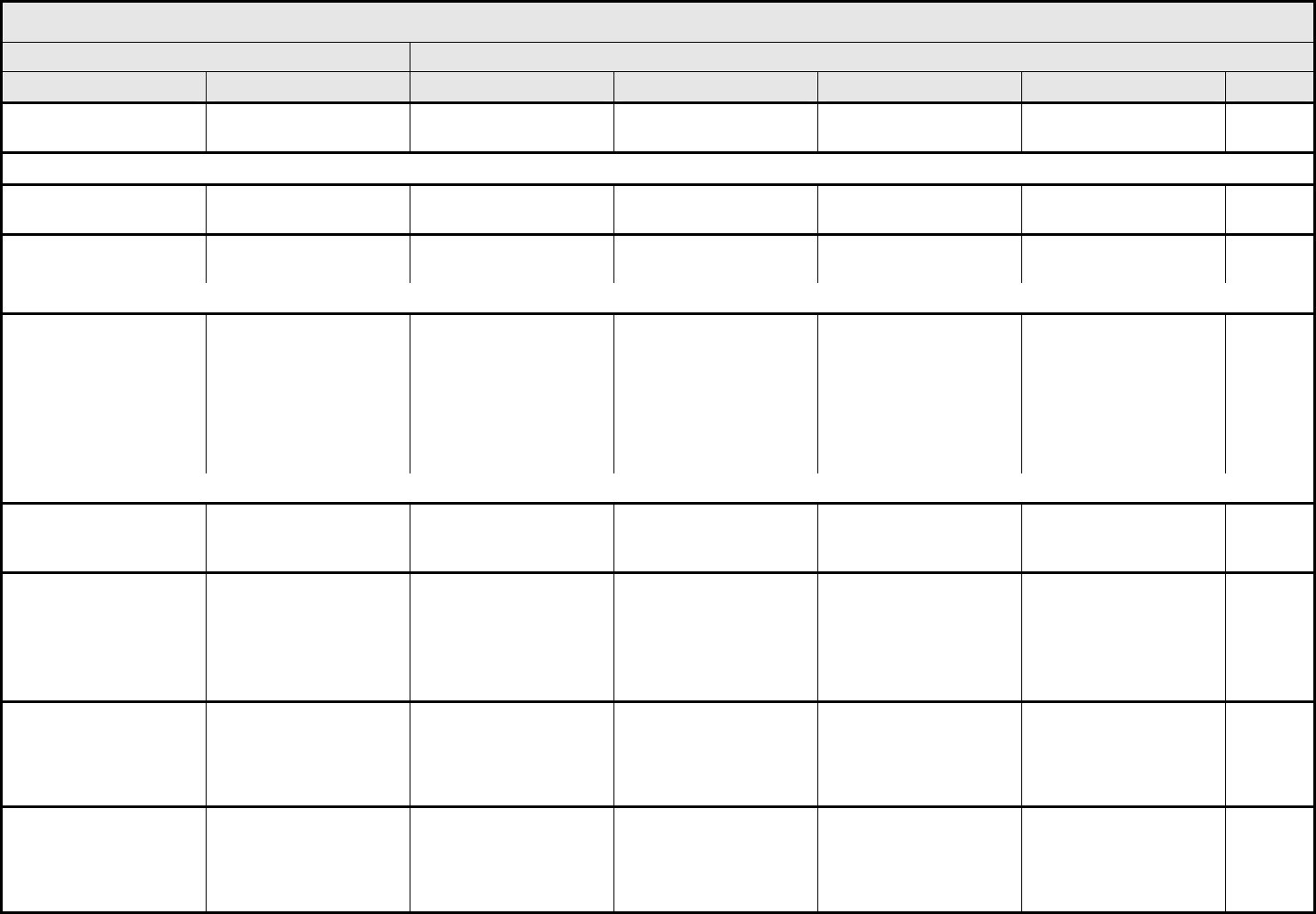
Nail changes Nail changes Discoloration; ridging
(koilonychias); pitting
Partial or complete loss of
nail(s); pain in nailbed(s)
Interfering with ADL — —
NAVIGATION NOTE: Petechiae is graded as Petechiae/purpura (hemorrhage/bleeding into skin or mucosa) in the HEMORRHAGE/BLEEDING CATEGORY.
Photosensitivity Photosensitivity Painless erythema Painful erythema Erythema with
desquamation
Life-threatening; disabling Death
Pruritus/itching Pruritus Mild or localized Intense or widespread Intense or widespread
and interfering with ADL
— —
ALSO CONSIDER: Rash/desquamation.
Rash/desquamation Rash Macular or papular
eruption or erythema
without associated
symptoms
Macular or papular
eruption or erythema with
pruritus or other
associated symptoms;
localized desquamation
or other lesions covering
<50% of body surface
area (BSA)
Severe, generalized
erythroderma or macular,
papular or vesicular
eruption; desquamation
covering ≥50% BSA
Generalized exfoliative,
ulcerative, or bullous
dermatitis
Death
REMARK: Rash/desquamation may be used for GVHD.
Rash:
acne/acneiform
Acne Intervention not indicated Intervention indicated Associated with pain,
disfigurement, ulceration,
or desquamation
— Death
Rash:
dermatitis associated with
radiation
– Select:
– Chemoradiation
– Radiation
Dermatitis – Select Faint erythema or dry
desquamation
Moderate to brisk
erythema; patchy moist
desquamation, mostly
confined to skin folds and
creases; moderate
edema
Moist desquamation other
than skin folds and
creases; bleeding
induced by minor trauma
or abrasion
Skin necrosis or
ulceration of full thickness
dermis; spontaneous
bleeding from involved
site
Death
Rash:
erythema multiforme
(e.g., Stevens-Johnson
syndrome, toxic
epidermal necrolysis)
Erythema multiforme — Scattered, but not
generalized eruption
Severe (e.g., generalized
rash or painful stomatitis);
IV fluids, tube feedings,
or TPN indicated
Life-threatening; disabling Death
Rash:
hand-foot skin reaction
Hand-foot Minimal skin changes or
dermatitis (e.g.,
erythema) without pain
Skin changes (e.g.,
peeling, blisters,
bleeding, edema) or pain,
not interfering with
function
Ulcerative dermatitis or
skin changes with pain
interfering with function
— —
DERMATOLOGY/SKIN Page 3 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 16 - March 31, 2003, Publish Date: August 9, 2006
Skin breakdown/
decubitus ulcer
Decubitus — Local wound care;
medical intervention
indicated
Operative debridement or
other invasive
intervention indicated
(e.g., hyperbaric oxygen)
Life-threatening
consequences; major
invasive intervention
indicated (e.g., tissue
reconstruction, flap, or
grafting)
Death
REMARK: Skin breakdown/decubitus ulcer is to be used for loss of skin integrity or decubitus ulcer from pressure or as the result of operative or medical intervention.
Striae Striae Mild Cosmetically significant — — —
Telangiectasia Telangiectasia Few Moderate number Many and confluent — —
Ulceration Ulceration — Superficial ulceration
<2 cm size; local wound
care; medical intervention
indicated
Ulceration ≥2 cm size;
operative debridement,
primary closure or other
invasive intervention
indicated (e.g., hyperbaric
oxygen)
Life-threatening
consequences; major
invasive intervention
indicated (e.g., complete
resection, tissue
reconstruction, flap, or
grafting)
Death
Urticaria
(hives, welts, wheals)
Urticaria Intervention not indicated Intervention indicated for
<24 hrs
Intervention indicated for
≥24 hrs
— —
ALSO CONSIDER: Allergic reaction/hypersensitivity (including drug fever).
Wound complication,
non-infectious
Wound complication,
non-infectious
Incisional separation of
≤25% of wound, no
deeper than superficial
fascia
Incisional separation
>25% of wound with local
care; asymptomatic
hernia
Symptomatic hernia
without evidence of
strangulation; fascial
disruption/dehiscence
without evisceration;
primary wound closure or
revision by operative
intervention indicated;
hospitalization or
hyperbaric oxygen
indicated
Symptomatic hernia with
evidence of strangulation;
fascial disruption with
evisceration; major
reconstruction flap,
grafting, resection, or
amputation indicated
Death
REMARK: Wound complication, non-infectious is to be used for separation of incision, hernia, dehiscence, evisceration, or second surgery for wound revision.
Dermatology/Skin – Other
(Specify, __)
Dermatology – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
ENDOCRINE Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 17 - March 31, 2003, Publish Date: August 9, 2006
Adrenal insufficiency Adrenal insufficiency Asymptomatic,
intervention not indicated
Symptomatic, intervention
indicated
Hospitalization Life-threatening; disabling Death
REMARK: Adrenal insufficiency includes any of the following signs and symptoms: abdominal pain, anorexia, constipation, diarrhea, hypotension, pigmentation of mucous membranes,
pigmentation of skin, salt craving, syncope (fainting), vitiligo, vomiting, weakness, weight loss. Adrenal insufficiency must be confirmed by laboratory studies (low cortisol frequently
accompanied by low aldosterone).
A
LSO CONSIDER: Potassium, serum-high (hyperkalemia); Thyroid function, low (hypothyroidism).
Cushingoid appearance
(e.g., moon face, buffalo
hump, centripetal obesity,
cutaneous striae)
Cushingoid — Present — — —
ALSO CONSIDER: Glucose, serum-high (hyperglycemia); Potassium, serum-low (hypokalemia).
Feminization of male Feminization of male — — Present — —
NAVIGATION NOTE: Gynecomastia is graded in the SEXUAL/REPRODUCTIVE FUNCTION CATEGORY.
Hot flashes/flushes
3
Hot flashes Mild Moderate Interfering with ADL — —
Masculinization of female Masculinization of female — — Present — —
Neuroendocrine:
ACTH deficiency
ACTH Asymptomatic Symptomatic, not
interfering with ADL;
intervention indicated
Symptoms interfering with
ADL; hospitalization
indicated
Life-threatening
consequences (e.g.,
severe hypotension)
Death
Neuroendocrine:
ADH secretion
abnormality (e.g., SIADH
or low ADH)
ADH Asymptomatic Symptomatic, not
interfering with ADL;
intervention indicated
Symptoms interfering with
ADL
Life-threatening
consequences
Death
Neuroendocrine:
gonadotropin secretion
abnormality
Gonadotropin Asymptomatic Symptomatic, not
interfering with ADL;
intervention indicated
Symptoms interfering with
ADL; osteopenia;
fracture; infertility
— —
Neuroendocrine:
growth hormone
secretion abnormality
Growth hormone Asymptomatic Symptomatic, not
interfering with ADL;
intervention indicated
— — —
Neuroendocrine:
prolactin hormone
secretion abnormality
Prolactin Asymptomatic Symptomatic, not
interfering with ADL;
intervention indicated
Symptoms interfering with
ADL; amenorrhea;
galactorrhea
— Death
3
Sloan JA, Loprinzi CL, Novotny PJ, Barton DL, Lavasseur BI, Windschitl HJ, "Methodologic Lessons Learned from Hot Flash Studies," J Clin Oncol 2001 Dec 1;19(23):4280-90
ENDOCRINE Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 18 - March 31, 2003, Publish Date: August 9, 2006
Pancreatic endocrine:
glucose intolerance
Diabetes Asymptomatic,
intervention not indicated
Symptomatic; dietary
modification or oral agent
indicated
Symptoms interfering with
ADL; insulin indicated
Life-threatening
consequences (e.g.,
ketoacidosis,
hyperosmolar non-ketotic
coma)
Death
Parathyroid function, low
(hypoparathyroidism)
Hypoparathyroidism Asymptomatic,
intervention not indicated
Symptomatic; intervention
indicated
— — —
Thyroid function, high
(hyperthyroidism,
thyrotoxicosis)
Hyperthyroidism Asymptomatic,
intervention not indicated
Symptomatic, not
interfering with ADL;
thyroid suppression
therapy indicated
Symptoms interfering with
ADL; hospitalization
indicated
Life-threatening
consequences (e.g.,
thyroid storm)
Death
Thyroid function, low
(hypothyroidism)
Hypothyroidism Asymptomatic,
intervention not indicated
Symptomatic, not
interfering with ADL;
thyroid replacement
indicated
Symptoms interfering with
ADL; hospitalization
indicated
Life-threatening
myxedema coma
Death
Endocrine – Other
(Specify, __)
Endocrine – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death

GASTROINTESTINAL Page 1 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 19 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Abdominal pain or cramping is graded as Pain – Select in the PAIN CATEGORY.
Anorexia Anorexia Loss of appetite without
alteration in eating habits
Oral intake altered
without significant weight
loss or malnutrition; oral
nutritional supplements
indicated
Associated with
significant weight loss or
malnutrition (e.g.,
inadequate oral caloric
and/or fluid intake); IV
fluids, tube feedings or
TPN indicated
Life-threatening
consequences
Death
ALSO CONSIDER: Weight loss.
Ascites (non-malignant) Ascites Asymptomatic Symptomatic, medical
intervention indicated
Symptomatic, invasive
procedure indicated
Life-threatening
consequences
Death
REMARK: Ascites (non-malignant) refers to documented non-malignant ascites or unknown etiology, but unlikely malignant, and includes chylous ascites.
Colitis Colitis Asymptomatic, pathologic
or radiographic findings
only
Abdominal pain; mucus
or blood in stool
Abdominal pain, fever,
change in bowel habits
with ileus; peritoneal
signs
Life-threatening
consequences (e.g.,
perforation, bleeding,
ischemia, necrosis, toxic
megacolon)
Death
ALSO CONSIDER: Hemorrhage, GI – Select.
Constipation Constipation Occasional or intermittent
symptoms; occasional
use of stool softeners,
laxatives, dietary
modification, or enema
Persistent symptoms with
regular use of laxatives
or enemas indicated
Symptoms interfering
with ADL; obstipation
with manual evacuation
indicated
Life-threatening
consequences (e.g.,
obstruction, toxic
megacolon)
Death
ALSO CONSIDER: Ileus, GI (functional obstruction of bowel, i.e., neuroconstipation); Obstruction, GI – Select.
Dehydration Dehydration Increased oral fluids
indicated; dry mucous
membranes; diminished
skin turgor
IV fluids indicated <24
hrs
IV fluids indicated ≥24 hrs
Life-threatening
consequences (e.g.,
hemodynamic collapse)
Death
ALSO CONSIDER: Diarrhea; Hypotension; Vomiting.
Dental:
dentures or prosthesis
Dentures Minimal discomfort, no
restriction in activities
Discomfort preventing
use in some activities
(e.g., eating), but not
others (e.g., speaking)
Unable to use dentures
or prosthesis at any time
— —
GASTROINTESTINAL Page 2 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 20 - March 31, 2003, Publish Date: August 9, 2006
Dental:
periodontal disease
Periodontal Gingival recession or
gingivitis; limited bleeding
on probing; mild local
bone loss
Moderate gingival
recession or gingivitis;
multiple sites of bleeding
on probing; moderate
bone loss
Spontaneous bleeding;
severe bone loss with or
without tooth loss;
osteonecrosis of maxilla
or mandible
— —
REMARK: Severe periodontal disease leading to osteonecrosis is graded as Osteonecrosis (avascular necrosis) in the MUSCULOSKELETAL CATEGORY.
Dental:
teeth
Teeth Surface stains; dental
caries; restorable, without
extractions
Less than full mouth
extractions; tooth fracture
or crown amputation or
repair indicated
Full mouth extractions
indicated
— —
Dental:
teeth development
Teeth development Hypoplasia of tooth or
enamel not interfering
with function
Functional impairment
correctable with oral
surgery
Maldevelopment with
functional impairment not
surgically correctable
— —
Diarrhea Diarrhea Increase of <4 stools per
day over baseline; mild
increase in ostomy output
compared to baseline
Increase of 4 – 6 stools
per day over baseline; IV
fluids indicated <24hrs;
moderate increase in
ostomy output compared
to baseline; not
interfering with ADL
Increase of ≥7 stools per
day over baseline;
incontinence; IV fluids
≥24 hrs; hospitalization;
severe increase in
ostomy output compared
to baseline; interfering
with ADL
Life-threatening
consequences (e.g.,
hemodynamic collapse)
Death
REMARK: Diarrhea includes diarrhea of small bowel or colonic origin, and/or ostomy diarrhea.
ALSO CONSIDER: Dehydration; Hypotension.
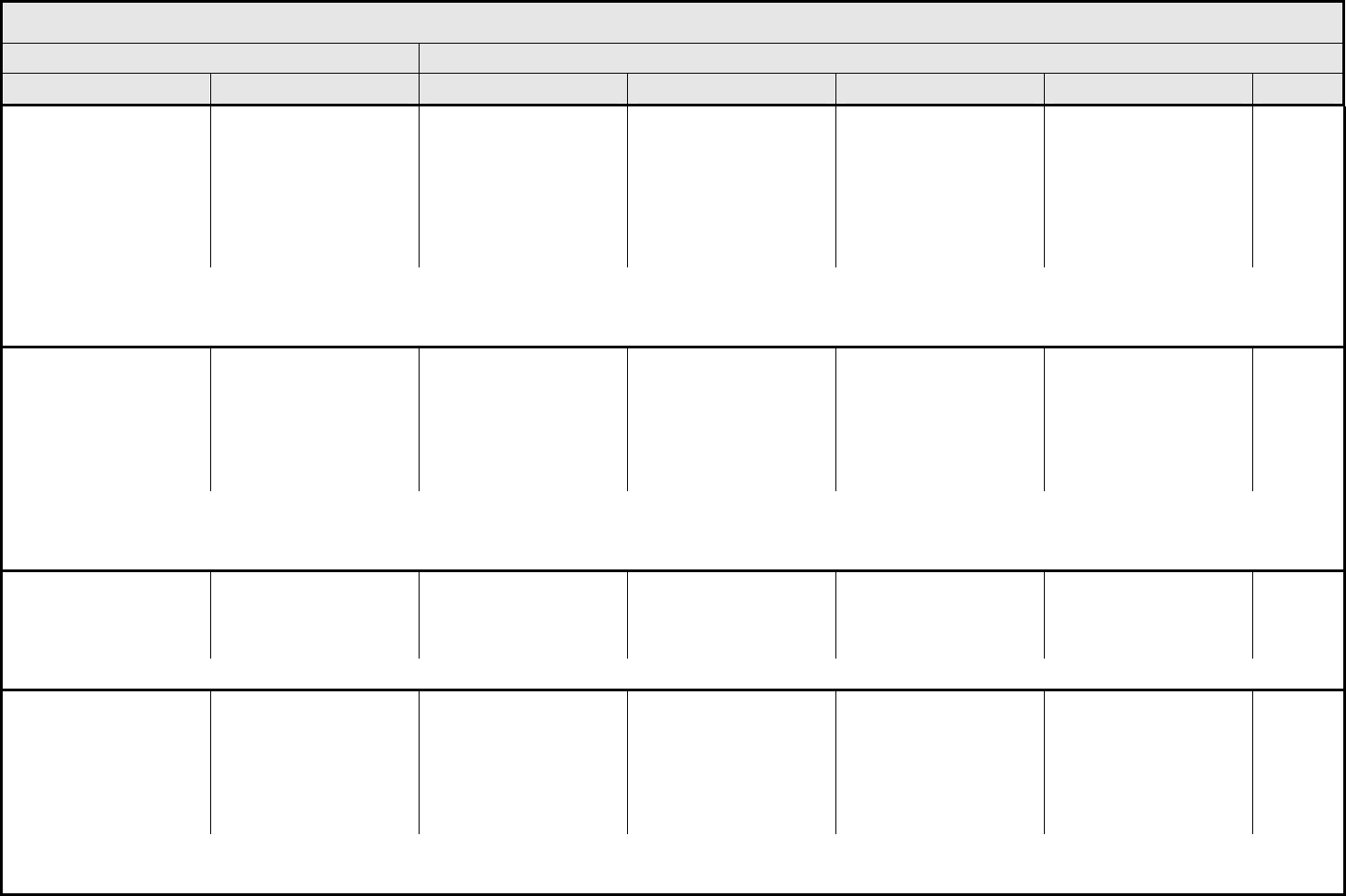
Distension/bloating,
abdominal
Distension Asymptomatic Symptomatic, but not
interfering with GI
function
Symptomatic, interfering
with GI function
— —
ALSO CONSIDER: Ascites (non-malignant); Ileus, GI (functional obstruction of bowel, i.e., neuroconstipation); Obstruction, GI – Select.
GASTROINTESTINAL Page 3 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 21 - March 31, 2003, Publish Date: August 9, 2006
Dry mouth/salivary gland
(xerostomia)
Dry mouth Symptomatic (dry or thick
saliva) without significant
dietary alteration;
unstimulated saliva flow
>0.2 ml/min
Symptomatic and
significant oral intake
alteration (e.g., copious
water, other lubricants,
diet limited to purees
and/or soft, moist foods);
unstimulated saliva
0.1 to 0.2 ml/min
Symptoms leading to
inability to adequately
aliment orally; IV fluids,
tube feedings, or TPN
indicated; unstimulated
saliva <0.1 ml/min
— —
REMARK: Dry mouth/salivary gland (xerostomia) includes descriptions of grade using both subjective and objective assessment parameters. Record this event consistently throughout
a patient’s participation on study. If salivary flow measurements are used for initial assessment, subsequent assessments must use salivary flow.
A
LSO CONSIDER: Salivary gland changes/saliva.
Dysphagia
(difficulty swallowing)
Dysphagia Symptomatic, able to eat
regular diet
Symptomatic and altered
eating/swallowing (e.g.,
altered dietary habits,
oral supplements); IV
fluids indicated <24 hrs
Symptomatic and
severely altered
eating/swallowing (e.g.,
inadequate oral caloric or
fluid intake); IV fluids,
tube feedings, or TPN
indicated ≥24 hrs
Life-threatening
consequences (e.g.,
obstruction, perforation)
Death
REMARK: Dysphagia (difficulty swallowing) is to be used for swallowing difficulty from oral, pharyngeal, esophageal, or neurologic origin. Dysphagia requiring dilation is graded as
Stricture/stenosis (including anastomotic), GI – Select.
A
LSO CONSIDER: Dehydration; Esophagitis.
Enteritis
(inflammation of the small
bowel)
Enteritis Asymptomatic, pathologic
or radiographic findings
only
Abdominal pain; mucus
or blood in stool
Abdominal pain, fever,
change in bowel habits
with ileus; peritoneal
signs
Life-threatening
consequences (e.g.,
perforation, bleeding,
ischemia, necrosis)
Death
ALSO CONSIDER: Hemorrhage, GI – Select; Typhlitis (cecal inflammation).
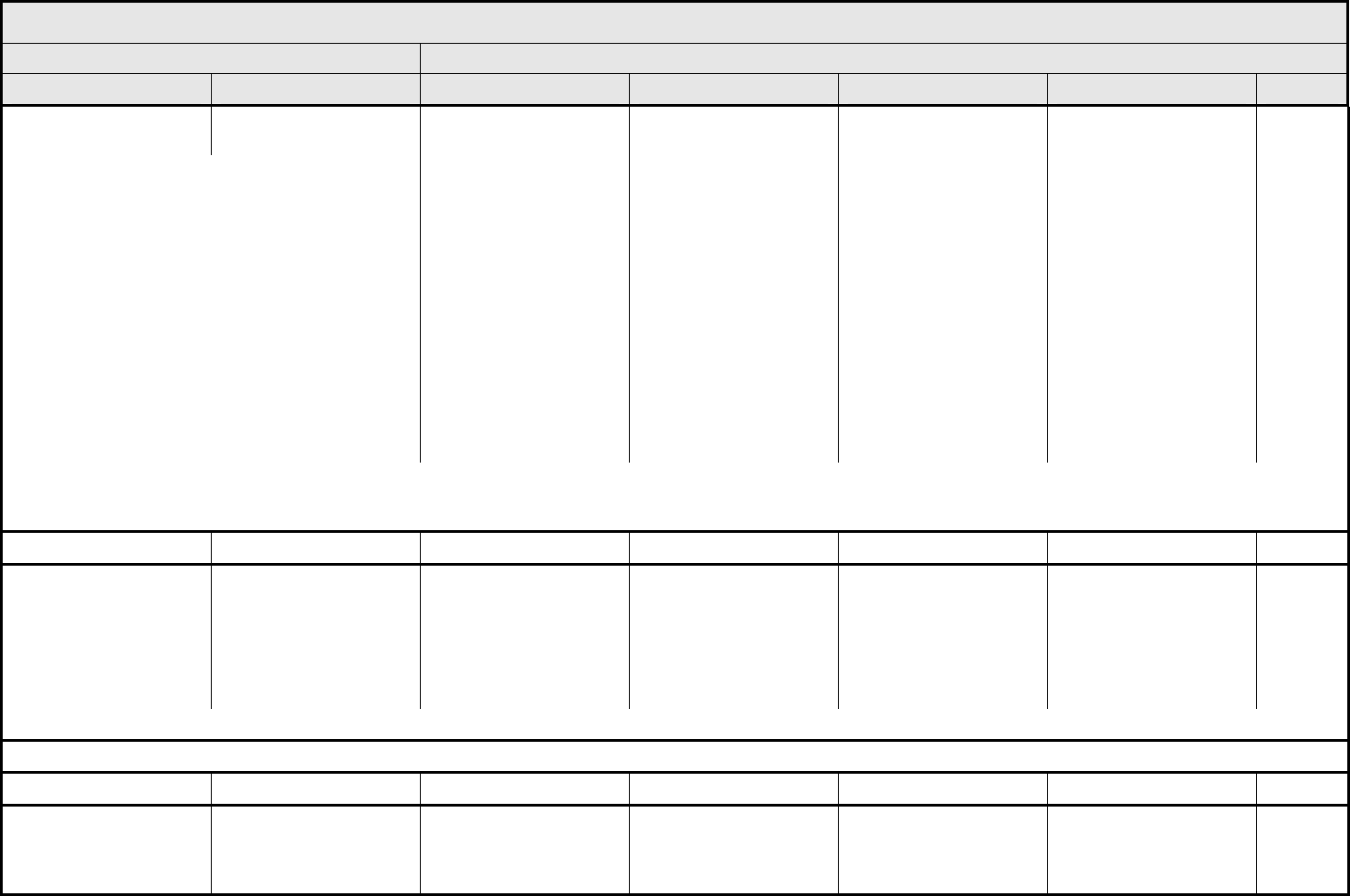
Esophagitis Esophagitis Asymptomatic pathologic,
radiographic, or
endoscopic findings only
Symptomatic; altered
eating/swallowing (e.g.,
altered dietary habits,
oral supplements); IV
fluids indicated <24 hrs
Symptomatic and
severely altered
eating/swallowing (e.g.,
inadequate oral caloric or
fluid intake); IV fluids,
tube feedings, or TPN
indicated ≥24 hrs
Life-threatening
consequences
Death
REMARK: Esophagitis includes reflux esophagitis.
A
LSO CONSIDER: Dysphagia (difficulty swallowing).
GASTROINTESTINAL Page 4 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 22 - March 31, 2003, Publish Date: August 9, 2006
Fistula, GI
– Select:
Fistula, GI – Select
– Abdomen NOS
– Anus
– Biliary tree
– Colon/cecum/appendix
– Duodenum
– Esophagus
– Gallbladder
– Ileum
– Jejunum
– Oral cavity
– Pancreas
– Pharynx
– Rectum
– Salivary gland
– Small bowel NOS
– Stomach
Asymptomatic,
radiographic findings only
Symptomatic; altered GI
function (e.g., altered
dietary habits, diarrhea,
or GI fluid loss); IV fluids
indicated <24 hrs
Symptomatic and
severely altered GI
function (e.g., altered
dietary habits, diarrhea,
or GI fluid loss); IV fluids,
tube feedings, or TPN
indicated ≥24 hrs
Life-threatening
consequences
Death
REMARK: A fistula is defined as an abnormal communication between two body cavities, potential spaces, and/or the skin. The site indicated for a fistula should be the site from which
the abnormal process is believed to have originated. For example, a tracheo-esophageal fistula arising in the context of a resected or irradiated esophageal cancer is graded as
Fistula, GI – esophagus.
Flatulence Flatulence Mild Moderate — — —
Gastritis (including bile
reflux gastritis)
Gastritis Asymptomatic
radiographic or
endoscopic findings only
Symptomatic; altered
gastric function (e.g.,
inadequate oral caloric or
fluid intake); IV fluids
indicated <24 hrs
Symptomatic and
severely altered gastric
function (e.g., inadequate
oral caloric or fluid
intake); IV fluids, tube
feedings, or TPN
indicated ≥24 hrs
Life-threatening
consequences; operative
intervention requiring
complete organ resection
(e.g., gastrectomy)
Death
ALSO CONSIDER: Hemorrhage, GI – Select; Ulcer, GI – Select.
NAVIGATION NOTE: Head and neck soft tissue necrosis is graded as Soft tissue necrosis – Select in the MUSCULOSKELETAL/SOFT TISSUE CATEGORY.
Heartburn/dyspepsia Heartburn Mild Moderate Severe — —
Hemorrhoids Hemorrhoids Asymptomatic Symptomatic; banding or
medical intervention
indicated
Interfering with ADL;
interventional radiology,
endoscopic, or operative
intervention indicated
Life-threatening
consequences
Death

GASTROINTESTINAL Page 5 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 23 - March 31, 2003, Publish Date: August 9, 2006
Ileus, GI (functional
obstruction of bowel, i.e.,
neuroconstipation)
Ileus Asymptomatic,
radiographic findings only
Symptomatic; altered GI
function (e.g., altered
dietary habits); IV fluids
indicated <24 hrs
Symptomatic and
severely altered GI
function; IV fluids, tube
feeding, or TPN indicated
≥24 hrs
Life-threatening
consequences
Death
REMARK: Ileus, GI is to be used for altered upper or lower GI function (e.g., delayed gastric or colonic emptying).
ALSO CONSIDER: Constipation; Nausea; Obstruction, GI – Select; Vomiting.
Incontinence, anal Incontinence, anal Occasional use of pads
required
Daily use of pads
required
Interfering with ADL;
operative intervention
indicated
Permanent bowel
diversion indicated
Death
REMARK: Incontinence, anal is to be used for loss of sphincter control as sequelae of operative or therapeutic intervention.
Leak (including
anastomotic), GI
– Select:
– Biliary tree
– Esophagus
– Large bowel
– Leak NOS
– Pancreas
– Pharynx
– Rectum
– Small bowel
– Stoma
– Stomach
Leak, GI – Select Asymptomatic
radiographic findings only
Symptomatic; medical
intervention indicated
Symptomatic and
interfering with GI
function; invasive or
endoscopic intervention
indicated
Life-threatening
consequences
Death
REMARK: Leak (including anasomotic), GI – Select is to be used for clinical signs/symptoms or radiographic confirmation of anastomotic or conduit leak (e.g., biliary, esophageal,
intestinal, pancreatic, pharyngeal, rectal), but without development of fistula.
Malabsorption Malabsorption — Altered diet; oral
therapies indicated (e.g.,
enzymes, medications,
dietary supplements)
Inability to aliment
adequately via GI tract
(i.e., TPN indicated)
Life-threatening
consequences
Death

GASTROINTESTINAL Page 6 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 24 - March 31, 2003, Publish Date: August 9, 2006
Mucositis/stomatitis
(clinical exam)
– Select:
– Anus
– Esophagus
– Large bowel
– Larynx
– Oral cavity
– Pharynx
– Rectum
– Small bowel
– Stomach
– Trachea
Mucositis (clinical exam)
– Select
Erythema of the mucosa Patchy ulcerations or
pseudomembranes
Confluent ulcerations or
pseudomembranes;
bleeding with minor
trauma
Tissue necrosis;
significant spontaneous
bleeding; life-threatening
consequences
Death
REMARK: Mucositis/stomatitis (functional/symptomatic) may be used for mucositis of the upper aero-digestive tract caused by radiation, agents, or GVHD.
Mucositis/stomatitis
(functional/symptomatic)
– Select:
– Anus
– Esophagus
– Large bowel
– Larynx
– Oral cavity
– Pharynx
– Rectum
– Small bowel
– Stomach
– Trachea
Mucositis (functional/
symptomatic) – Select
Upper aerodigestive tract
sites: Minimal symptoms,
normal diet; minimal
respiratory symptoms but
not interfering with
function
Lower GI sites:
Minimal discomfort,
intervention not indicated
Upper aerodigestive tract
sites: Symptomatic but
can eat and swallow
modified diet; respiratory
symptoms interfering with
function but not
interfering with ADL
Lower GI sites:
Symptomatic, medical
intervention indicated but
not interfering with ADL
Upper aerodigestive tract
sites: Symptomatic and
unable to adequately
aliment or hydrate orally;
respiratory symptoms
interfering with ADL
Lower GI sites:
Stool incontinence or
other symptoms
interfering with ADL
Symptoms associated
with life-threatening
consequences
Death
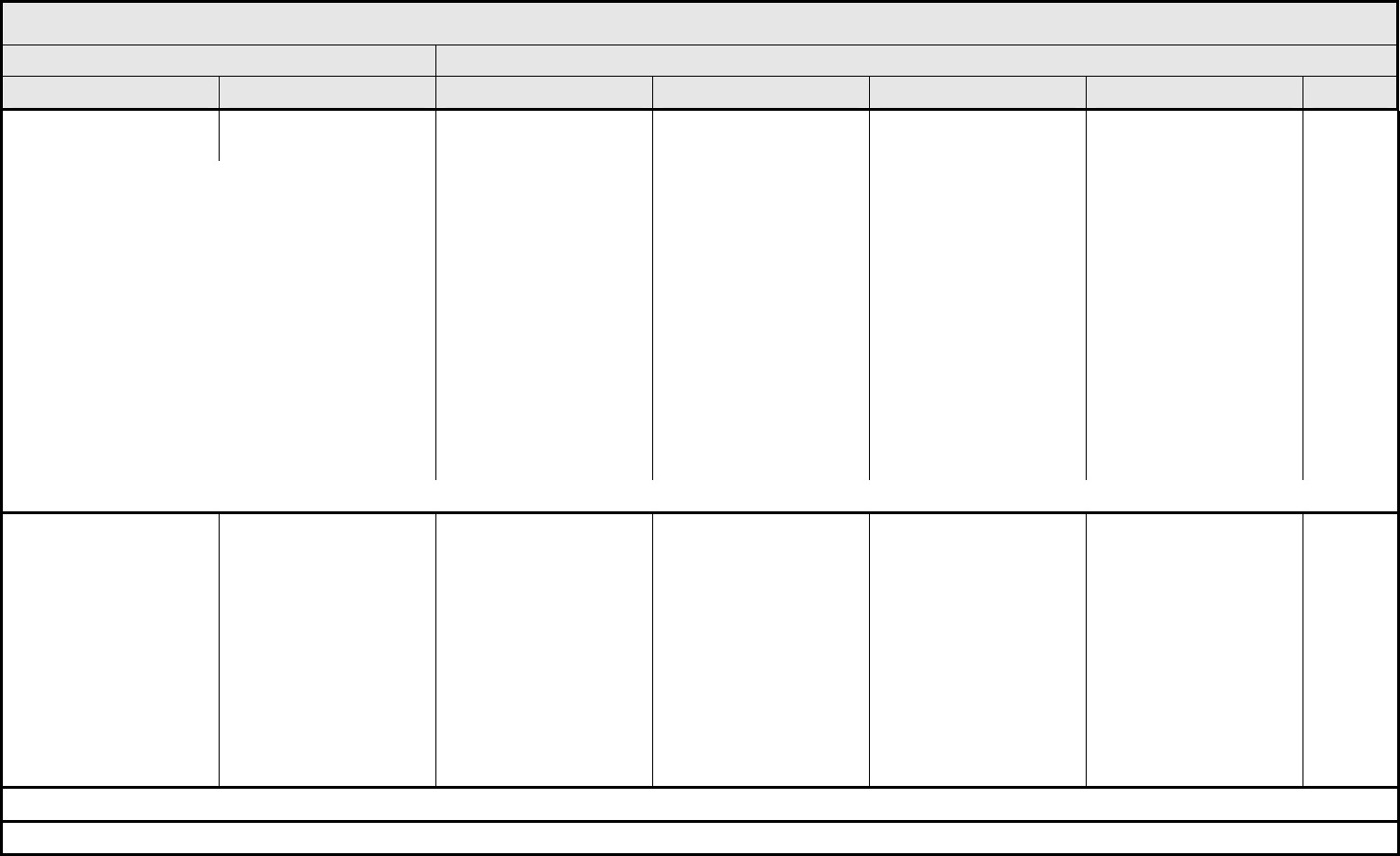
Nausea Nausea Loss of appetite without
alteration in eating habits
Oral intake decreased
without significant weight
loss, dehydration or
malnutrition; IV fluids
indicated <24 hrs
Inadequate oral caloric or
fluid intake; IV fluids, tube
feedings, or TPN
indicated ≥24 hrs
Life-threatening
consequences
Death
ALSO CONSIDER: Anorexia; Vomiting.
GASTROINTESTINAL Page 7 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 25 - March 31, 2003, Publish Date: August 9, 2006
Necrosis, GI
– Select:
Necrosis, GI – Select
– Anus
– Colon/cecum/appendix
– Duodenum
– Esophagus
– Gallbladder
– Hepatic
– Ileum
– Jejunum
– Oral
– Pancreas
– Peritoneal cavity
– Pharynx
– Rectum
– Small bowel NOS
– Stoma
– Stomach
— — Inability to aliment
adequately by GI tract
(e.g., requiring enteral or
parenteral nutrition);
interventional radiology,
endoscopic, or operative
intervention indicated
Life-threatening
consequences; operative
intervention requiring
complete organ resection
(e.g., total colectomy)
Death
ALSO CONSIDER: Visceral arterial ischemia (non-myocardial).
Obstruction, GI
– Select:
– Cecum
– Colon
– Duodenum
– Esophagus
– Gallbladder
– Ileum
– Jejunum
– Rectum
– Small bowel NOS
– Stoma
– Stomach
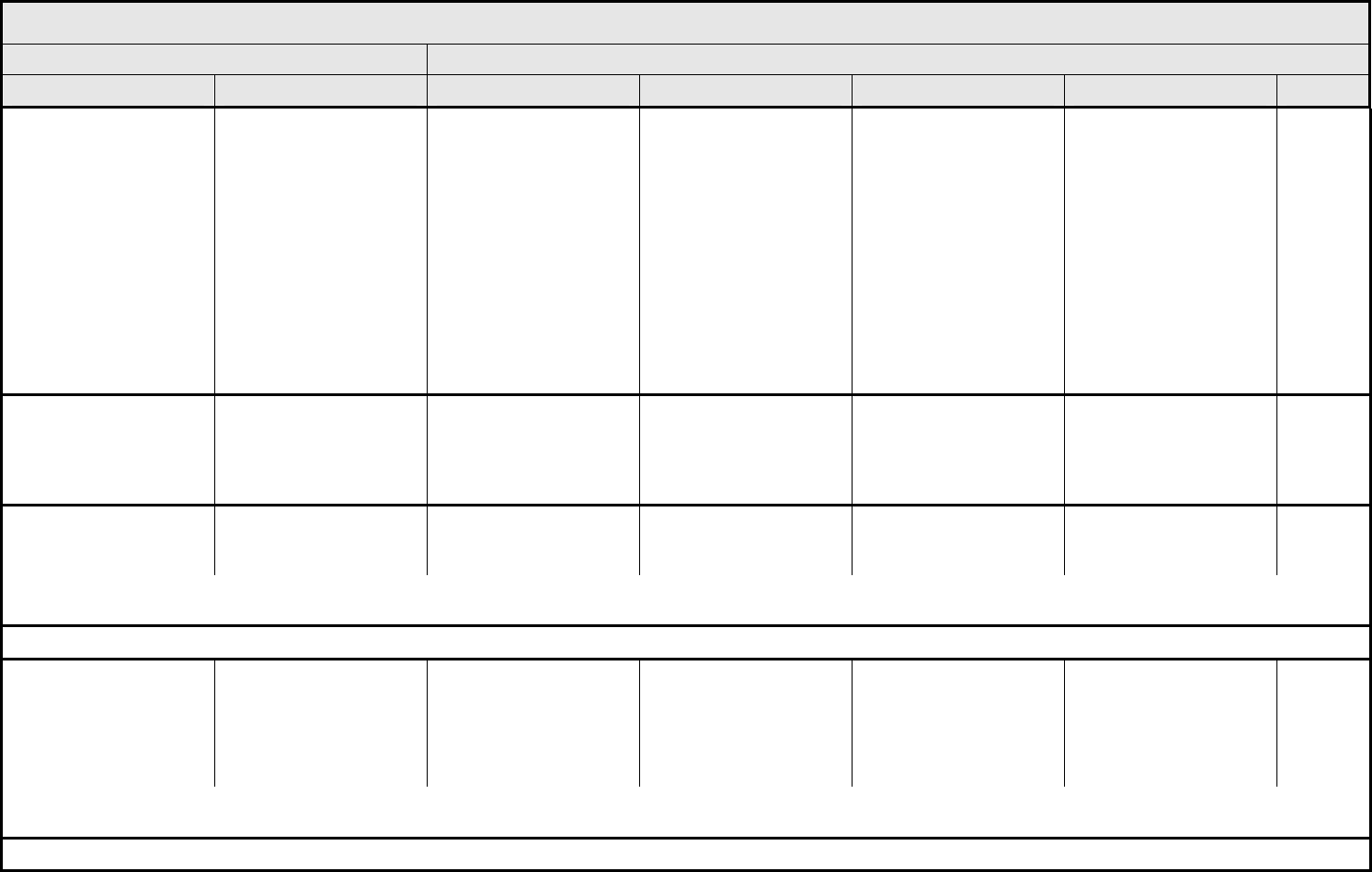
Obstruction, GI – Select Asymptomatic
radiographic findings only
Symptomatic; altered GI
function (e.g., altered
dietary habits, vomiting,
diarrhea, or GI fluid loss);
IV fluids indicated <24
hrs
Symptomatic and
severely altered GI
function (e.g., altered
dietary habits, vomiting,
diarrhea, or GI fluid loss);
IV fluids, tube feedings,
or TPN indicated ≥24 hrs;
operative intervention
indicated
Life-threatening
consequences; operative
intervention requiring
complete organ resection
(e.g., total colectomy)
Death
NAVIGATION NOTE: Operative injury is graded as Intra-operative injury – Select Organ or Structure in the SURGERY/INTRA-OPERATIVE INJURY CATEGORY.
NAVIGATION NOTE: Pelvic pain is graded as Pain – Select in the PAIN CATEGORY.
GASTROINTESTINAL Page 8 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 26 - March 31, 2003, Publish Date: August 9, 2006
Perforation, GI
– Select:
– Appendix
– Biliary tree
– Cecum
– Colon
– Duodenum
– Esophagus
– Gallbladder
– Ileum
– Jejunum
– Rectum
– Small bowel NOS
– Stomach
Perforation, GI – Select Asymptomatic
radiographic findings only
Medical intervention
indicated; IV fluids
indicated <24 hrs
IV fluids, tube feedings,
or TPN indicated ≥24 hrs;
operative intervention
indicated
Life-threatening
consequences
Death
Proctitis Proctitis Rectal discomfort,
intervention not indicated
Symptoms not interfering
with ADL; medical
intervention indicated
Stool incontinence or
other symptoms
interfering with ADL;
operative intervention
indicated
Life-threatening
consequences (e.g.,
perforation)
Death
Prolapse of stoma, GI Prolapse of stoma, GI Asymptomatic Extraordinary local care
or maintenance; minor
revision indicated
Dysfunctional stoma;
major revision indicated
Life-threatening
consequences
Death
REMARK: Other stoma complications may be graded as Fistula, GI – Select; Leak (including anastomotic), GI – Select; Obstruction, GI – Select; Perforation, GI – Select;
Stricture/stenosis (including anastomotic), GI – Select.
NAVIGATION NOTE: Rectal or perirectal pain (proctalgia) is graded as Pain – Select in the PAIN CATEGORY.
Salivary gland
changes/saliva
Salivary gland changes Slightly thickened saliva;
slightly altered taste (e.g.,
metallic)
Thick, ropy, sticky saliva;
markedly altered taste;
alteration in diet
indicated; secretion-
induced symptoms not
interfering with ADL
Acute salivary gland
necrosis; severe
secretion-induced
symptoms interfering with
ADL
Disabling —
ALSO CONSIDER: Dry mouth/salivary gland (xerostomia); Mucositis/stomatitis (clinical exam) – Select; Mucositis/stomatitis (functional/symptomatic) – Select; Taste alteration
(dysgeusia).
NAVIGATION NOTE: Splenic function is graded in the BLOOD/BONE MARROW CATEGORY.
GASTROINTESTINAL Page 9 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 27 - March 31, 2003, Publish Date: August 9, 2006
Stricture/stenosis
(including anastomotic),
GI
– Select:
Stricture, GI – Select
– Anus
– Biliary tree
– Cecum
– Colon
– Duodenum
– Esophagus
– Ileum
– Jejunum
– Pancreas/pancreatic duct
– Pharynx
– Rectum
– Small bowel NOS
– Stoma
– Stomach
Asymptomatic
radiographic findings only
Symptomatic; altered GI
function (e.g., altered
dietary habits, vomiting,
bleeding, diarrhea); IV
fluids indicated <24 hrs
Symptomatic and
severely altered GI
function (e.g., altered
dietary habits, diarrhea,
or GI fluid loss); IV fluids,
tube feedings, or TPN
indicated ≥24 hrs;
operative intervention
indicated
Life-threatening
consequences; operative
intervention requiring
complete organ resection
(e.g., total colectomy)
Death
Taste alteration
(dysgeusia)
Taste alteration Altered taste but no
change in diet
Altered taste with change
in diet (e.g., oral
supplements); noxious or
unpleasant taste; loss of
taste
— — —
Typhlitis
(cecal inflammation)
Typhlitis Asymptomatic, pathologic
or radiographic findings
only
Abdominal pain; mucus
or blood in stool
Abdominal pain, fever,
change in bowel habits
with ileus; peritoneal
signs
Life-threatening
consequences (e.g.,
perforation, bleeding,
ischemia, necrosis);
operative intervention
indicated
Death
ALSO CONSIDER: Colitis; Hemorrhage, GI – Select ; Ileus, GI (functional obstruction of bowel, i.e., neuroconstipation).

GASTROINTESTINAL Page 10 of 10
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 28 - March 31, 2003, Publish Date: August 9, 2006
Ulcer, GI
– Select:
– Anus
– Cecum
– Colon
– Duodenum
– Esophagus
– Ileum
– Jejunum
– Rectum
– Small bowel NOS
– Stoma
– Stomach
Ulcer, GI – Select Asymptomatic,
radiographic or
endoscopic findings only
Symptomatic; altered GI
function (e.g., altered
dietary habits, oral
supplements); IV fluids
indicated <24 hrs
Symptomatic and
severely altered GI
function (e.g., inadequate
oral caloric or fluid
intake); IV fluids, tube
feedings, or TPN
indicated ≥24 hrs
Life-threatening
consequences
Death
ALSO CONSIDER: Hemorrhage, GI – Select.
Vomiting Vomiting 1 episode in 24 hrs 2 – 5 episodes in 24 hrs;
IV fluids indicated
<24 hrs
≥6 episodes in 24 hrs; IV
fluids, or TPN indicated
≥24 hrs
Life-threatening
consequences
Death
ALSO CONSIDER: Dehydration.
Gastrointestinal – Other
(Specify, __)
GI – Other (Specify) Mild Moderate Severe Life-threatening;
disabling
Death
GROWTH AND DEVELOPMENT Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 29 - March 31, 2003, Publish Date: August 9, 2006
Bone age
(alteration in bone age)
Bone age — +2 SD (standard
deviation) from normal
— — —
Bone growth:
femoral head; slipped
capital femoral epiphysis
Femoral head growth Mild valgus/varus
deformity
Moderate valgus/varus
deformity, symptomatic,
interfering with function
but not interfering with
ADL
Mild slipped capital
femoral epiphysis;
operative intervention
(e.g., fixation) indicated;
interfering with ADL
Disabling; severe slipped
capital femoral epiphysis
>60%; avascular necrosis
—
Bone growth:
limb length discrepancy
Limb length Mild length discrepancy
<2 cm
Moderate length
discrepancy 2 – 5 cm;
shoe lift indicated
Severe length
discrepancy >5 cm;
operative intervention
indicated; interfering with
ADL
Disabling; epiphysiodesis —
Bone growth:
spine kyphosis/lordosis
Kyphosis/lordosis Mild radiographic
changes
Moderate accentuation;
interfering with function
but not interfering with
ADL
Severe accentuation;
operative intervention
indicated; interfering with
ADL
Disabling (e.g., cannot lift
head)
—
Growth velocity
(reduction in growth
velocity)
Reduction in growth
velocity
10 – 29% reduction in
growth from the baseline
growth curve
30 – 49% reduction in
growth from the baseline
growth curve
≥50% reduction in growth
from the baseline growth
curve
— —
Puberty (delayed) Delayed puberty — No breast development
by age 13 yrs for females;
no Tanner Stage 2
development by age 14.5
yrs for males
No sexual development
by age 14 yrs for girls,
age 16 yrs for boys;
hormone replacement
indicated
— —
REMARK: Do not use testicular size for Tanner Stage in male cancer survivors.
Puberty (precocious) Precocious puberty — Physical signs of puberty
<7 years for females,
<9 years for males
— — —
Short stature Short stature Beyond two standard
deviations of age and
gender mean height
Altered ADL — — —
REMARK: Short stature is secondary to growth hormone deficiency.
A
LSO CONSIDER: Neuroendocrine: growth hormone secretion abnormality.
Growth and Development
– Other (Specify, __)
Growth and Development
– Other (Specify)
Mild Moderate Severe Life-threatening; disabling Death
HEMORRHAGE/BLEEDING Page 1 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 30 - March 31, 2003, Publish Date: August 9, 2006
Hematoma Hematoma Minimal symptoms,
invasive intervention not
indicated
Minimally invasive
evacuation or aspiration
indicated
Transfusion,
interventional radiology,
or operative intervention
indicated
Life-threatening
consequences; major
urgent intervention
indicated
Death
REMARK: Hematoma refers to extravasation at wound or operative site or secondary to other intervention. Transfusion implies pRBC.
A
LSO CONSIDER: Fibrinogen; INR (International Normalized Ratio of prothrombin time); Platelets; PTT (Partial Thromboplastin Time).
Hemorrhage/bleeding
associated with surgery,
intra-operative or
postoperative
Hemorrhage with surgery — — Requiring transfusion of
2 units non-autologous
(10 cc/kg for pediatrics)
pRBCs beyond protocol
specification;
postoperative
interventional radiology,
endoscopic, or operative
intervention indicated
Life-threatening
consequences
Death
REMARK: Postoperative period is defined as ≤72 hours after surgery. Verify protocol-specific acceptable guidelines regarding pRBC transfusion.
ALSO CONSIDER: Fibrinogen; INR (International Normalized Ratio of prothrombin time); Platelets; PTT (Partial Thromboplastin Time).
Hemorrhage, CNS CNS hemorrhage Asymptomatic,
radiographic findings only
Medical intervention
indicated
Ventriculostomy, ICP
monitoring,
intraventricular
thrombolysis, or operative
intervention indicated
Life-threatening
consequences;
neurologic deficit or
disability
Death
ALSO CONSIDER: Fibrinogen; INR (International Normalized Ratio of prothrombin time); Platelets; PTT (Partial Thromboplastin Time).

HEMORRHAGE/BLEEDING Page 2 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 31 - March 31, 2003, Publish Date: August 9, 2006
Hemorrhage, GI
– Select:
– Abdomen NOS
– Anus
– Biliary tree
– Cecum/appendix
– Colon
– Duodenum
– Esophagus
– Ileum
– Jejunum
– Liver
– Lower GI NOS
– Oral cavity
– Pancreas
– Peritoneal cavity
– Rectum
– Stoma
– Stomach
– Upper GI NOS
– Varices (esophageal)
– Varices (rectal)
Hemorrhage, GI – Select Mild, intervention (other
than iron supplements)
not indicated
Symptomatic and medical
intervention or minor
cauterization indicated
Transfusion,
interventional radiology,
endoscopic, or operative
intervention indicated;
radiation therapy (i.e.,
hemostasis of bleeding
site)
Life-threatening
consequences; major
urgent intervention
indicated
Death
REMARK: Transfusion implies pRBC.
ALSO CONSIDER: Fibrinogen; INR (International Normalized Ratio of prothrombin time); Platelets; PTT (Partial Thromboplastin Time).

HEMORRHAGE/BLEEDING Page 3 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 32 - March 31, 2003, Publish Date: August 9, 2006
Hemorrhage, GU
– Select:
– Bladder
– Fallopian tube
– Kidney
– Ovary
– Prostate
– Retroperitoneum
– Spermatic cord
– Stoma
– Testes
– Ureter
– Urethra
– Urinary NOS
– Uterus
– Vagina
– Vas deferens
Hemorrhage, GU – Select Minimal or microscopic
bleeding; intervention not
indicated
Gross bleeding, medical
intervention, or urinary
tract irrigation indicated
Transfusion,
interventional radiology,
endoscopic, or operative
intervention indicated;
radiation therapy (i.e.,
hemostasis of bleeding
site)
Life-threatening
consequences; major
urgent intervention
indicated
Death
REMARK: Transfusion implies pRBC.
A
LSO CONSIDER: Fibrinogen; INR (International Normalized Ratio of prothrombin time); Platelets; PTT (Partial Thromboplastin Time).
Hemorrhage, pulmonary/
upper respiratory
– Select:
Hemorrhage pulmonary
– Select
– Bronchopulmonary NOS
– Bronchus
– Larynx
– Lung
– Mediastinum
– Nose
– Pharynx
– Pleura
– Respiratory tract NOS
– Stoma
– Trachea
Mild, intervention not
indicated
Symptomatic and
medical intervention
indicated
Transfusion,
interventional radiology,
endoscopic, or operative
intervention indicated;
radiation therapy (i.e.,
hemostasis of bleeding
site)
Life-threatening
consequences; major
urgent intervention
indicated
Death
REMARK: Transfusion implies pRBC.
A
LSO CONSIDER: Fibrinogen; INR (International Normalized Ratio of prothrombin time); Platelets; PTT (Partial Thromboplastin Time).
Petechiae/purpura
(hemorrhage/bleeding
into skin or mucosa)
Petechiae Few petechiae Moderate petechiae;
purpura
Generalized petechiae or
purpura
— —
ALSO CONSIDER: Fibrinogen; INR (International Normalized Ratio of prothrombin time); Platelets; PTT (Partial Thromboplastin Time).

HEMORRHAGE/BLEEDING Page 4 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 33 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Vitreous hemorrhage is graded in the OCULAR/VISUAL CATEGORY.
Hemorrhage/Bleeding –
Other (Specify, __)
Hemorrhage – Other
(Specify)
Mild without transfusion — Transfusion indicated Catastrophic bleeding,
requiring major non-
elective intervention
Death

HEPATOBILIARY/PANCREAS Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 34 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Biliary tree damage is graded as Fistula, GI – Select; Leak (including anastomotic), GI – Select; Necrosis, GI – Select; Obstruction, GI – Select; Perforation, GI –
Select; Stricture/stenosis (including anastomotic), GI – Select in the GASTROINTESTINAL CATEGORY.
Cholecystitis Cholecystitis Asymptomatic,
radiographic findings only
Symptomatic, medical
intervention indicated
Interventional radiology,
endoscopic, or operative
intervention indicated
Life-threatening
consequences (e.g.,
sepsis or perforation)
Death
ALSO CONSIDER: Infection (documented clinically or microbiologically) with Grade 3 or 4 neutrophils – Select; Infection with normal ANC or Grade 1 or 2 neutrophils – Select; Infection
with unknown ANC – Select.
Liver dysfunction/failure
(clinical)
Liver dysfunction — Jaundice Asterixis Encephalopathy or coma Death
REMARK: Jaundice is not an AE, but occurs when the liver is not working properly or when a bile duct is blocked. It is graded as a result of liver dysfunction/failure or elevated bilirubin.
A
LSO CONSIDER: Bilirubin (hyperbilirubinemia).
Pancreas, exocrine
enzyme deficiency
Pancreas, exocrine
enzyme deficiency
— Increase in stool
frequency, bulk, or odor;
steatorrhea
Sequelae of absorption
deficiency (e.g., weight
loss)
Life-threatening
consequences
Death
ALSO CONSIDER: Diarrhea.
Pancreatitis Pancreatitis Asymptomatic, enzyme
elevation and/or
radiographic findings
Symptomatic, medical
intervention indicated
Interventional radiology or
operative intervention
indicated
Life-threatening
consequences
(e.g., circulatory failure,
hemorrhage, sepsis)
Death
ALSO CONSIDER: Amylase.
NAVIGATION NOTE: Stricture (biliary tree, hepatic or pancreatic) is graded as Stricture/stenosis (including anastomotic), GI – Select in the GASTROINTESTINAL CATEGORY.
Hepatobiliary/Pancreas –
Other (Specify, __)
Hepatobiliary – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death

INFECTION Page 1 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 35 - March 31, 2003, Publish Date: August 9, 2006
Colitis, infectious
(e.g., Clostridium difficile)
Colitis, infectious Asymptomatic, pathologic
or radiographic findings
only
Abdominal pain with
mucus and/or blood in
stool
IV antibiotics or TPN
indicated
Life-threatening
consequences (e.g.,
perforation, bleeding,
ischemia, necrosis or
toxic megacolon);
operative resection or
diversion indicated
Death
ALSO CONSIDER: Hemorrhage, GI – Select; Typhlitis (cecal inflammation).
Febrile neutropenia
(fever of unknown origin
without clinically or
microbiologically
documented infection)
(ANC <1.0 x 10
9
/L, fever
≥38.5°C)
Febrile neutropenia — — Present Life-threatening
consequences (e.g.,
septic shock,
hypotension, acidosis,
necrosis)
Death
ALSO CONSIDER: Neutrophils/granulocytes (ANC/AGC).
Infection
(documented clinically or
microbiologically) with
Grade 3 or 4 neutrophils
(ANC <1.0 x 10
9
/L)
– Select
‘Select’ AEs appear at the
end of the CATEGORY.
Infection (documented
clinically) with Grade 3 or
4 ANC – Select
— Localized, local
intervention indicated
IV antibiotic, antifungal, or
antiviral intervention
indicated; interventional
radiology or operative
intervention indicated
Life-threatening
consequences (e.g.,
septic shock,
hypotension, acidosis,
necrosis)
Death
REMARK: Fever with Grade 3 or 4 neutrophils in the absence of documented infection is graded as Febrile neutropenia (fever of unknown origin without clinically or microbiologically
documented infection).
A
LSO CONSIDER: Neutrophils/granulocytes (ANC/AGC).
Infection with normal
ANC or Grade 1 or 2
neutrophils
– Select
‘Select’ AEs appear at the
end of the CATEGORY.
Infection with normal
ANC – Select
— Localized, local
intervention indicated
IV antibiotic, antifungal, or
antiviral intervention
indicated; interventional
radiology or operative
intervention indicated
Life-threatening
consequences (e.g.,
septic shock,
hypotension, acidosis,
necrosis)
Death
INFECTION Page 2 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 36 - March 31, 2003, Publish Date: August 9, 2006
Infection with unknown
ANC
– Select
‘Select’ AEs appear at the
end of the CATEGORY.
Infection with unknown
ANC – Select
— Localized, local
intervention indicated
IV antibiotic, antifungal, or
antiviral intervention
indicated; interventional
radiology or operative
intervention indicated
Life-threatening
consequences (e.g.,
septic shock,
hypotension, acidosis,
necrosis)
Death
REMARK: Infection with unknown ANC – Select is to be used in the rare case when ANC is unknown.
Opportunistic infection
associated with ≥Grade 2
Lymphopenia
Opportunistic infection — Localized, local
intervention indicated
IV antibiotic, antifungal, or
antiviral intervention
indicated; interventional
radiology or operative
intervention indicated
Life-threatening
consequences (e.g.,
septic shock,
hypotension, acidosis,
necrosis)
Death
ALSO CONSIDER: Lymphopenia.
Viral hepatitis Viral hepatitis Present; transaminases
and liver function normal
Transaminases
abnormal, liver function
normal
Symptomatic liver
dysfunction; fibrosis by
biopsy; compensated
cirrhosis
Decompensated liver
function (e.g., ascites,
coagulopathy,
encephalopathy, coma)
Death
REMARK: Non-viral hepatitis is graded as Infection – Select.
ALSO CONSIDER: Albumin, serum-low (hypoalbuminemia); ALT, SGPT (serum glutamic pyruvic transaminase); AST, SGOT (serum glutamic oxaloacetic transaminase); Bilirubin
(hyperbilirubinemia); Encephalopathy.
Infection – Other
(Specify, __)
Infection – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death

INFECTION – SELECT Page 3 of 3
CTCAE v3.0 - 37 - March 31, 2003, Publish Date: August 9, 2006
AUDITORY/EAR
– External ear (otitis externa)
– Middle ear (otitis media)
CARDIOVASCULAR
– Artery
– Heart (endocarditis)
– Spleen
– Vein
DERMATOLOGY/SKIN
– Lip/perioral
– Peristomal
– Skin (cellulitis)
– Ungual (nails)
GASTROINTESTINAL
– Abdomen NOS
– Anal/perianal
– Appendix
– Cecum
– Colon
– Dental-tooth
– Duodenum
– Esophagus
– Ileum
– Jejunum
– Oral cavity-gums (gingivitis)
– Peritoneal cavity
– Rectum
– Salivary gland
– Small bowel NOS
– Stomach
GENERAL
– Blood
– Catheter-related
– Foreign body (e.g., graft, implant,
prosthesis, stent)
– Wound
HEPATOBILIARY/PANCREAS
– Biliary tree
– Gallbladder (cholecystitis)
– Liver
– Pancreas
LYMPHATIC
– Lymphatic
MUSCULOSKELETAL
– Bone (osteomyelitis)
– Joint
– Muscle (infection myositis)
– Soft tissue NOS
NEUROLOGY
– Brain (encephalitis, infectious)
– Brain + Spinal cord (encephalomyelitis)
– Meninges (meningitis)
– Nerve-cranial
– Nerve-peripheral
– Spinal cord (myelitis)
OCULAR
– Conjunctiva
– Cornea
– Eye NOS
– Lens
PULMONARY/UPPER RESPIRATORY
– Bronchus
– Larynx
– Lung (pneumonia)
– Mediastinum NOS
– Mucosa
– Neck NOS
– Nose
– Paranasal
– Pharynx
– Pleura (empyema)
– Sinus
– Trachea
– Upper aerodigestive NOS
– Upper airway NOS
RENAL/GENITOURINARY
– Bladder (urinary)
– Kidney
– Prostate
– Ureter
– Urethra
– Urinary tract NOS
SEXUAL/REPRODUCTIVE FUNCTION
– Cervix
– Fallopian tube
– Pelvis NOS
– Penis
– Scrotum
– Uterus
– Vagina
– Vulva
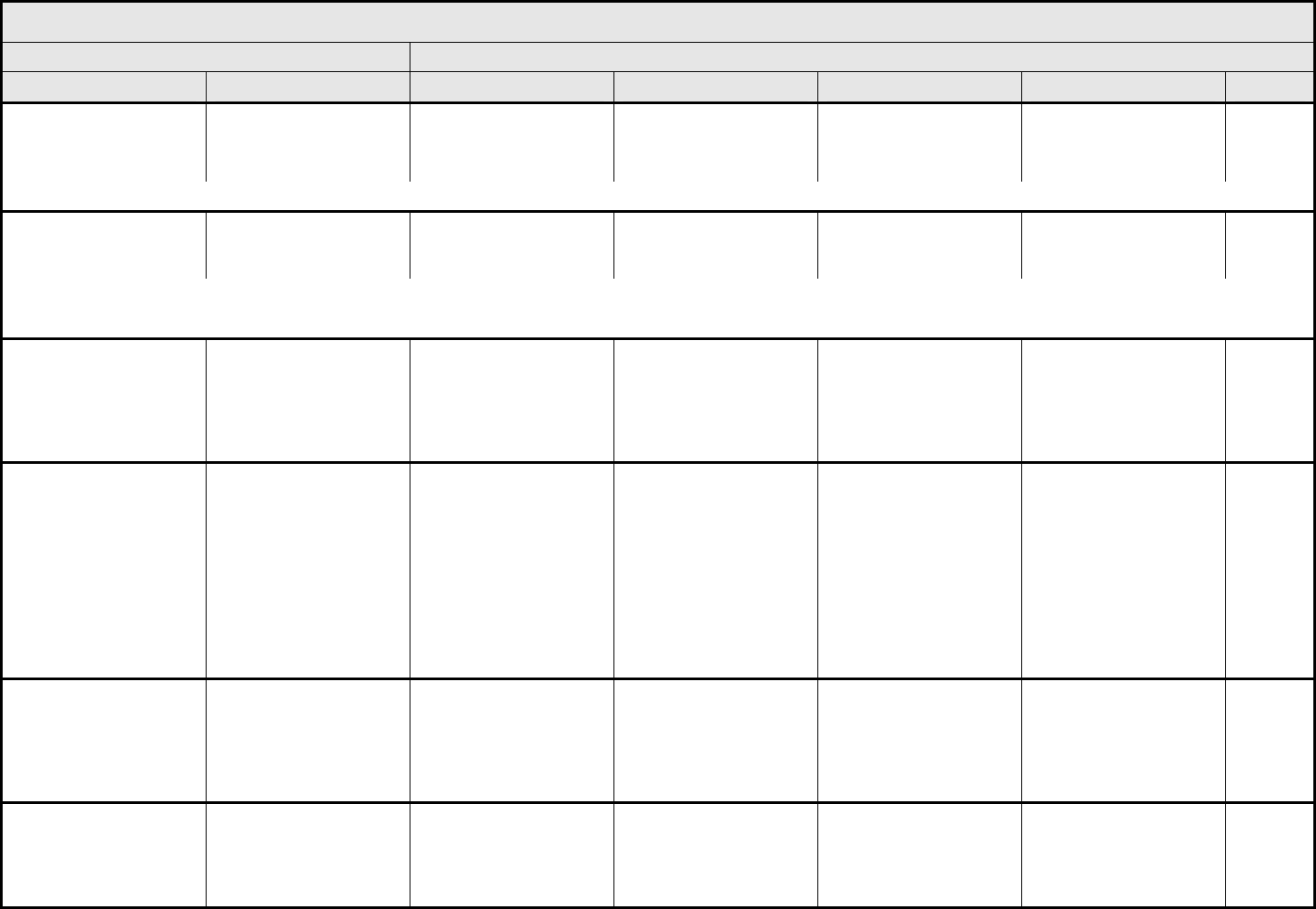
LYMPHATICS Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 38 - March 31, 2003, Publish Date: August 9, 2006
Chyle or lymph leakage Chyle or lymph leakage Asymptomatic, clinical or
radiographic findings
Symptomatic, medical
intervention indicated
Interventional radiology
or operative intervention
indicated
Life-threatening
complications
Death
ALSO CONSIDER: Chylothorax.
Dermal change
lymphedema,
phlebolymphedema
Dermal change Trace thickening or faint
discoloration
Marked discoloration;
leathery skin texture;
papillary formation
— — —
REMARK: Dermal change lymphedema, phlebolymphedema refers to changes due to venous stasis.
ALSO CONSIDER: Ulceration.
Edema:
head and neck
Edema: head and neck Localized to dependent
areas, no disability or
functional impairment
Localized facial or neck
edema with functional
impairment
Generalized facial or
neck edema with
functional impairment
(e.g., difficulty in turning
neck or opening mouth
compared to baseline)
Severe with ulceration or
cerebral edema;
tracheotomy or feeding
tube indicated
Death
Edema:
limb
Edema: limb 5 – 10% inter-limb
discrepancy in volume or
circumference at point of
greatest visible
difference; swelling or
obscuration of anatomic
architecture on close
inspection; pitting edema
>10 – 30% inter-limb
discrepancy in volume or
circumference at point of
greatest visible
difference; readily
apparent obscuration of
anatomic architecture;
obliteration of skin folds;
readily apparent
deviation from normal
anatomic contour
>30% inter-limb
discrepancy in volume;
lymphorrhea; gross
deviation from normal
anatomic contour;
interfering with ADL
Progression to
malignancy (i.e.,
lymphangiosarcoma);
amputation indicated;
disabling
Death
Edema:
trunk/genital
Edema: trunk/genital Swelling or obscuration
of anatomic architecture
on close inspection;
pitting edema
Readily apparent
obscuration of anatomic
architecture; obliteration
of skin folds; readily
apparent deviation from
normal anatomic contour
Lymphorrhea; interfering
with ADL; gross deviation
from normal anatomic
contour
Progression to
malignancy (i.e.,
lymphangiosarcoma);
disabling
Death
Edema:
viscera
Edema: viscera Asymptomatic; clinical or
radiographic findings only
Symptomatic; medical
intervention indicated
Symptomatic and unable
to aliment adequately
orally; interventional
radiology or operative
intervention indicated
Life-threatening
consequences
Death
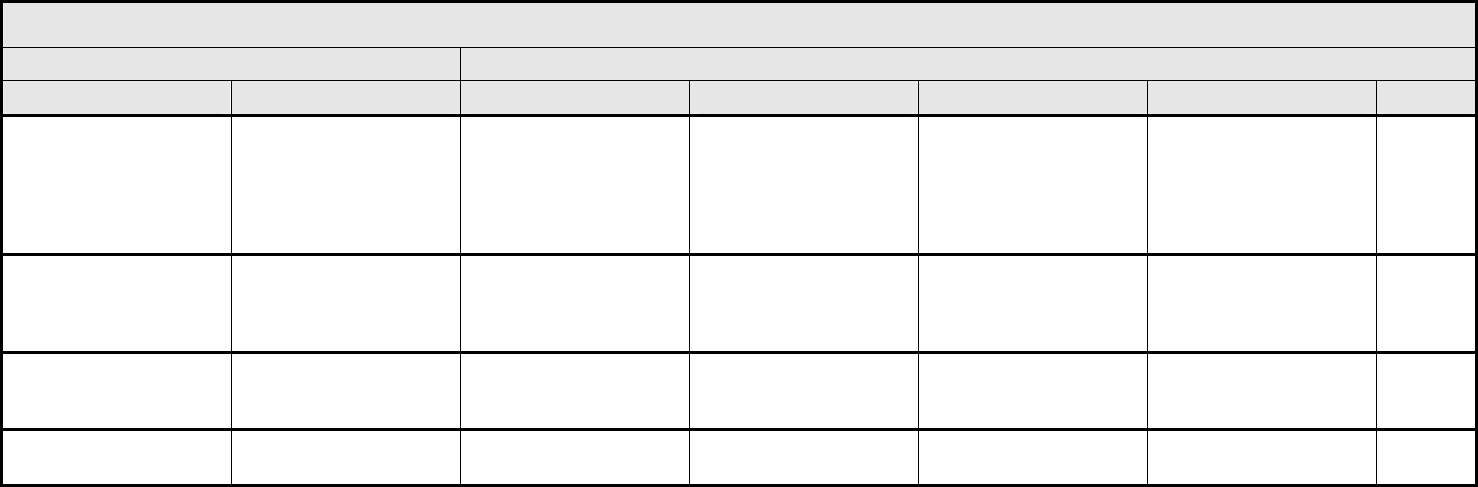
LYMPHATICS Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 39 - March 31, 2003, Publish Date: August 9, 2006
Lymphedema-related
fibrosis
Lymphedema-related
fibrosis
Minimal to moderate
redundant soft tissue,
unresponsive to elevation
or compression, with
moderately firm texture or
spongy feel
Marked increase in
density and firmness,
with or without tethering
Very marked density and
firmness with tethering
affecting ≥40% of the
edematous area
— —
Lymphocele Lymphocele Asymptomatic, clinical or
radiographic findings only
Symptomatic; medical
intervention indicated
Symptomatic and
interventional radiology
or operative intervention
indicated
— —
Phlebolymphatic cording Phlebolymphatic cording Asymptomatic, clinical
findings only
Symptomatic; medical
intervention indicated
Symptomatic and leading
to contracture or reduced
range of motion
— —
Lymphatics – Other
(Specify, __)
Lymphatics – Other
(Specify)
Mild Moderate Severe Life-threatening;
disabling
Death
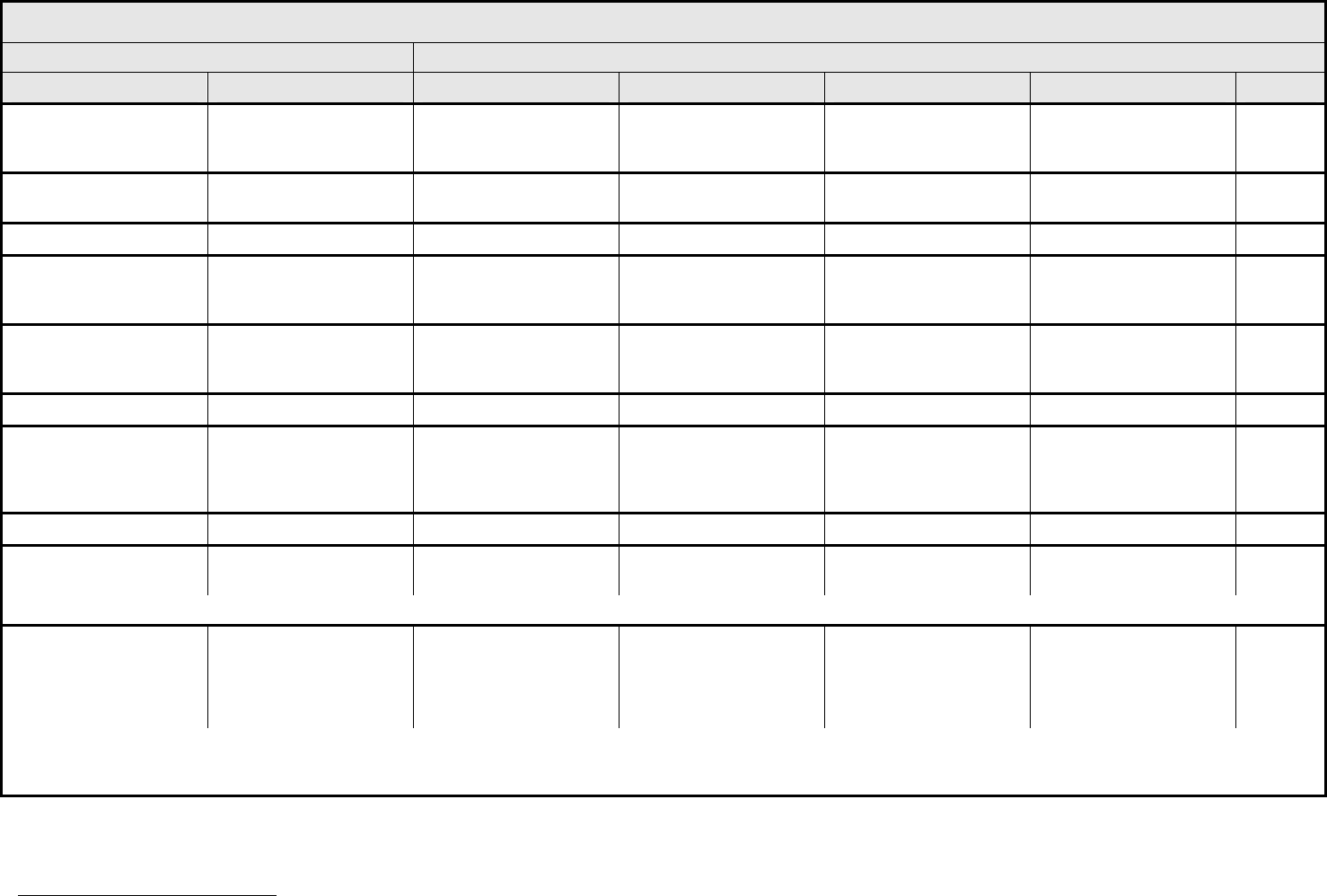
METABOLIC/LABORATORY Page 1 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 40 - March 31, 2003, Publish Date: August 9, 2006
Acidosis
(metabolic or respiratory)
Acidosis
pH <normal, but ≥7.3
— pH <7.3 pH <7.3 with life-
threatening
consequences
Death
Albumin, serum-low
(hypoalbuminemia)
Hypoalbuminemia <LLN – 3 g/dL
<LLN – 30 g/L
<3 – 2 g/dL
<30 – 20 g/L
<2 g/dL
<20 g/L
— Death
Alkaline phosphatase Alkaline phosphatase >ULN – 2.5 x ULN >2.5 – 5.0 x ULN >5.0 – 20.0 x ULN >20.0 x ULN —
Alkalosis
(metabolic or respiratory)
Alkalosis
pH >normal, but ≤7.5
— pH >7.5 pH >7.5 with life-
threatening
consequences
Death
ALT, SGPT
(serum glutamic pyruvic
transaminase)
ALT >ULN – 2.5 x ULN >2.5 – 5.0 x ULN >5.0 – 20.0 x ULN >20.0 x ULN —
Amylase Amylase >ULN – 1.5 x ULN >1.5 – 2.0 x ULN >2.0 – 5.0 x ULN >5.0 x ULN —
AST, SGOT
(serum glutamic
oxaloacetic
transaminase)
AST >ULN – 2.5 x ULN >2.5 – 5.0 x ULN >5.0 – 20.0 x ULN >20.0 x ULN —
Bicarbonate, serum-low Bicarbonate, serum-low <LLN – 16 mmol/L <16 – 11 mmol/L <11 – 8 mmol/L <8 mmol/L Death
Bilirubin
(hyperbilirubinemia)
Bilirubin >ULN – 1.5 x ULN >1.5 – 3.0 x ULN >3.0 – 10.0 x ULN >10.0 x ULN —
REMARK: Jaundice is not an AE, but may be a manifestation of liver dysfunction/failure or elevated bilirubin. If jaundice is associated with elevated bilirubin, grade bilirubin.
Calcium, serum-low
(hypocalcemia)
Hypocalcemia <LLN – 8.0 mg/dL
<LLN – 2.0 mmol/L
Ionized calcium:
<LLN – 1.0 mmol/L
<8.0 – 7.0 mg/dL
<2.0 – 1.75 mmol/L
Ionized calcium:
<1.0 – 0.9 mmol/L
<7.0 – 6.0 mg/dL
<1.75 – 1.5 mmol/L
Ionized calcium:
<0.9 – 0.8 mmol/L
<6.0 mg/dL
<1.5 mmol/L
Ionized calcium:
<0.8 mmol/L
Death
REMARK: Calcium can be falsely low if hypoalbuminemia is present. Serum albumin is <4.0 g/dL, hypocalcemia is reported after the following corrective calculation has been
performed: Corrected Calcium (mg/dL) = Total Calcium (mg/dL) – 0.8 [Albumin (g/dL) – 4]
4
. Alternatively, direct measurement of ionized calcium is the definitive method to diagnose
metabolically relevant alterations in serum calcium.
4
Crit Rev Clin Lab Sci 1984;21(1):51-97
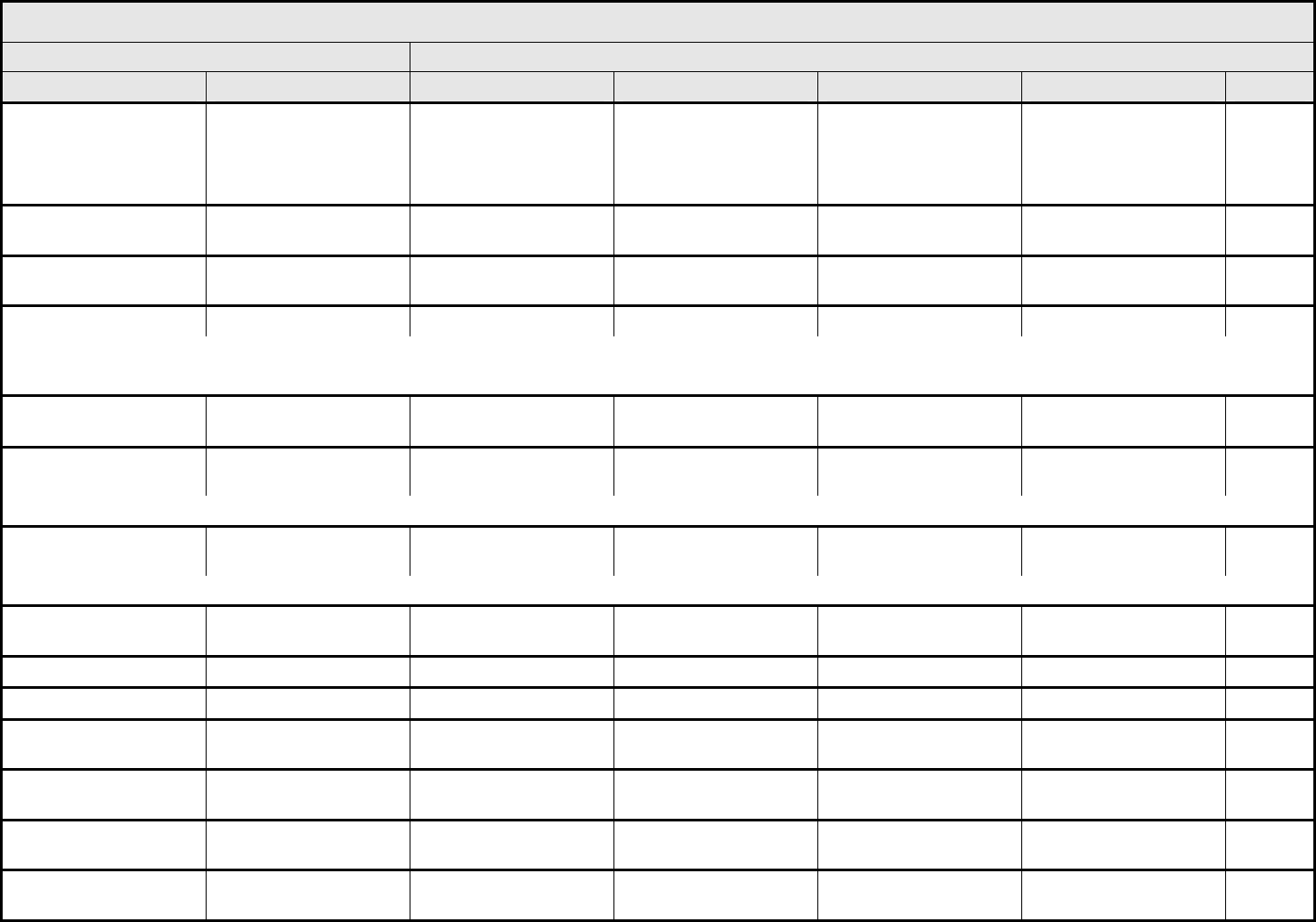
METABOLIC/LABORATORY Page 2 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 41 - March 31, 2003, Publish Date: August 9, 2006
Calcium, serum-high
(hypercalcemia)
Hypercalcemia >ULN – 11.5 mg/dL
>ULN – 2.9 mmol/L
Ionized calcium:
>ULN – 1.5 mmol/L
>11.5 – 12.5 mg/dL
>2.9 – 3.1 mmol/L
Ionized calcium:
>1.5 – 1.6 mmol/L
>12.5 – 13.5 mg/dL
>3.1 – 3.4 mmol/L
Ionized calcium:
>1.6 – 1.8 mmol/L
>13.5 mg/dL
>3.4 mmol/L
Ionized calcium:
>1.8 mmol/L
Death
Cholesterol, serum-high
(hypercholesteremia)
Cholesterol >ULN – 300 mg/dL
>ULN – 7.75 mmol/L
>300 – 400 mg/dL
>7.75 – 10.34 mmol/L
>400 – 500 mg/dL
>10.34 – 12.92 mmol/L
>500 mg/dL
>12.92 mmol/L
Death
CPK (creatine
phosphokinase)
CPK >ULN – 2.5 x ULN >2.5 x ULN – 5 x ULN >5 x ULN – 10 x ULN >10 x ULN Death
Creatinine Creatinine >ULN – 1.5 x ULN >1.5 – 3.0 x ULN >3.0 – 6.0 x ULN >6.0 x ULN Death
REMARK: Adjust to age-appropriate levels for pediatric patients.
A
LSO CONSIDER: Glomerular filtration rate.
GGT (γ-Glutamyl
transpeptidase)
GGT >ULN – 2.5 x ULN >2.5 – 5.0 x ULN >5.0 – 20.0 x ULN >20.0 x ULN —
Glomerular filtration rate GFR <75 – 50% LLN <50 – 25% LLN <25% LLN, chronic
dialysis not indicated
Chronic dialysis or renal
transplant indicated
Death
ALSO CONSIDER: Creatinine.
Glucose, serum-high
(hyperglycemia)
Hyperglycemia >ULN – 160 mg/dL
>ULN – 8.9 mmol/L
>160 – 250 mg/dL
>8.9 – 13.9 mmol/L
>250 – 500 mg/dL
>13.9 – 27.8 mmol/L
>500 mg/dL
>27.8 mmol/L or acidosis
Death
REMARK: Hyperglycemia, in general, is defined as fasting unless otherwise specified in protocol.
Glucose, serum-low
(hypoglycemia)
Hypoglycemia <LLN – 55 mg/dL
<LLN – 3.0 mmol/L
<55 – 40 mg/dL
<3.0 – 2.2 mmol/L
<40 – 30 mg/dL
<2.2 – 1.7 mmol/L
<30 mg/dL
<1.7 mmol/L
Death
Hemoglobinuria Hemoglobinuria Present — — — Death
Lipase Lipase >ULN – 1.5 x ULN >1.5 – 2.0 x ULN >2.0 – 5.0 x ULN >5.0 x ULN —
Magnesium, serum-high
(hypermagnesemia)
Hypermagnesemia >ULN – 3.0 mg/dL
>ULN – 1.23 mmol/L
— >3.0 – 8.0 mg/dL
>1.23 – 3.30 mmol/L
>8.0 mg/dL
>3.30 mmol/L
Death
Magnesium, serum-low
(hypomagnesemia)
Hypomagnesemia <LLN – 1.2 mg/dL
<LLN – 0.5 mmol/L
<1.2 – 0.9 mg/dL
<0.5 – 0.4 mmol/L
<0.9 – 0.7 mg/dL
<0.4 – 0.3 mmol/L
<0.7 mg/dL
<0.3 mmol/L
Death
Phosphate, serum-low
(hypophosphatemia)
Hypophosphatemia <LLN – 2.5 mg/dL
<LLN – 0.8 mmol/L
<2.5 – 2.0 mg/dL
<0.8 – 0.6 mmol/L
<2.0 – 1.0 mg/dL
<0.6 – 0.3 mmol/L
<1.0 mg/dL
<0.3 mmol/L
Death
Potassium, serum-high
(hyperkalemia)
Hyperkalemia >ULN – 5.5 mmol/L >5.5 – 6.0 mmol/L >6.0 – 7.0 mmol/L >7.0 mmol/L Death
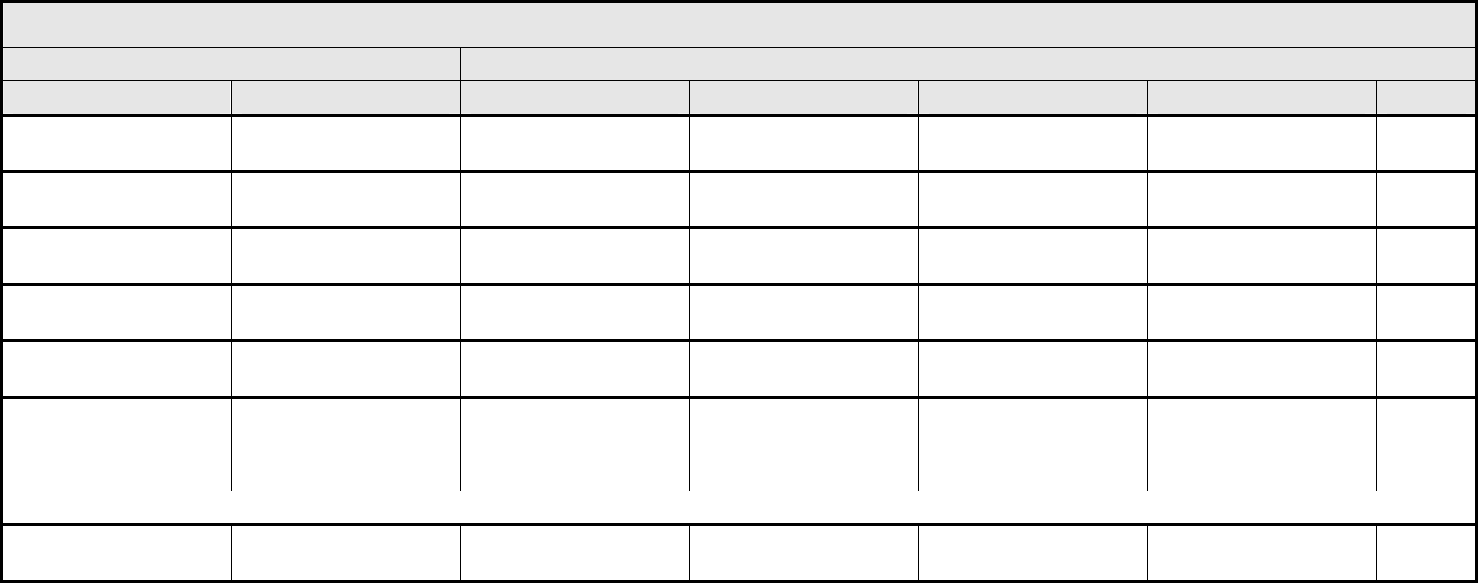
METABOLIC/LABORATORY Page 3 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 42 - March 31, 2003, Publish Date: August 9, 2006
Potassium, serum-low
(hypokalemia)
Hypokalemia <LLN – 3.0 mmol/L — <3.0 – 2.5 mmol/L <2.5 mmol/L Death
Proteinuria Proteinuria 1+ or
0.15 – 1.0 g/24 hrs
2+ to 3+ or
>1.0 – 3.5 g/24 hrs
4+ or
>3.5 g/24 hrs
Nephrotic syndrome Death
Sodium, serum-high
(hypernatremia)
Hypernatremia >ULN – 150 mmol/L >150 – 155 mmol/L >155 – 160 mmol/L >160 mmol/L Death
Sodium, serum-low
(hyponatremia)
Hyponatremia <LLN – 130 mmol/L — <130 – 120 mmol/L <120 mmol/L Death
Triglyceride, serum-high
(hypertriglyceridemia)
Hypertriglyceridemia >ULN – 2.5 x ULN >2.5 – 5.0 x ULN >5.0 – 10 x ULN >10 x ULN Death
Uric acid, serum-high
(hyperuricemia)
Hyperuricemia
>ULN – 10 mg/dL
≤0.59 mmol/L without
physiologic
consequences
—
>ULN – 10 mg/dL
≤0.59 mmol/L with
physiologic
consequences
>10 mg/dL
>0.59 mmol/L
Death
ALSO CONSIDER: Creatinine; Potassium, serum-high (hyperkalemia); Renal failure; Tumor lysis syndrome.
Metabolic/Laboratory –
Other (Specify, __)
Metabolic/Lab – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
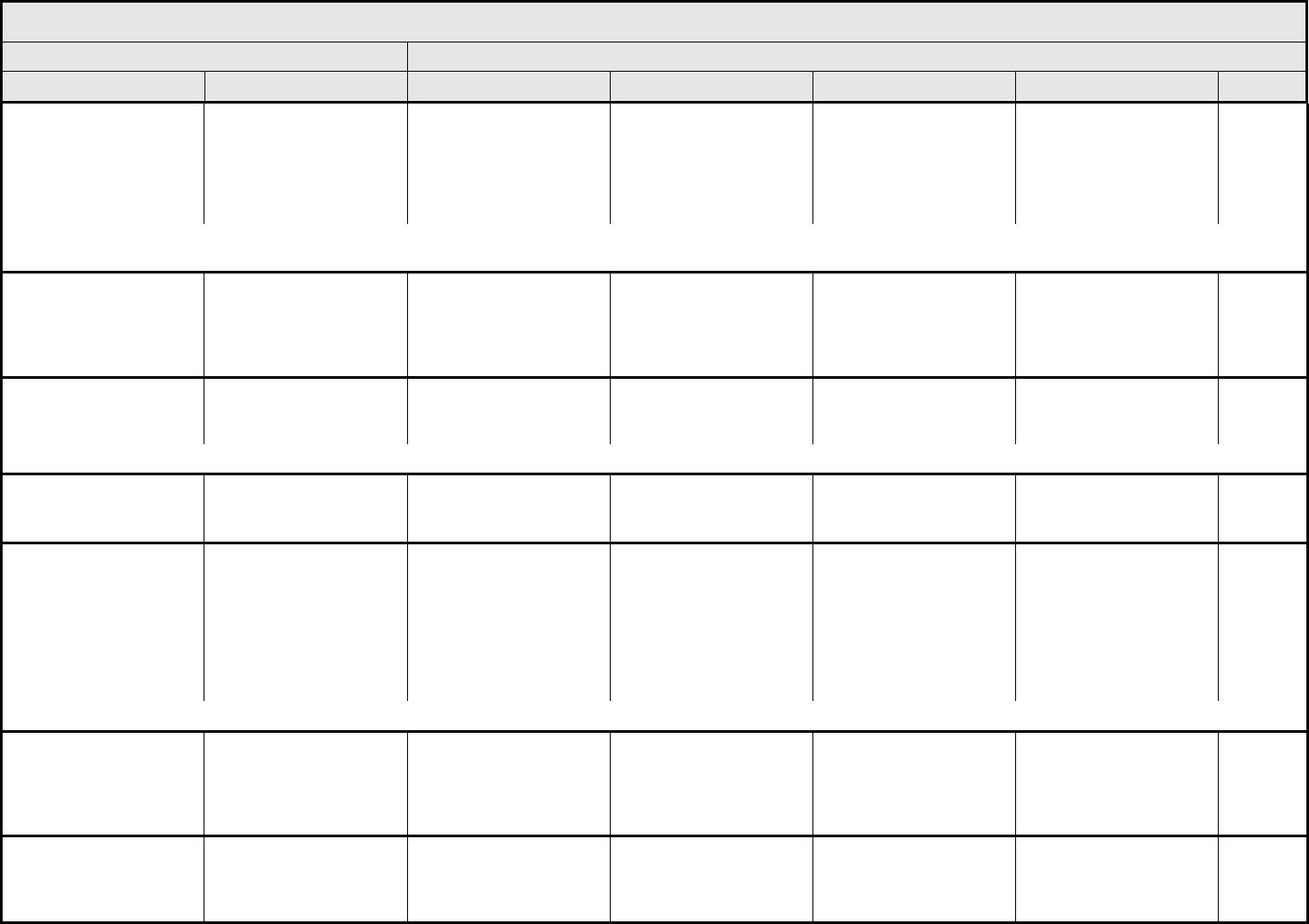
MUSCULOSKELETAL/SOFT TISSUE Page 1 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 43 - March 31, 2003, Publish Date: August 9, 2006
Arthritis (non-septic) Arthritis Mild pain with
inflammation, erythema,
or joint swelling, but not
interfering with function
Moderate pain with
inflammation, erythema,
or joint swelling
interfering with function,
but not interfering with
ADL
Severe pain with
inflammation, erythema,
or joint swelling and
interfering with ADL
Disabling Death
REMARK: Report only when the diagnosis of arthritis (e.g., inflammation of a joint or a state characterized by inflammation of joints) is made. Arthralgia (sign or symptom of pain in a
joint, especially non-inflammatory in character) is graded as Pain – Select in the PAIN CATEGORY.
Bone:
spine-scoliosis
Scoliosis
≤20 degrees; clinically
undetectable
>20 – 45 degrees; visible
by forward flexion;
interfering with function
but not interfering with
ADL
>45 degrees; scapular
prominence in forward
flexion; operative
intervention indicated;
interfering with ADL
Disabling (e.g., interfering
with cardiopulmonary
function)
Death
Cervical spine-range of
motion
Cervical spine ROM Mild restriction of rotation
or flexion between
60 – 70 degrees
Rotation <60 degrees to
right or left; <60 degrees
of flexion
Ankylosed/fused over
multiple segments with
no C-spine rotation
— —
REMARK: 60 – 65 degrees of rotation is required for reversing a car; 60 – 65 degrees of flexion is required to tie shoes.
Exostosis Exostosis Asymptomatic Involving multiple sites;
pain or interfering with
function
Excision indicated Progression to
malignancy (i.e.,
chondrosarcoma)
Death
Extremity-lower
(gait/walking)
Gait/walking Limp evident only to
trained observer and able
to walk ≥1 kilometer;
cane indicated for
walking
Noticeable limp, or
limitation of limb function,
but able to walk ≥0.1
kilometer (1 city block);
quad cane indicated for
walking
Severe limp with stride
modified to maintain
balance (widened base of
support, marked
reduction in step length);
ambulation limited to
walker; crutches
indicated
Unable to walk —
ALSO CONSIDER: Ataxia (incoordination); Muscle weakness, generalized or specific area (not due to neuropathy) – Select.
Extremity-upper
(function)
Extremity-upper
(function)
Able to perform most
household or work
activities with affected
limb
Able to perform most
household or work
activities with
compensation from
unaffected limb
Interfering with ADL Disabling; no function of
affected limb
—
Fibrosis-cosmesis Fibrosis-cosmesis Visible only on close
examination
Readily apparent but not
disfiguring
Significant disfigurement;
operative intervention
indicated if patient
chooses
— —
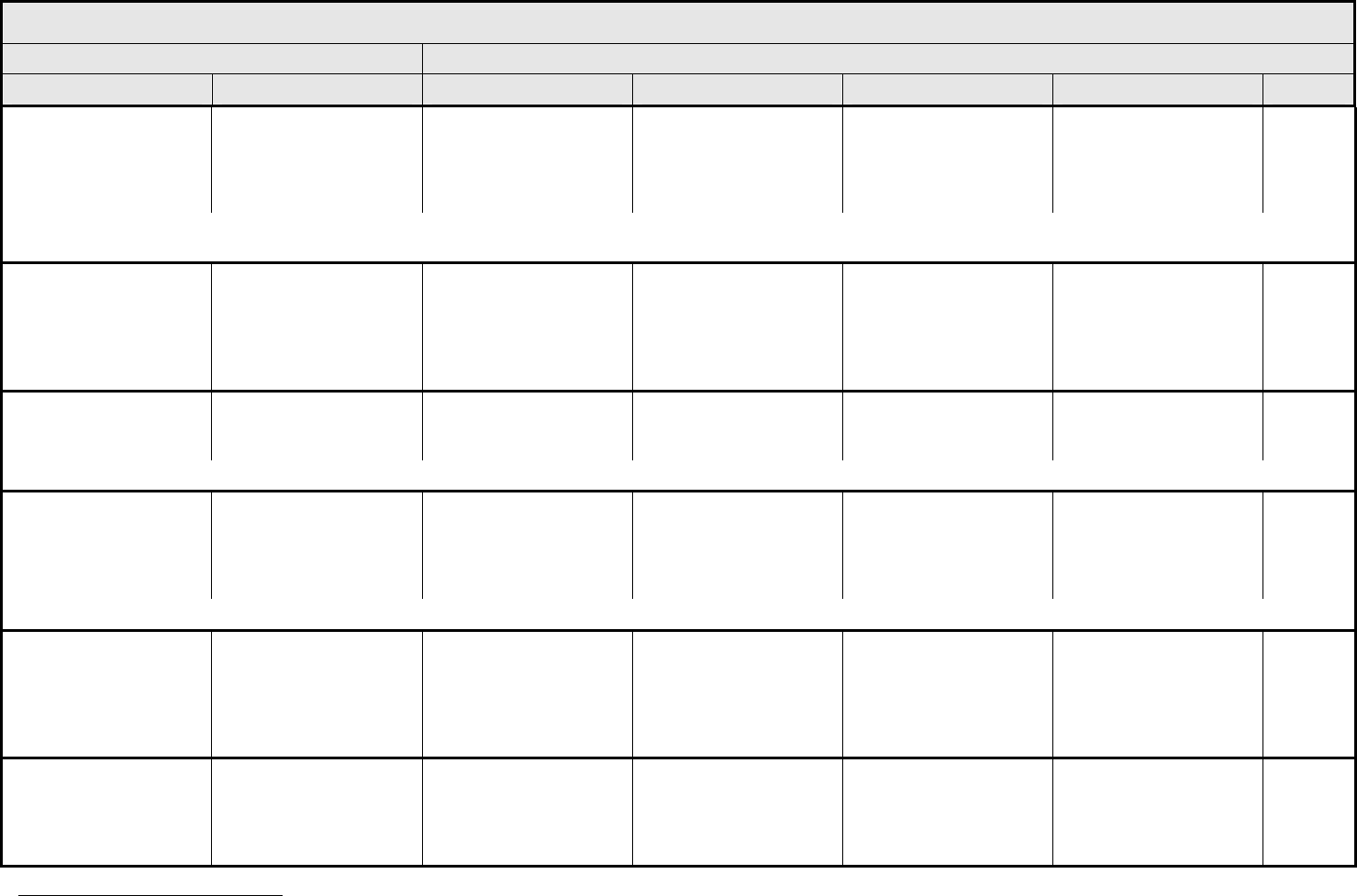
MUSCULOSKELETAL/SOFT TISSUE Page 2 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 44 - March 31, 2003, Publish Date: August 9, 2006
Fibrosis-deep connective
tissue
Fibrosis-deep connective
tissue
Increased density,
“spongy” feel
Increased density with
firmness or tethering
Increased density with
fixation of tissue;
operative intervention
indicated; interfering with
ADL
Life-threatening;
disabling; loss of limb;
interfering with vital organ
function
Death
ALSO CONSIDER: Induration/fibrosis (skin and subcutaneous tissue); Muscle weakness, generalized or specific area (not due to neuropathy) – Select; Neuropathy: motor; Neuropathy:
sensory.
Fracture Fracture Asymptomatic,
radiographic findings only
(e.g., asymptomatic rib
fracture on plain x-ray,
pelvic insufficiency
fracture on MRI, etc.)
Symptomatic but non-
displaced; immobilization
indicated
Symptomatic and
displaced or open wound
with bone exposure;
operative intervention
indicated
Disabling; amputation
indicated
Death
Joint-effusion Joint-effusion Asymptomatic, clinical or
radiographic findings only
Symptomatic; interfering
with function but not
interfering with ADL
Symptomatic and
interfering with ADL
Disabling Death
ALSO CONSIDER: Arthritis (non-septic).
Joint-function
5
Joint-function Stiffness interfering with
athletic activity; ≤25%
loss of range of motion
(ROM)
Stiffness interfering with
function but not
interfering with ADL;
>25 – 50% decrease in
ROM
Stiffness interfering with
ADL; >50 – 75%
decrease in ROM
Fixed or non-functional
joint (arthrodesis);
>75% decrease in ROM
—
ALSO CONSIDER: Arthritis (non-septic).
Local complication –
device/prosthesis-related
Device/prosthesis Asymptomatic Symptomatic, but not
interfering with ADL; local
wound care; medical
intervention indicated
Symptomatic, interfering
with ADL; operative
intervention indicated
(e.g., hardware/device
replacement or removal,
reconstruction)
Life-threatening;
disabling; loss of limb or
organ
Death
Lumbar spine-range of
motion
Lumbar spine ROM Stiffness and difficulty
bending to the floor to
pick up a very light object
but able to do activity
Some lumbar spine
flexion but requires a
reaching aid to pick up a
very light object from the
floor
Ankylosed/fused over
multiple segments with
no L-spine flexion (i.e.,
unable to reach to floor to
pick up a very light
— —
5
Adapted from the International SFTR Method of Measuring and Recording Joint Motion, International Standard Orthopedic Measurements (ISOM), Jon J. Gerhardt and Otto A.
Russee, Bern, Switzerland, Han Huber 9 Publisher), 1975.
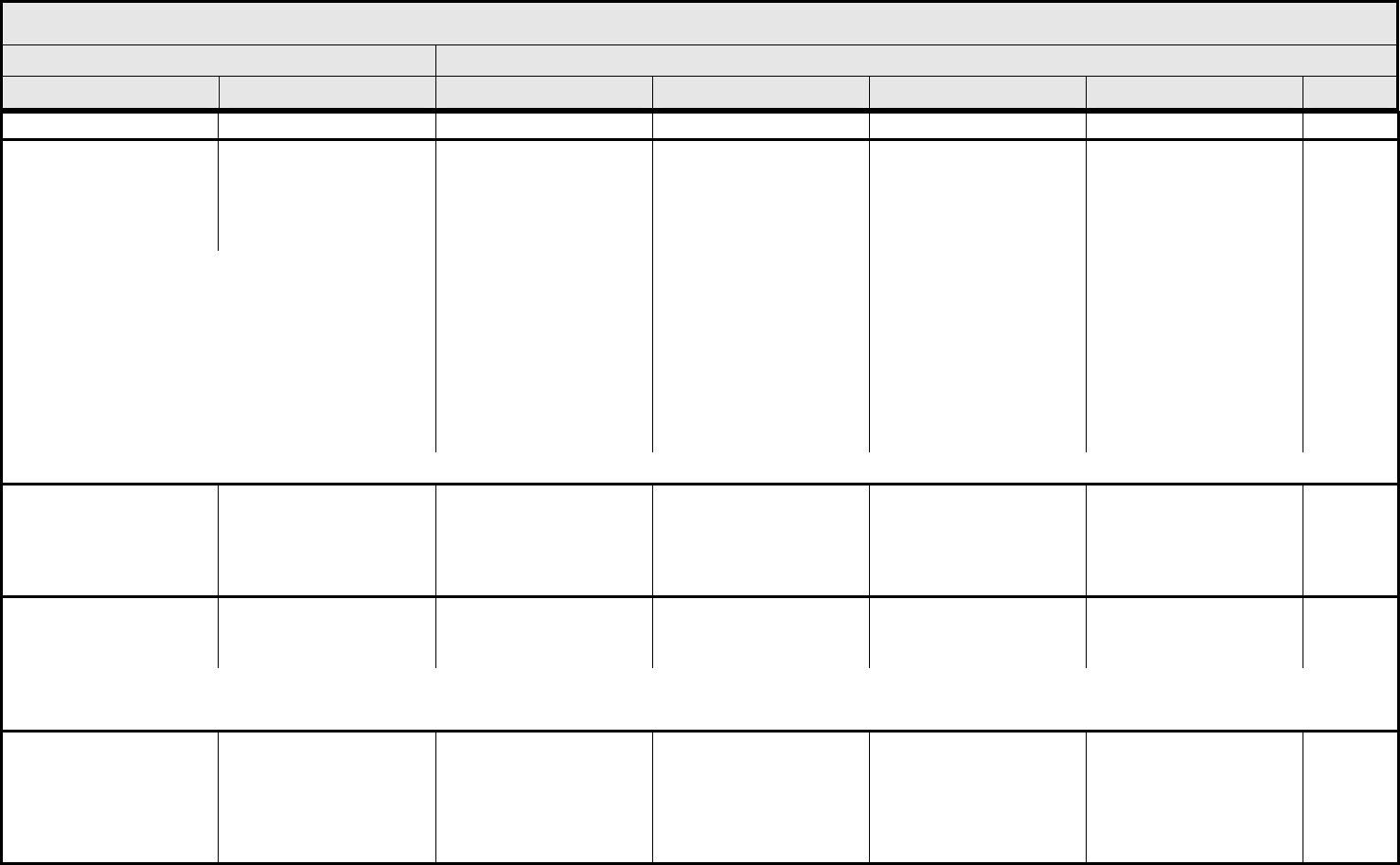
MUSCULOSKELETAL/SOFT TISSUE Page 3 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 45 - March 31, 2003, Publish Date: August 9, 2006
object)
Muscle weakness,
generalized or specific
area (not due to
neuropathy)
– Select:
Muscle weakness
– Select
– Extraocular
– Extremity-lower
– Extremity-upper
– Facial
– Left-sided
– Ocular
– Pelvic
– Right-sided
– Trunk
– Whole body/generalized
Asymptomatic, weakness
on physical exam
Symptomatic and
interfering with function,
but not interfering with
ADL
Symptomatic and
interfering with ADL
Life-threatening; disabling Death
ALSO CONSIDER: Fatigue (asthenia, lethargy, malaise).
Muscular/skeletal
hypoplasia
Muscular/skeletal
hypoplasia
Cosmetically and
functionally insignificant
hypoplasia
Deformity, hypoplasia, or
asymmetry able to be
remediated by prosthesis
(e.g., shoe insert) or
covered by clothing
Functionally significant
deformity, hypoplasia, or
asymmetry, unable to be
remediated by prosthesis
or covered by clothing
Disabling —
Myositis
(inflammation/damage of
muscle)
Myositis Mild pain, not interfering
with function
Pain interfering with
function, but not
interfering with ADL
Pain interfering with ADL Disabling Death
REMARK: Myositis implies muscle damage (i.e., elevated CPK).
A
LSO CONSIDER: CPK (creatine phosphokinase); Pain – Select.
Osteonecrosis
(avascular necrosis)
Osteonecrosis Asymptomatic,
radiographic findings only
Symptomatic and
interfering with function,
but not interfering with
ADL; minimal bone
removal indicated (i.e.,
minor sequestrectomy)
Symptomatic and
interfering with ADL;
operative intervention or
hyperbaric oxygen
indicated
Disabling Death

MUSCULOSKELETAL/SOFT TISSUE Page 4 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 46 - March 31, 2003, Publish Date: August 9, 2006
Osteoporosis
6
Osteoporosis Radiographic evidence of
osteoporosis or Bone
Mineral Density (BMD)
t-score –1 to –2.5
(osteopenia) and no loss
of height or therapy
indicated
BMD t-score < –2.5; loss
of height <2 cm; anti-
osteoporotic therapy
indicated
Fractures; loss of height
≥2 cm
Disabling Death
Seroma Seroma Asymptomatic Symptomatic; medical
intervention or simple
aspiration indicated
Symptomatic,
interventional radiology
or operative intervention
indicated
— —
Soft tissue necrosis
– Select:
– Abdomen
– Extremity-lower
– Extremity-upper
– Head
– Neck
– Pelvic
– Thorax
Soft tissue necrosis
– Select
— Local wound care;
medical intervention
indicated
Operative debridement or
other invasive
intervention indicated
(e.g., hyperbaric oxygen)
Life-threatening
consequences; major
invasive intervention
indicated (e.g., tissue
reconstruction, flap, or
grafting)
Death
Trismus (difficulty,
restriction or pain when
opening mouth)
Trismus Decreased range of
motion without impaired
eating
Decreased range of
motion requiring small
bites, soft foods or
purees
Decreased range of
motion with inability to
adequately aliment or
hydrate orally
— —
NAVIGATION NOTE: Wound-infectious is graded as Infection – Select in the INFECTION CATEGORY.
NAVIGATION NOTE: Wound non-infectious is graded as Wound complication, non-infectious in the DERMATOLOGY/SKIN CATEGORY.
Musculoskeletal/Soft
Tissue – Other
(Specify, __)
Musculoskeletal – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
6
"Assessment of Fracture Risk and its Application to Screening for Postmenopausal Osteoporosis," Report of a WHO Study Group Technical Report Series, No. 843, 1994, v + 129
pages [C*, E, F, R, S], ISBN 92 4 120843 0, Sw.fr. 22.-/US $19.80; in developing countries: Sw.fr. 15.40, Order no. 1100843
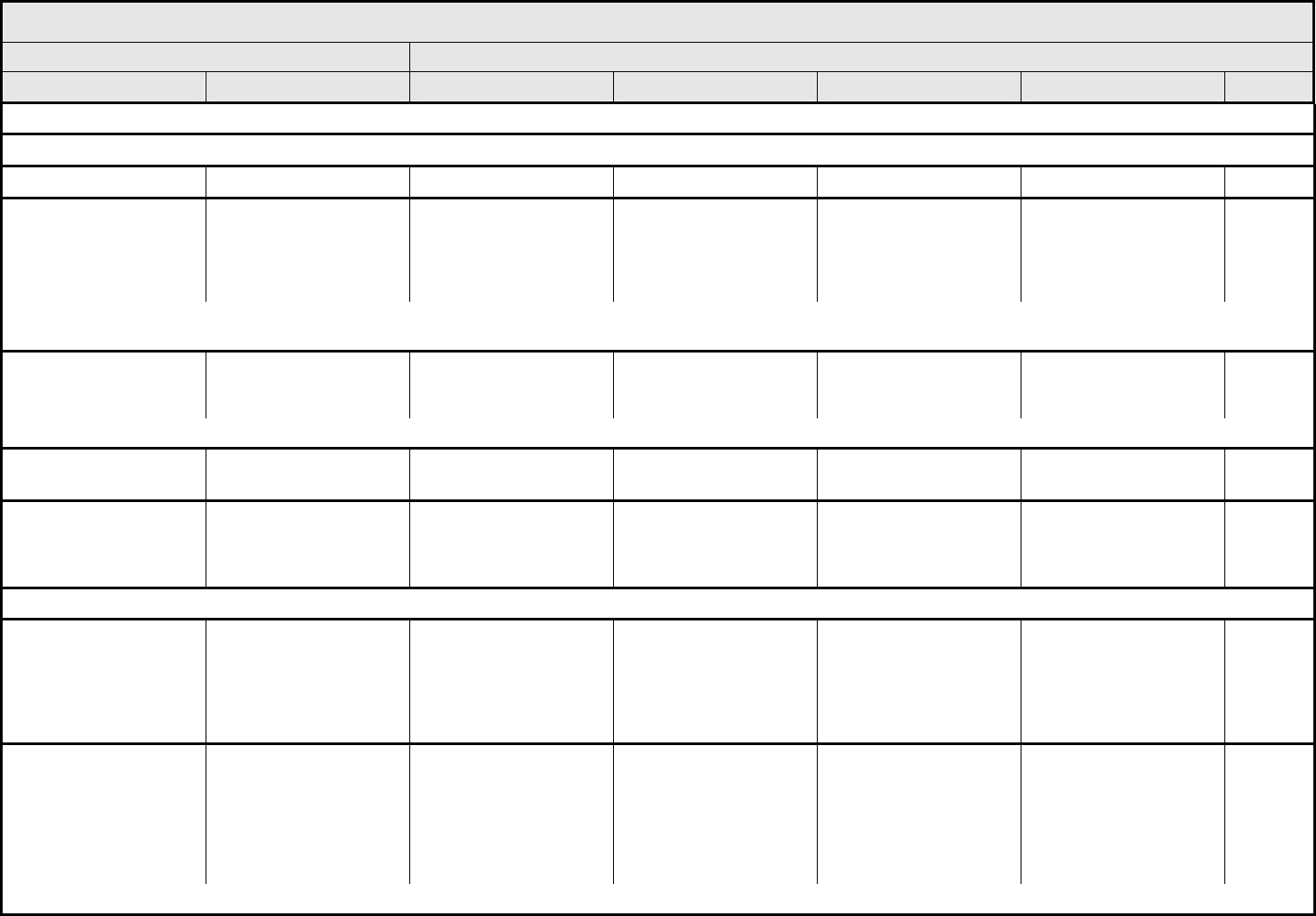
NEUROLOGY Page 1 of 5
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 47 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: ADD (Attention Deficit Disorder) is graded as Cognitive disturbance.
NAVIGATION NOTE: Aphasia, receptive and/or expressive, is graded as Speech impairment (e.g., dysphasia or aphasia).
Apnea Apnea — — Present Intubation indicated Death
Arachnoiditis/
meningismus/radiculitis
Arachnoiditis Symptomatic, not
interfering with function;
medical intervention
indicated
Symptomatic (e.g.,
photophobia, nausea)
interfering with function
but not interfering with
ADL
Symptomatic, interfering
with ADL
Life-threatening; disabling
(e.g., paraplegia)
Death
ALSO CONSIDER: Fever (in the absence of neutropenia, where neutropenia is defined as ANC <1.0 x 10
9
/L); Infection (documented clinically or microbiologically) with Grade 3 or 4
neutrophils (ANC <1.0 x 109/L) – Select; Infection with normal ANC or Grade 1 or 2 neutrophils – Select; Infection with unknown ANC – Select; Pain – Select; Vomiting.
Ataxia (incoordination) Ataxia Asymptomatic Symptomatic, not
interfering with ADL
Symptomatic, interfering
with ADL; mechanical
assistance indicated
Disabling Death
REMARK: Ataxia (incoordination) refers to the consequence of medical or operative intervention.
Brachial plexopathy Brachial plexopathy Asymptomatic Symptomatic, not
interfering with ADL
Symptomatic, interfering
with ADL
Disabling Death
CNS cerebrovascular
ischemia
CNS ischemia — Asymptomatic,
radiographic findings only
Transient ischemic event
or attack (TIA) ≤24 hrs
duration
Cerebral vascular
accident (CVA, stroke),
neurologic deficit
>24 hrs
Death
NAVIGATION NOTE: CNS hemorrhage/bleeding is graded as Hemorrhage, CNS in the HEMORRHAGE/BLEEDING CATEGORY.
CNS necrosis/cystic
progression
CNS necrosis Asymptomatic,
radiographic findings only
Symptomatic, not
interfering with ADL;
medical intervention
indicated
Symptomatic and
interfering with ADL;
hyperbaric oxygen
indicated
Life-threatening;
disabling; operative
intervention indicated to
prevent or treat CNS
necrosis/cystic
progression
Death
Cognitive disturbance Cognitive disturbance Mild cognitive disability;
not interfering with
work/school/life
performance; specialized
educational
services/devices not
indicated
Moderate cognitive
disability; interfering with
work/school/life
performance but capable
of independent living;
specialized resources on
part-time basis indicated
Severe cognitive
disability; significant
impairment of
work/school/life
performance
Unable to perform ADL;
full-time specialized
resources or
institutionalization
indicated
Death
REMARK: Cognitive disturbance may be used for Attention Deficit Disorder (ADD).
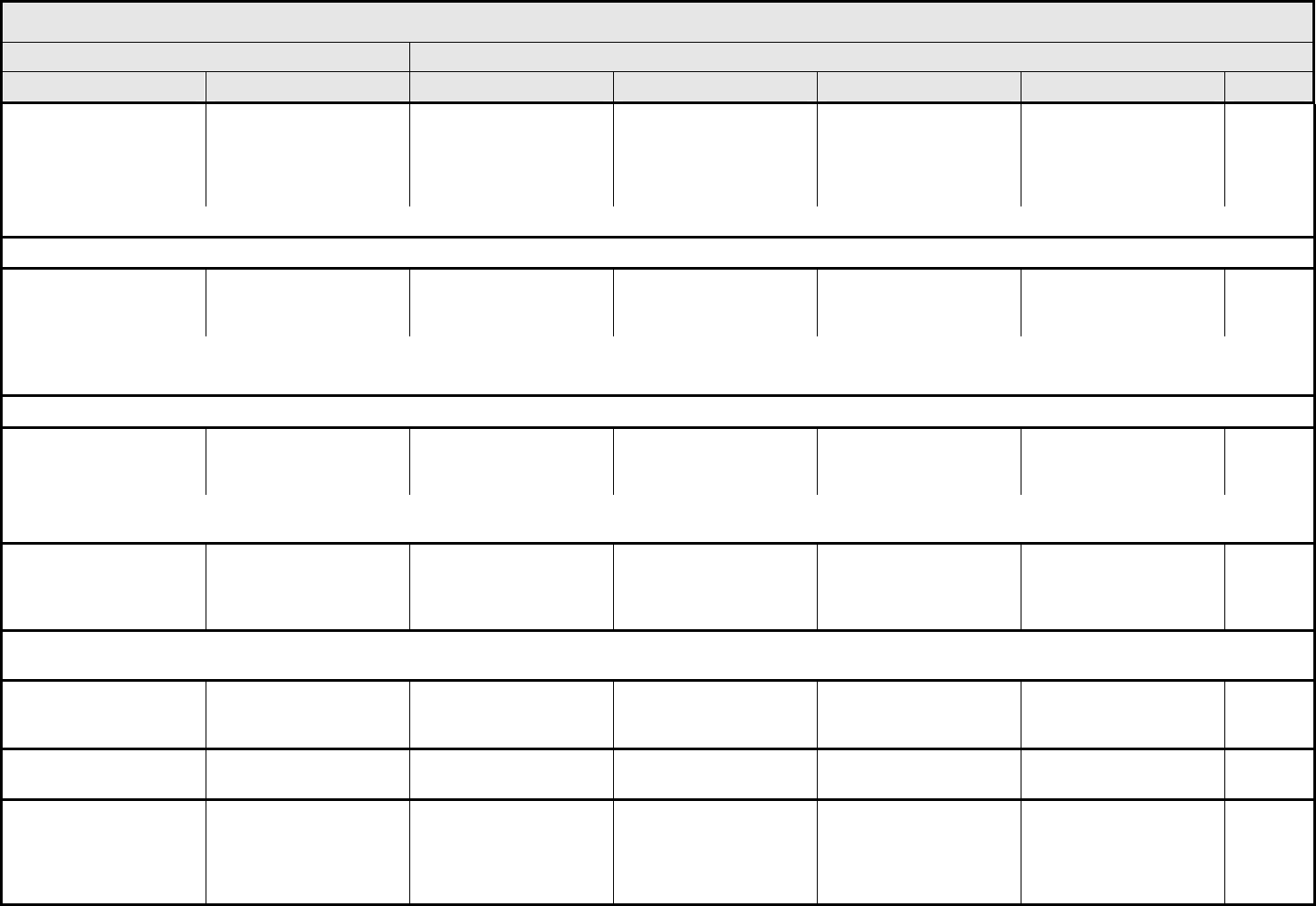
NEUROLOGY Page 2 of 5
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 48 - March 31, 2003, Publish Date: August 9, 2006
Confusion Confusion
Transient confusion,
disorientation, or attention
deficit
Confusion, disorientation,
or attention deficit
interfering with function,
but not interfering with
ADL
Confusion or delirium
interfering with ADL
Harmful to others or self;
hospitalization indicated
Death
REMARK: Attention Deficit Disorder (ADD) is graded as Cognitive disturbance.
NAVIGATION NOTE: Cranial neuropathy is graded as Neuropathy-cranial – Select.
Dizziness Dizziness With head movements or
nystagmus only; not
interfering with function
Interfering with function,
but not interfering with
ADL
Interfering with ADL Disabling —
REMARK: Dizziness includes disequilibrium, lightheadedness, and vertigo.
ALSO CONSIDER: Neuropathy: cranial – Select; Syncope (fainting).
NAVIGATION NOTE: Dysphasia, receptive and/or expressive, is graded as Speech impairment (e.g., dysphasia or aphasia).
Encephalopathy Encephalopathy — Mild signs or symptoms;
not interfering with ADL
Signs or symptoms
interfering with ADL;
hospitalization indicated
Life-threatening; disabling Death
ALSO CONSIDER: Cognitive disturbance; Confusion; Dizziness; Memory impairment; Mental status; Mood alteration – Select; Psychosis (hallucinations/delusions);
Somnolence/depressed level of consciousness.
Extrapyramidal/
involuntary movement/
restlessness
Involuntary movement Mild involuntary
movements not
interfering with function
Moderate involuntary
movements interfering
with function, but not
interfering with ADL
Severe involuntary
movements or torticollis
interfering with ADL
Disabling
Death
NAVIGATION NOTE: Headache/neuropathic pain (e.g., jaw pain, neurologic pain, phantom limb pain, post-infectious neuralgia, or painful neuropathies) is graded as Pain – Select in the
PAIN CATEGORY.
Hydrocephalus Hydrocephalus Asymptomatic,
radiographic findings only
Mild to moderate
symptoms not interfering
with ADL
Severe symptoms or
neurological deficit
interfering with ADL
Disabling Death
Irritability (children <3
years of age)
Irritability Mild; easily consolable Moderate; requiring
increased attention
Severe; inconsolable — —
Laryngeal nerve
dysfunction
Laryngeal nerve Asymptomatic, weakness
on clinical
examination/testing only
Symptomatic, but not
interfering with ADL;
intervention not indicated
Symptomatic, interfering
with ADL; intervention
indicated (e.g.,
thyroplasty, vocal cord
injection)
Life-threatening;
tracheostomy indicated
Death
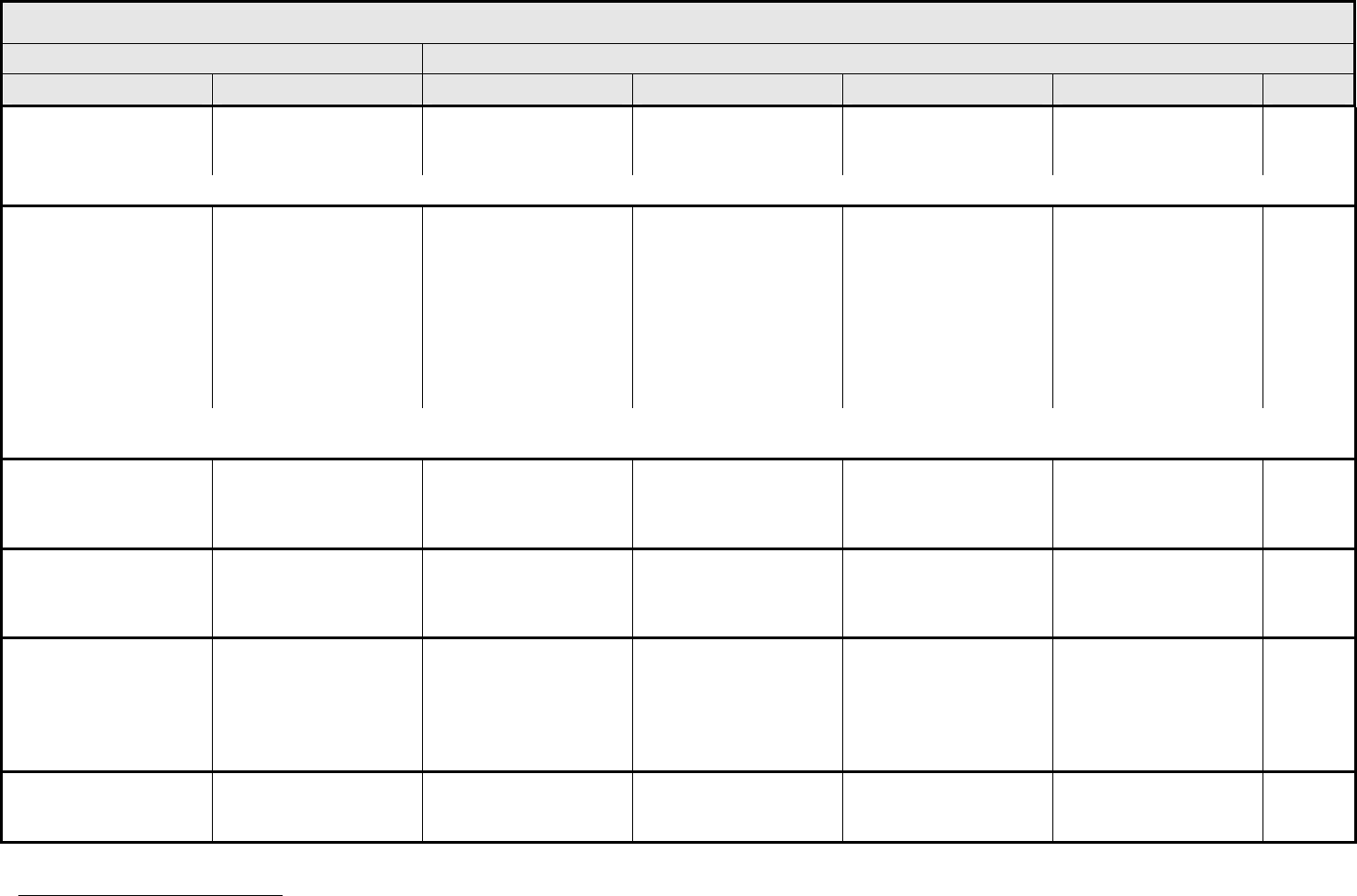
NEUROLOGY Page 3 of 5
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 49 - March 31, 2003, Publish Date: August 9, 2006
Leak, cerebrospinal fluid
(CSF)
CSF leak Transient headache;
postural care indicated
Symptomatic, not
interfering with ADL;
blood patch indicated
Symptomatic, interfering
with ADL; operative
intervention indicated
Life-threatening; disabling Death
REMARK: Leak, cerebrospinal fluid (CSF) may be used for CSF leak associated with operation and persisting >72 hours.
Leukoencephalopathy
(radiographic findings)
Leukoencephalopathy Mild increase in
subarachnoid space
(SAS); mild
ventriculomegaly; small
(+/- multiple) focal T2
hyperintensities, involving
periventricular white
matter or <1/3 of
susceptible areas of
cerebrum
Moderate increase in
SAS; moderate
ventriculomegaly; focal
T2 hyperintensities
extending into centrum
ovale or involving 1/3 to
2/3 of susceptible areas
of cerebrum
Severe increase in SAS;
severe ventriculomegaly;
near total white matter T2
hyperintensities or diffuse
low attenuation (CT)
— —
REMARK: Leukoencephalopathy is a diffuse white matter process, specifically NOT associated with necrosis. Leukoencephalopathy (radiographic findings) does not include lacunas,
which are areas that become void of neural tissue.
Memory impairment Memory impairment Memory impairment not
interfering with function
Memory impairment
interfering with function,
but not interfering with
ADL
Memory impairment
interfering with ADL
Amnesia —
Mental status
7
Mental status — 1 – 3 point below age
and educational norm in
Folstein Mini-Mental
Status Exam (MMSE)
>3 point below age and
educational norm in
Folstein MMSE
— —
Mood alteration
– Select:
– Agitation
– Anxiety
– Depression
– Euphoria
Mood alteration – Select Mild mood alteration not
interfering with function
Moderate mood alteration
interfering with function,
but not interfering with
ADL; medication
indicated
Severe mood alteration
interfering with ADL
Suicidal ideation; danger
to self or others
Death
Myelitis Myelitis Asymptomatic, mild signs
(e.g., Babinski’s or
Lhermitte’s sign)
Weakness or sensory
loss not interfering with
ADL
Weakness or sensory
loss interfering with ADL
Disabling Death
7
Folstein MF, Folstein, SE and McHugh PR (1975) "Mini-Mental State: A Practical Method for Grading the State of Patients for the Clinician," Journal of Psychiatric Research, 12:
189-198

NEUROLOGY Page 4 of 5
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 50 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Neuropathic pain is graded as Pain – Select in the PAIN CATEGORY.
Neuropathy:
cranial
– Select:
Neuropathy: cranial
– Select
Asymptomatic, detected
on exam/testing only
– CN I Smell
– CN II Vision
– CN III Pupil, upper eyelid, extra ocular movements
– CN IV Downward, inward movement of eye
– CN V Motor-jaw muscles; Sensory-facial
– CN VI Lateral deviation of eye
– CN VII Motor-face; Sensory-taste
– CN VIII Hearing and balance
– CN IX Motor-pharynx; Sensory-ear, pharynx, tongue
– CN X Motor-palate; pharynx, larynx
– CN XI Motor-sternomastoid and trapezius
– CN XII Motor-tongue
Symptomatic, not
interfering with ADL
Symptomatic, interfering
with ADL
Life-threatening; disabling Death
Neuropathy:
motor
Neuropathy-motor Asymptomatic, weakness
on exam/testing only
Symptomatic weakness
interfering with function,
but not interfering with
ADL
Weakness interfering with
ADL; bracing or
assistance to walk (e.g.,
cane or walker) indicated
Life-threatening; disabling
(e.g., paralysis)
Death
REMARK: Cranial nerve motor neuropathy is graded as Neuropathy: cranial – Select.
ALSO CONSIDER: Laryngeal nerve dysfunction; Phrenic nerve dysfunction.
Neuropathy:
sensory
Neuropathy-sensory Asymptomatic; loss of
deep tendon reflexes or
paresthesia (including
tingling) but not
interfering with function
Sensory alteration or
paresthesia (including
tingling), interfering with
function, but not
interfering with ADL
Sensory alteration or
paresthesia interfering
with ADL
Disabling Death
REMARK: Cranial nerve sensory neuropathy is graded as Neuropathy: cranial – Select.
Personality/behavioral Personality Change, but not
adversely affecting
patient or family
Change, adversely
affecting patient or family
Mental health intervention
indicated
Change harmful to others
or self; hospitalization
indicated
Death
Phrenic nerve dysfunction Phrenic nerve Asymptomatic weakness
on exam/testing only
Symptomatic but not
interfering with ADL;
intervention not indicated
Significant dysfunction;
intervention indicated
(e.g., diaphragmatic
plication)
Life-threatening
respiratory compromise;
mechanical ventilation
indicated
Death
Psychosis (hallucinations/
delusions)
Psychosis — Transient episode Interfering with ADL;
medication, supervision
Harmful to others or self;
life-threatening
Death
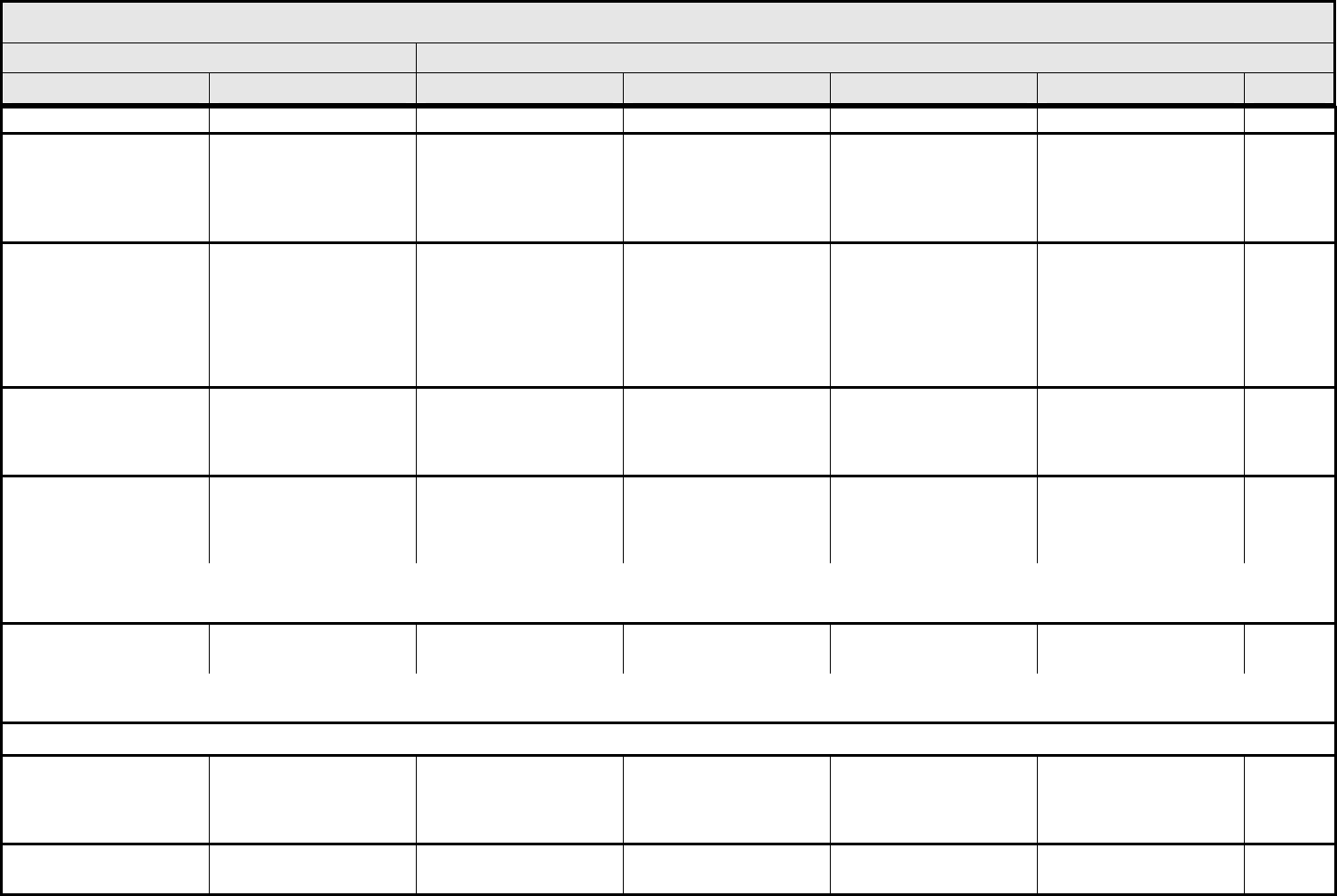
NEUROLOGY Page 5 of 5
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 51 - March 31, 2003, Publish Date: August 9, 2006
or restraints indicated consequences
Pyramidal tract
dysfunction (e.g., ↑ tone,
hyperreflexia, positive
Babinski, ↓ fine motor
coordination)
Pyramidal tract
dysfunction
Asymptomatic,
abnormality on exam or
testing only
Symptomatic; interfering
with function but not
interfering with ADL
Interfering with ADL Disabling; paralysis Death
Seizure Seizure — One brief generalized
seizure; seizure(s) well
controlled by
anticonvulsants or
infrequent focal motor
seizures not interfering
with ADL
Seizures in which
consciousness is altered;
poorly controlled seizure
disorder, with
breakthrough generalized
seizures despite medical
intervention
Seizures of any kind
which are prolonged,
repetitive, or difficult to
control (e.g., status
epilepticus, intractable
epilepsy)
Death
Somnolence/depressed
level of consciousness
Somnolence — Somnolence or sedation
interfering with function,
but not interfering with
ADL
Obtundation or stupor;
difficult to arouse;
interfering with ADL
Coma Death
Speech impairment
(e.g., dysphasia or
aphasia)
Speech impairment — Awareness of receptive
or expressive dysphasia,
not impairing ability to
communicate
Receptive or expressive
dysphasia, impairing
ability to communicate
Inability to communicate —
REMARK: Speech impairment refers to a primary CNS process, not neuropathy or end organ dysfunction.
ALSO CONSIDER: Laryngeal nerve dysfunction; Voice changes/dysarthria (e.g., hoarseness, loss, or alteration in voice, laryngitis).
Syncope (fainting) Syncope (fainting) — — Present Life-threatening
consequences
Death
ALSO CONSIDER: CNS cerebrovascular ischemia; Conduction abnormality/atrioventricular heart block – Select; Dizziness; Supraventricular and nodal arrhythmia – Select; Vasovagal
episode; Ventricular arrhythmia – Select.
NAVIGATION NOTE: Taste alteration (CN VII, IX) is graded as Taste alteration (dysgeusia) in the GASTROINTESTINAL CATEGORY.
Tremor Tremor Mild and brief or
intermittent but not
interfering with function
Moderate tremor
interfering with function,
but not interfering with
ADL
Severe tremor interfering
with ADL
Disabling —
Neurology – Other
(Specify, __)
Neurology – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
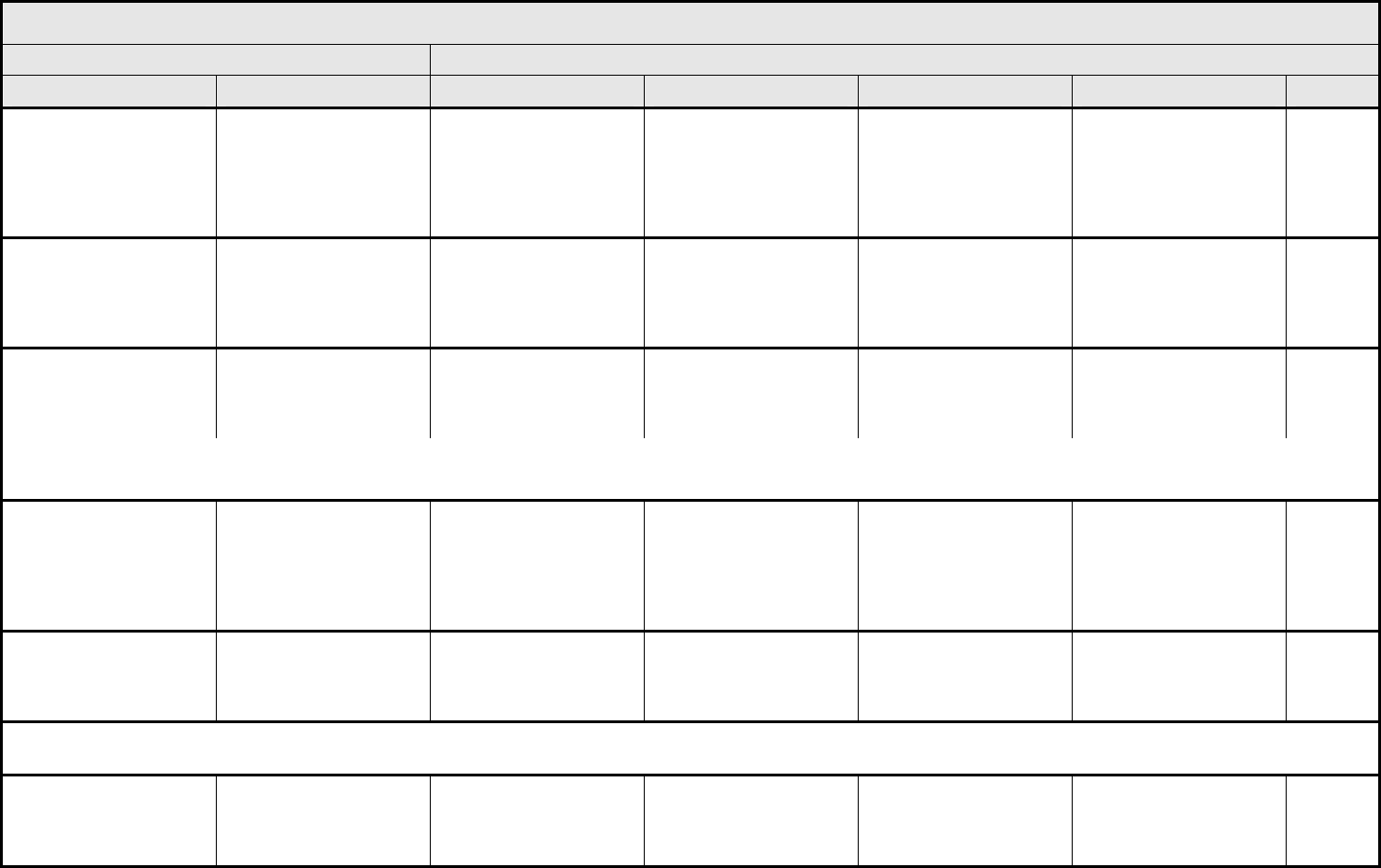
OCULAR/VISUAL Page 1 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 52 - March 31, 2003, Publish Date: August 9, 2006
Cataract Cataract Asymptomatic, detected
on exam only
Symptomatic, with
moderate decrease in
visual acuity
(20/40 or better);
decreased visual function
correctable with glasses
Symptomatic with marked
decrease in visual acuity
(worse than 20/40);
operative intervention
indicated (e.g., cataract
surgery)
— —
Dry eye syndrome Dry eye Mild, intervention not
indicated
Symptomatic, interfering
with function but not
interfering with ADL;
medical intervention
indicated
Symptomatic or decrease
in visual acuity interfering
with ADL; operative
intervention indicated
— —
Eyelid dysfunction Eyelid dysfunction Asymptomatic Symptomatic, interfering
with function but not ADL;
requiring topical agents or
epilation
Symptomatic; interfering
with ADL; surgical
intervention indicated
— —
REMARK: Eyelid dysfunction includes canalicular stenosis, ectropion, entropion, erythema, madarosis, symblepharon, telangiectasis, thickening, and trichiasis.
A
LSO CONSIDER: Neuropathy: cranial – Select.
Glaucoma Glaucoma Elevated intraocular
pressure (EIOP) with
single topical agent for
intervention; no visual
field deficit
EIOP causing early visual
field deficit (i.e., nasal
step or arcuate deficit);
multiple topical or oral
agents indicated
EIOP causing marked
visual field deficits (i.e.,
involving both superior
and inferior visual fields);
operative intervention
indicated
EIOP resulting in
blindness (20/200 or
worse); enucleation
indicated
—
Keratitis (corneal
inflammation/corneal
ulceration)
Keratitis Abnormal ophthalmologic
changes only;
intervention not indicated
Symptomatic and
interfering with function,
but not interfering with
ADL
Symptomatic and
interfering with ADL;
operative intervention
indicated
Perforation or blindness
(20/200 or worse)
—
NAVIGATION NOTE: Ocular muscle weakness is graded as Muscle weakness, generalized or specific area (not due to neuropathy) – Select in the MUSCULOSKELETAL/SOFT TISSUE
CATEGORY.
Night blindness
(nyctalopia)
Nyctalopia Symptomatic, not
interfering with function
Symptomatic and
interfering with function
but not interfering with
ADL
Symptomatic and
interfering with ADL
Disabling —
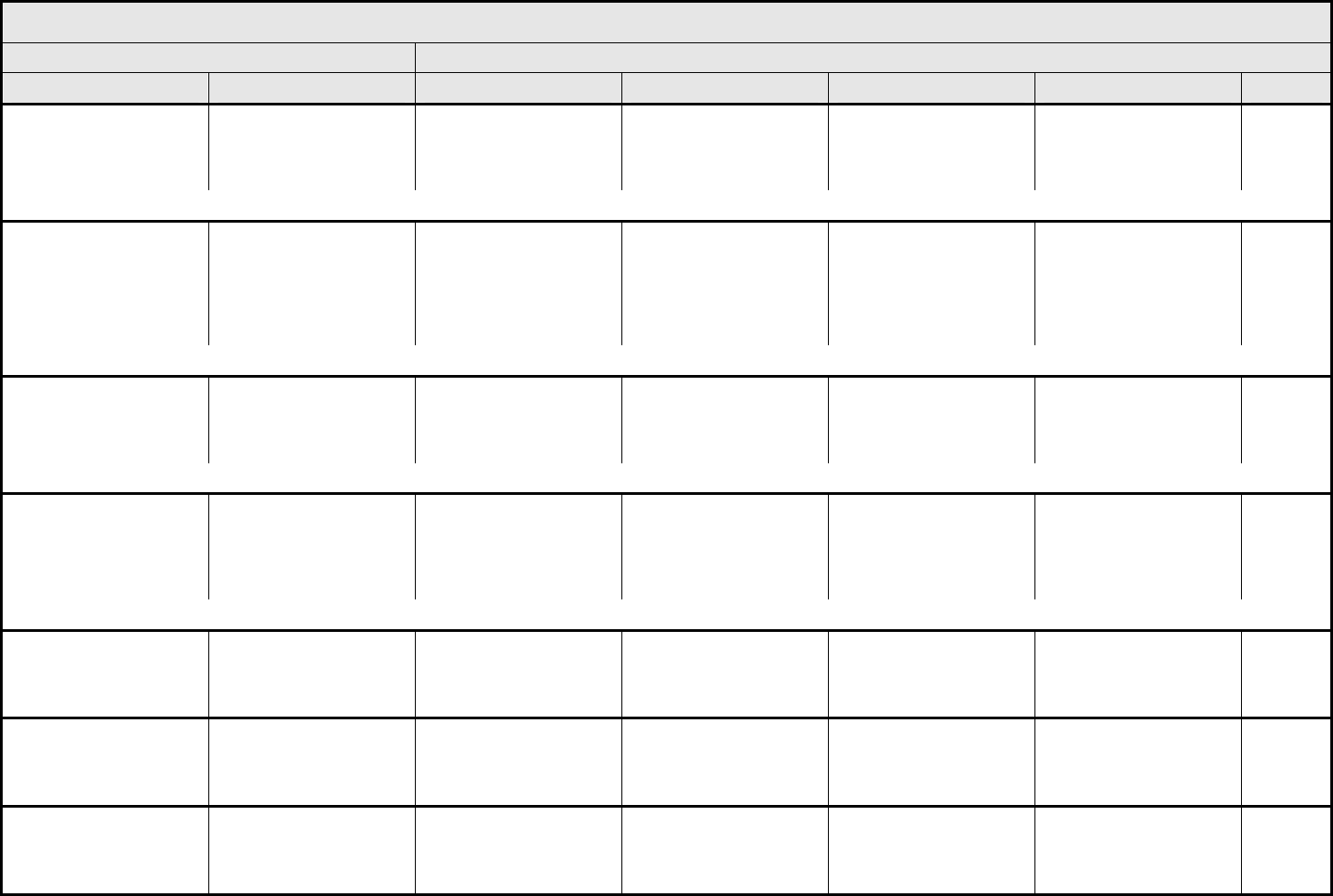
OCULAR/VISUAL Page 2 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 53 - March 31, 2003, Publish Date: August 9, 2006
Nystagmus Nystagmus Asymptomatic Symptomatic and
interfering with function
but not interfering with
ADL
Symptomatic and
interfering with ADL
Disabling —
ALSO CONSIDER: Neuropathy: cranial – Select; Ophthalmoplegia/diplopia (double vision).
Ocular surface disease Ocular surface disease Asymptomatic or
minimally symptomatic
but not interfering with
function
Symptomatic, interfering
with function but not
interfering with ADL;
topical antibiotics or other
topical intervention
indicated
Symptomatic, interfering
with ADL; operative
intervention indicated
— —
REMARK: Ocular surface disease includes conjunctivitis, keratoconjunctivitis sicca, chemosis, keratinization, and palpebral conjunctival epithelial metaplasia.
Ophthalmoplegia/
diplopia (double vision)
Diplopia Intermittently
symptomatic, intervention
not indicated
Symptomatic and
interfering with function
but not interfering with
ADL
Symptomatic and
interfering with ADL;
surgical intervention
indicated
Disabling —
ALSO CONSIDER: Neuropathy: cranial – Select.
Optic disc edema Optic disc edema Asymptomatic Decreased visual acuity
(20/40 or better); visual
field defect present
Decreased visual acuity
(worse than 20/40);
marked visual field defect
but sparing the central 20
degrees
Blindness
(20/200 or worse)
—
ALSO CONSIDER: Neuropathy: cranial – Select.
Proptosis/enophthalmos Proptosis/enophthalmos Asymptomatic,
intervention not indicated
Symptomatic and
interfering with function,
but not interfering with
ADL
Symptomatic and
interfering with ADL
— —
Retinal detachment Retinal detachment Exudative; no central
vision loss; intervention
not indicated
Exudative and visual
acuity 20/40 or better but
intervention not indicated
Rhegmatogenous or
exudative detachment;
operative intervention
indicated
Blindness
(20/200 or worse)
—
Retinopathy Retinopathy Asymptomatic Symptomatic with
moderate decrease in
visual acuity
(20/40 or better)
Symptomatic with marked
decrease in visual acuity
(worse than 20/40)
Blindness
(20/200 or worse)
—
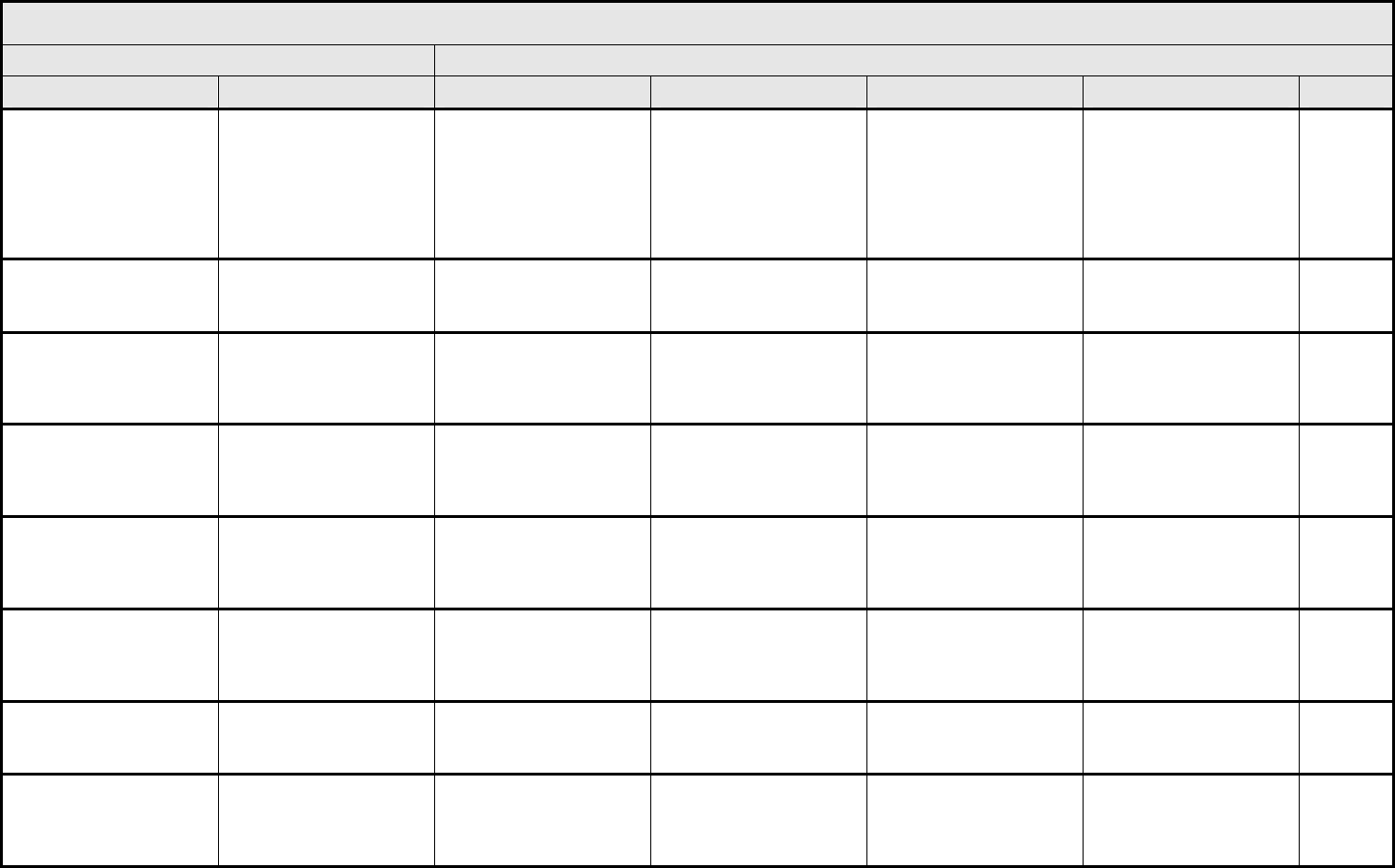
OCULAR/VISUAL Page 3 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 54 - March 31, 2003, Publish Date: August 9, 2006
Scleral necrosis/melt Scleral necrosis Asymptomatic or
symptomatic but not
interfering with function
Symptomatic, interfering
with function but not
interfering with ADL;
moderate decrease in
visual acuity (20/40 or
better); medical
intervention indicated
Symptomatic, interfering
with ADL; marked
decrease in visual acuity
(worse than 20/40);
operative intervention
indicated
Blindness (20/200 or
worse); painful eye with
enucleation indicated
—
Uveitis Uveitis Asymptomatic Anterior uveitis; medical
intervention indicated
Posterior or pan-uveitis;
operative intervention
indicated
Blindness
(20/200 or worse)
—
Vision-blurred vision Blurred vision Symptomatic not
interfering with function
Symptomatic and
interfering with function,
but not interfering with
ADL
Symptomatic and
interfering with ADL
Disabling —
Vision-flashing
lights/floaters
Flashing lights Symptomatic not
interfering with function
Symptomatic and
interfering with function,
but not interfering with
ADL
Symptomatic and
interfering with ADL
Disabling —
Vision-photophobia Photophobia Symptomatic not
interfering with function
Symptomatic and
interfering with function,
but not interfering with
ADL
Symptomatic and
interfering with ADL
Disabling —
Vitreous hemorrhage Vitreous hemorrhage Asymptomatic, clinical
findings only
Symptomatic, interfering
with function, but not
interfering with ADL;
intervention not indicated
Symptomatic, interfering
with ADL; vitrectomy
indicated
— —
Watery eye (epiphora,
tearing)
Watery eye Symptomatic, intervention
not indicated
Symptomatic, interfering
with function but not
interfering with ADL
Symptomatic, interfering
with ADL
— —
Ocular/Visual – Other
(Specify, __)
Ocular – Other
(Specify)
Symptomatic not
interfering with function
Symptomatic and
interfering with function,
but not interfering with
ADL
Symptomatic and
interfering with ADL
Blindness
(20/200 or worse)
Death

PAIN Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 55 - March 31, 2003, Publish Date: August 9, 2006
Pain
– Select:
‘Select’ AEs appear at the
end of the CATEGORY.
Pain – Select Mild pain not interfering
with function
Moderate pain; pain or
analgesics interfering with
function, but not
interfering with ADL
Severe pain; pain or
analgesics severely
interfering with ADL
Disabling —
Pain – Other
(Specify, __)
Pain – Other (Specify) Mild pain not interfering
with function
Moderate pain; pain or
analgesics interfering with
function, but not
interfering with ADL
Severe pain; pain or
analgesics severely
interfering with ADL
Disabling —
PAIN – SELECT
AUDITORY/EAR
– External ear
– Middle ear
CARDIOVASCULAR
– Cardiac/heart
– Pericardium
DERMATOLOGY/SKIN
– Face
– Lip
– Oral-gums
– Scalp
– Skin
GASTROINTESTINAL
– Abdomen NOS
– Anus
– Dental/teeth/peridontal
– Esophagus
– Oral cavity
– Peritoneum
– Rectum
– Stomach
GENERAL
– Pain NOS
– Tumor pain
HEPATOBILIARY/PANCREAS
– Gallbladder
– Liver
LYMPHATIC
– Lymph node
MUSCULOSKELETAL
– Back
– Bone
– Buttock
– Extremity-limb
– Intestine
– Joint
– Muscle
– Neck
– Phantom (pain associated with missing limb)
NEUROLOGY
– Head/headache
– Neuralgia/peripheral nerve
OCULAR
– Eye
PULMONARY/UPPER RESPIRATORY
– Chest wall
– Chest/thorax NOS
PULMONARY/UPPER RESPIRATORY
(continued)
– Larynx
– Pleura
– Sinus
– Throat/pharynx/larynx
RENAL/GENITOURINARY
– Bladder
– Kidney
SEXUAL/REPRODUCTIVE FUNCTION
– Breast
– Ovulatory
– Pelvis
– Penis
– Perineum
– Prostate
– Scrotum
– Testicle
– Urethra
– Uterus
– Vagina
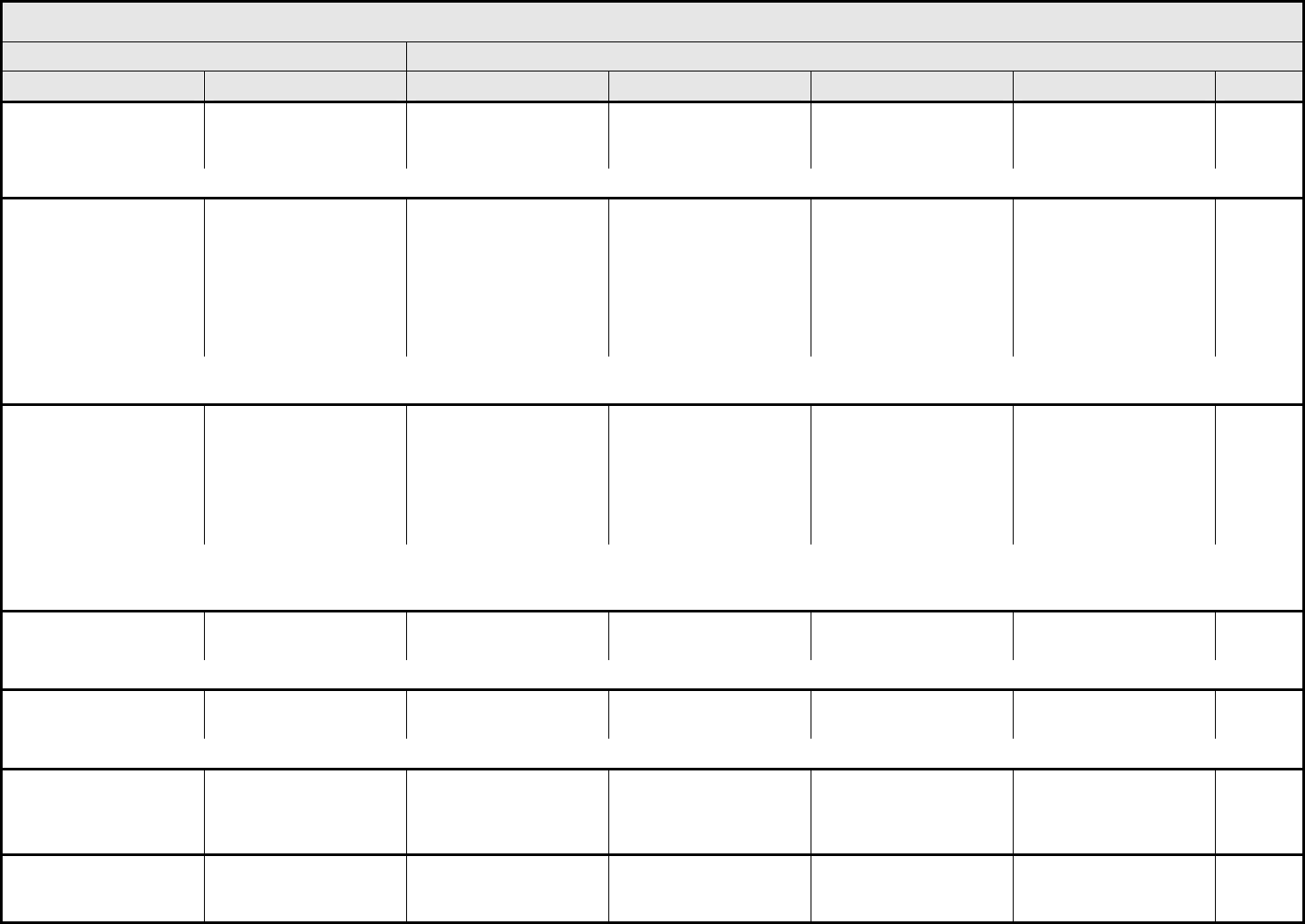
PULMONARY/UPPER RESPIRATORY Page 1 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 56 - March 31, 2003, Publish Date: August 9, 2006
Adult Respiratory
Distress Syndrome
(ARDS)
ARDS — — Present, intubation not
indicated
Present, intubation
indicated
Death
ALSO CONSIDER: Dyspnea (shortness of breath); Hypoxia; Pneumonitis/pulmonary infiltrates.
Aspiration Aspiration Asymptomatic (“silent
aspiration”); endoscopy
or radiographic (e.g.,
barium swallow) findings
Symptomatic (e.g.,
altered eating habits,
coughing or choking
episodes consistent with
aspiration); medical
intervention indicated
(e.g., antibiotics, suction
or oxygen)
Clinical or radiographic
signs of pneumonia or
pneumonitis; unable to
aliment orally
Life-threatening (e.g.,
aspiration pneumonia or
pneumonitis)
Death
ALSO CONSIDER: Infection (documented clinically or microbiologically) with Grade 3 or 4 neutrophils (ANC <1.0 x 10
9
/L) – Select; Infection with normal ANC or Grade 1 or 2 neutrophils
– Select; Infection with unknown ANC – Select; Laryngeal nerve dysfunction; Neuropathy: cranial – Select; Pneumonitis/pulmonary infiltrates.
Atelectasis Atelectasis Asymptomatic Symptomatic (e.g.,
dyspnea, cough), medical
intervention indicated
(e.g., bronchoscopic
suctioning, chest
physiotherapy,
suctioning)
Operative (e.g., stent,
laser) intervention
indicated
Life-threatening
respiratory compromise
Death
ALSO CONSIDER: Adult Respiratory Distress Syndrome (ARDS); Cough; Dyspnea (shortness of breath); Hypoxia; Infection (documented clinically or microbiologically) with Grade 3 or 4
neutrophils (ANC <1.0 x 109/L) – Select; Infection with normal ANC or Grade 1 or 2 neutrophils – Select; Infection with unknown ANC – Select; Obstruction/stenosis of airway –
Select; Pneumonitis/pulmonary infiltrates; Pulmonary fibrosis (radiographic changes).
Bronchospasm,
wheezing
Bronchospasm Asymptomatic Symptomatic not
interfering with function
Symptomatic interfering
with function
Life-threatening Death
ALSO CONSIDER: Allergic reaction/hypersensitivity (including drug fever); Dyspnea (shortness of breath).
Carbon monoxide
diffusion capacity (DL
CO
)
DL
CO
90 – 75% of predicted
value
<75 – 50% of predicted
value
<50 – 25% of predicted
value
<25% of predicted value Death
ALSO CONSIDER: Hypoxia; Pneumonitis/pulmonary infiltrates; Pulmonary fibrosis (radiographic changes).
Chylothorax Chylothorax Asymptomatic Symptomatic;
thoracentesis or tube
drainage indicated
Operative intervention
indicated
Life-threatening (e.g.,
hemodynamic instability
or ventilatory support
indicated)
Death
Cough Cough Symptomatic, non-
narcotic medication only
indicated
Symptomatic and
narcotic medication
indicated
Symptomatic and
significantly interfering
with sleep or ADL
— —
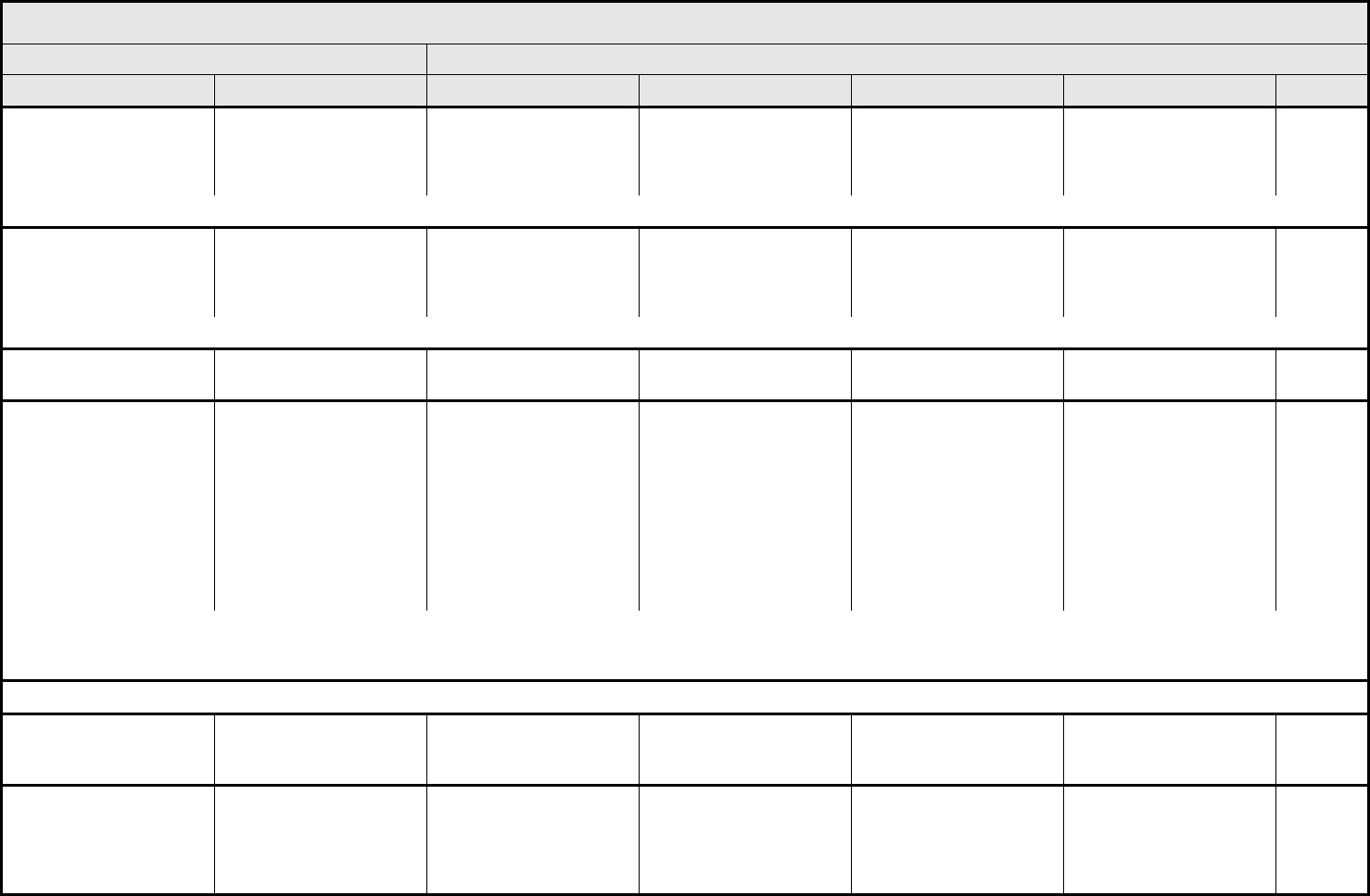
PULMONARY/UPPER RESPIRATORY Page 2 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 57 - March 31, 2003, Publish Date: August 9, 2006
Dyspnea (shortness of
breath)
Dyspnea Dyspnea on exertion, but
can walk 1 flight of stairs
without stopping
Dyspnea on exertion but
unable to walk 1 flight of
stairs or 1 city block
(0.1km) without stopping
Dyspnea with ADL Dyspnea at rest;
intubation/ventilator
indicated
Death
ALSO CONSIDER: Hypoxia; Neuropathy: motor; Pneumonitis/pulmonary infiltrates; Pulmonary fibrosis (radiographic changes).
Edema, larynx Edema, larynx Asymptomatic edema by
exam only
Symptomatic edema, no
respiratory distress
Stridor; respiratory
distress; interfering with
ADL
Life-threatening airway
compromise;
tracheotomy, intubation,
or laryngectomy indicated
Death
ALSO CONSIDER: Allergic reaction/hypersensitivity (including drug fever).
FEV
1
FEV
1
90 – 75% of predicted
value
<75 – 50% of predicted
value
<50 – 25% of predicted
value
<25% of predicted Death
Fistula, pulmonary/upper
respiratory
– Select:
– Bronchus
– Larynx
– Lung
– Oral cavity
– Pharynx
– Pleura
– Trachea
Fistula, pulmonary
– Select
Asymptomatic,
radiographic findings only
Symptomatic, tube
thoracostomy or medical
management indicated;
associated with altered
respiratory function but
not interfering with ADL
Symptomatic and
associated with altered
respiratory function
interfering with ADL; or
endoscopic (e.g., stent)
or primary closure by
operative intervention
indicated
Life-threatening
consequences; operative
intervention with
thoracoplasty, chronic
open drainage or multiple
thoracotomies indicated
Death
REMARK: A fistula is defined as an abnormal communication between two body cavities, potential spaces, and/or the skin. The site indicated for a fistula should be the site from which
the abnormal process is believed to have arisen. For example, a tracheo-esophageal fistula arising in the context of a resected or irradiated esophageal cancer should be graded as
Fistula, GI – esophagus in the GASTROINTESTINAL CATEGORY.
NAVIGATION NOTE: Hemoptysis is graded as Hemorrhage, pulmonary/upper respiratory – Select in the HEMORRHAGE/BLEEDING CATEGORY.
Hiccoughs (hiccups,
singultus)
Hiccoughs Symptomatic,
intervention not indicated
Symptomatic,
intervention indicated
Symptomatic,
significantly interfering
with sleep or ADL
— —
Hypoxia Hypoxia — Decreased O
2
saturation
with exercise (e.g., pulse
oximeter <88%);
intermittent supplemental
oxygen
Decreased O
2
saturation
at rest; continuous
oxygen indicated
Life-threatening;
intubation or ventilation
indicated
Death

PULMONARY/UPPER RESPIRATORY Page 3 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 58 - March 31, 2003, Publish Date: August 9, 2006
Nasal cavity/paranasal
sinus reactions
Nasal/paranasal
reactions
Asymptomatic mucosal
crusting, blood-tinged
secretions
Symptomatic stenosis or
edema/narrowing
interfering with airflow
Stenosis with significant
nasal obstruction;
interfering with ADL
Necrosis of soft tissue or
bone
Death
ALSO CONSIDER: Infection (documented clinically or microbiologically) with Grade 3 or 4 neutrophils (ANC <1.0 x 10
9
/L) – Select; Infection with normal ANC or Grade 1 or 2 neutrophils
– Select; Infection with unknown ANC – Select.
Obstruction/stenosis of
airway
– Select:
– Bronchus
– Larynx
– Pharynx
– Trachea
Airway obstruction
– Select
Asymptomatic obstruction
or stenosis on exam,
endoscopy, or radiograph
Symptomatic (e.g., noisy
airway breathing), but
causing no respiratory
distress; medical
management indicated
(e.g., steroids)
Interfering with ADL;
stridor or endoscopic
intervention indicated
(e.g., stent, laser)
Life-threatening airway
compromise; tracheotomy
or intubation indicated
Death
Pleural effusion
(non-malignant)
Pleural effusion Asymptomatic Symptomatic,
intervention such as
diuretics or up to 2
therapeutic
thoracenteses indicated
Symptomatic and
supplemental oxygen,
>2 therapeutic
thoracenteses, tube
drainage, or pleurodesis
indicated
Life-threatening (e.g.,
causing hemodynamic
instability or ventilatory
support indicated)
Death
ALSO CONSIDER: Atelectasis; Cough; Dyspnea (shortness of breath); Hypoxia; Pneumonitis/pulmonary infiltrates; Pulmonary fibrosis (radiographic changes).
NAVIGATION NOTE: Pleuritic pain is graded as Pain – Select in the PAIN CATEGORY.
Pneumonitis/pulmonary
infiltrates
Pneumonitis Asymptomatic,
radiographic findings only
Symptomatic, not
interfering with ADL
Symptomatic, interfering
with ADL; O
2
indicated
Life-threatening;
ventilatory support
indicated
Death
ALSO CONSIDER: Adult Respiratory Distress Syndrome (ARDS); Cough; Dyspnea (shortness of breath); Hypoxia; Infection (documented clinically or microbiologically) with Grade 3 or 4
neutrophils (ANC <1.0 x 10
9
/L) – Select; Infection with normal ANC or Grade 1 or 2 neutrophils – Select; Infection with unknown ANC – Select; Pneumonitis/pulmonary infiltrates;
Pulmonary fibrosis (radiographic changes).
Pneumothorax Pneumothorax Asymptomatic,
radiographic findings only
Symptomatic;
intervention indicated
(e.g., hospitalization for
observation, tube
placement without
sclerosis)
Sclerosis and/or
operative intervention
indicated
Life-threatening, causing
hemodynamic instability
(e.g., tension
pneumothorax);
ventilatory support
indicated
Death
Prolonged chest tube
drainage or air leak after
pulmonary resection
Chest tube drainage or
leak
— Sclerosis or additional
tube thoracostomy
indicated
Operative intervention
indicated (e.g.,
thoracotomy with stapling
or sealant application)
Life-threatening;
debilitating; organ
resection indicated
Death
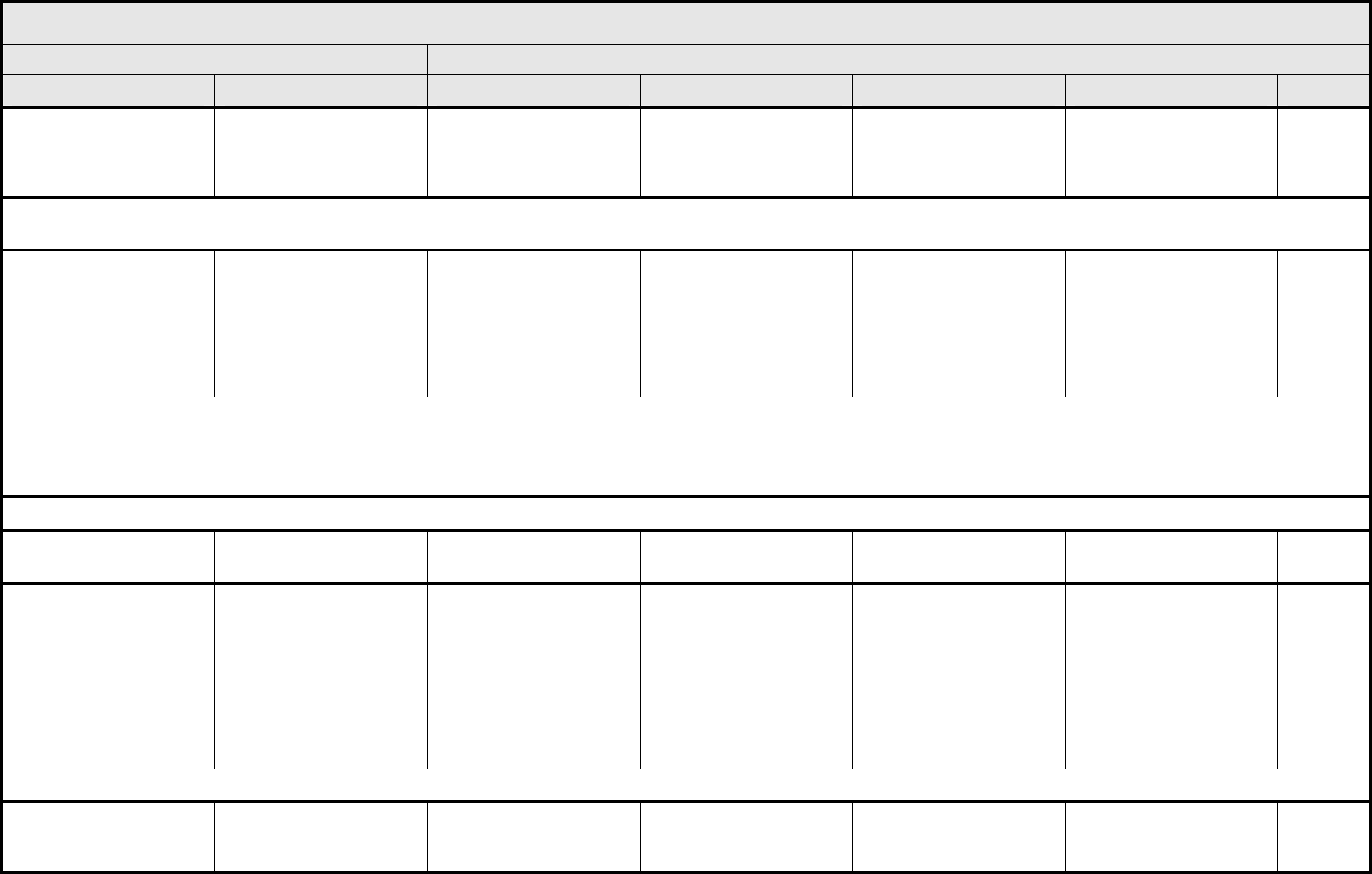
PULMONARY/UPPER RESPIRATORY Page 4 of 4
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 59 - March 31, 2003, Publish Date: August 9, 2006
Prolonged intubation
after pulmonary resection
(>24 hrs after surgery)
Prolonged intubation — Extubated within
24 – 72 hrs
postoperatively
Extubated >72 hrs
postoperatively, but
before tracheostomy
indicated
Tracheostomy indicated Death
NAVIGATION NOTE: Pulmonary embolism is graded as Grade 4 either as Thrombosis/embolism (vascular access-related) or Thrombosis/thrombus/embolism in the VASCULAR
CATEGORY.
Pulmonary fibrosis
(radiographic changes)
Pulmonary fibrosis Minimal radiographic
findings (or patchy or bi-
basilar changes) with
estimated radiographic
proportion of total lung
volume that is fibrotic of
<25%
Patchy or bi-basilar
changes with estimated
radiographic proportion of
total lung volume that is
fibrotic of 25 – <50%
Dense or widespread
infiltrates/consolidation
with estimated
radiographic proportion of
total lung volume that is
fibrotic of 50 – <75%
Estimated radiographic
proportion of total lung
volume that is fibrotic is
≥75%; honeycombing
Death
REMARK: Fibrosis is usually a “late effect” seen >3 months after radiation or combined modality therapy (including surgery). It is thought to represent scar/fibrotic lung tissue. It may be
difficult to distinguish from pneumonitis that is generally seen within 3 months of radiation or combined modality therapy.
A
LSO CONSIDER: Adult Respiratory Distress Syndrome (ARDS); Cough; Dyspnea (shortness of breath); Hypoxia; Infection (documented clinically or microbiologically) with Grade 3 or 4
neutrophils (ANC <1.0 x 10
9
/L) – Select; Infection with normal ANC or Grade 1 or 2 neutrophils – Select; Infection with unknown ANC – Select.
NAVIGATION NOTE: Recurrent laryngeal nerve dysfunction is graded as Laryngeal nerve dysfunction in the NEUROLOGY CATEGORY.
Vital capacity Vital capacity 90 – 75% of predicted
value
<75 – 50% of predicted
value
<50 – 25% of predicted
value
<25% of predicted value Death
Voice changes/dysarthria
(e.g., hoarseness, loss or
alteration in voice,
laryngitis)
Voice changes Mild or intermittent
hoarseness or voice
change, but fully
understandable
Moderate or persistent
voice changes, may
require occasional
repetition but
understandable on
telephone
Severe voice changes
including predominantly
whispered speech; may
require frequent repetition
or face-to-face contact for
understandability;
requires voice aid (e.g.,
electrolarynx) for ≤50% of
communication
Disabling;
non-understandable voice
or aphonic; requires voice
aid (e.g., electrolarynx)
for >50% of
communication or
requires >50% written
communication
Death
ALSO CONSIDER: Laryngeal nerve dysfunction; Speech impairment (e.g., dysphasia or aphasia).
Pulmonary/Upper
Respiratory – Other
(Specify, __)
Pulmonary – Other
(Specify)
Mild Moderate Severe Life-threatening;
disabling
Death
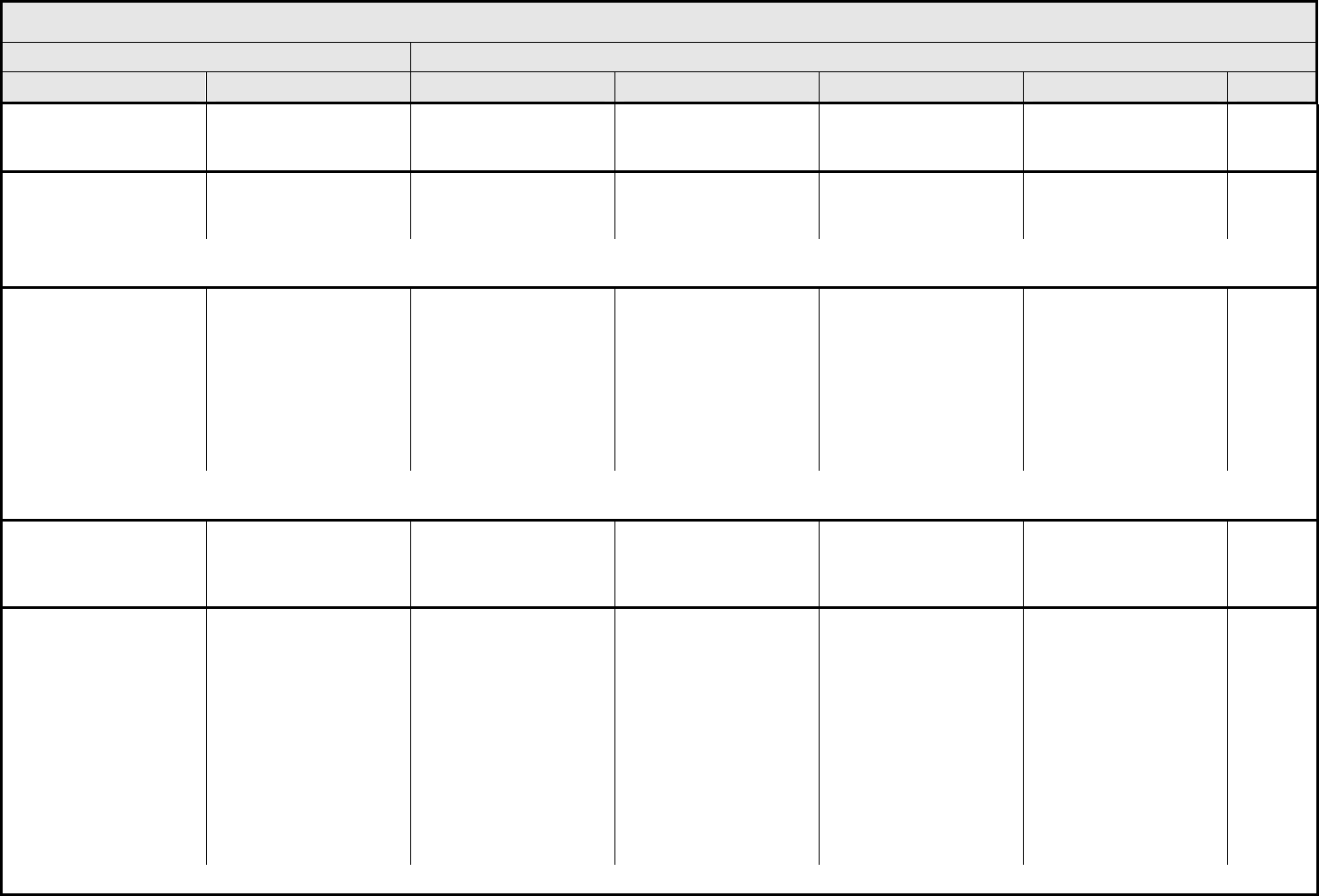
RENAL/GENITOURINARY Page 1 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 60 - March 31, 2003, Publish Date: August 9, 2006
Bladder spasms Bladder spasms Symptomatic, intervention
not indicated
Symptomatic,
antispasmodics indicated
Narcotics indicated Major surgical
intervention indicated
(e.g., cystectomy)
—
Cystitis Cystitis Asymptomatic Frequency with dysuria;
macroscopic hematuria
Transfusion; IV pain
medications; bladder
irrigation indicated
Catastrophic bleeding;
major non-elective
intervention indicated
Death
ALSO CONSIDER: Infection (documented clinically or microbiologically) with Grade 3 or 4 neutrophils (ANC <1.0 x 109/L) – Select; Infection with normal ANC or Grade 1 or 2 neutrophils
– Select; Infection with unknown ANC – Select; Pain – Select.
Fistula, GU
– Select:
– Bladder
– Genital tract-female
– Kidney
– Ureter
– Urethra
– Uterus
– Vagina
Fistula, GU – Select Asymptomatic,
radiographic findings only
Symptomatic;
noninvasive intervention
indicated
Symptomatic interfering
with ADL; invasive
intervention indicated
Life-threatening
consequences; operative
intervention requiring
partial or full organ
resection; permanent
urinary diversion
Death
REMARK: A fistula is defined as an abnormal communication between two body cavities, potential spaces, and/or the skin. The site indicated for a fistula should be the site from which
the abnormal process is believed to have originated.
Incontinence, urinary Incontinence, urinary Occasional (e.g., with
coughing, sneezing, etc.),
pads not indicated
Spontaneous, pads
indicated
Interfering with ADL;
intervention indicated
(e.g., clamp, collagen
injections)
Operative intervention
indicated (e.g.,
cystectomy or permanent
urinary diversion)
—
Leak (including
anastomotic), GU
– Select:
– Bladder
– Fallopian tube
– Kidney
– Spermatic cord
– Stoma
– Ureter
– Urethra
– Uterus
– Vagina
– Vas deferens
Leak, GU – Select Asymptomatic,
radiographic findings only
Symptomatic; medical
intervention indicated
Symptomatic, interfering
with GU function; invasive
or endoscopic
intervention indicated
Life-threatening Death
REMARK: Leak (including anastomotic), GU – Select refers to clinical signs and symptoms or radiographic confirmation of anastomotic leak but without development of fistula.

RENAL/GENITOURINARY Page 2 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 61 - March 31, 2003, Publish Date: August 9, 2006
Obstruction, GU
– Select:
– Bladder
– Fallopian tube
– Prostate
– Spermatic cord
– Stoma
– Testes
– Ureter
– Urethra
– Uterus
– Vagina
– Vas deferens
Obstruction, GU – Select Asymptomatic,
radiographic or
endoscopic findings only
Symptomatic but no
hydronephrosis, sepsis or
renal dysfunction; dilation
or endoscopic repair or
stent placement indicated
Symptomatic and altered
organ function (e.g.,
sepsis or hydronephrosis,
or renal dysfunction);
operative intervention
indicated
Life-threatening
consequences; organ
failure or operative
intervention requiring
complete organ resection
indicated
Death
NAVIGATION NOTE: Operative injury is graded as Intra-operative injury – Select Organ or Structure in the SURGERY/INTRA-OPERATIVE INJURY CATEGORY.
Perforation, GU
– Select:
– Bladder
– Fallopian tube
– Kidney
– Ovary
– Prostate
– Spermatic cord
– Stoma
– Testes
– Ureter
– Urethra
– Uterus
– Vagina
– Vas deferens
Perforation, GU – Select Asymptomatic
radiographic findings only
Symptomatic, associated
with altered renal/GU
function
Symptomatic, operative
intervention indicated
Life-threatening
consequences or organ
failure; operative
intervention requiring
organ resection indicated
Death
Prolapse of stoma, GU Prolapse stoma, GU Asymptomatic; special
intervention,
extraordinary care not
indicated
Extraordinary local care
or maintenance; minor
revision under local
anesthesia indicated
Dysfunctional stoma;
operative intervention or
major stomal revision
indicated
Life-threatening
consequences
Death
REMARK: Other stoma complications may be graded as Fistula, GU – Select; Leak (including anastomotic), GU – Select; Obstruction, GU – Select; Perforation, GU – Select;
Stricture/stenosis (including anastomotic), GU – Select.
Renal failure Renal failure — — Chronic dialysis not
indicated
Chronic dialysis or renal
transplant indicated
Death
ALSO CONSIDER: Glomerular filtration rate.

RENAL/GENITOURINARY Page 3 of 3
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 62 - March 31, 2003, Publish Date: August 9, 2006
Stricture/stenosis
(including anastomotic),
GU
– Select:
– Bladder
– Fallopian tube
– Prostate
– Spermatic cord
– Stoma
– Testes
– Ureter
– Urethra
– Uterus
– Vagina
– Vas deferens
Stricture, anastomotic,
GU – Select
Asymptomatic,
radiographic or
endoscopic findings only
Symptomatic but no
hydronephrosis, sepsis or
renal dysfunction; dilation
or endoscopic repair or
stent placement indicated
Symptomatic and altered
organ function (e.g.,
sepsis or hydronephrosis,
or renal dysfunction);
operative intervention
indicated
Life-threatening
consequences; organ
failure or operative
intervention requiring
organ resection indicated
Death
ALSO CONSIDER: Obstruction, GU – Select.
Urinary electrolyte
wasting (e.g., Fanconi’s
syndrome, renal tubular
acidosis)
Urinary electrolyte
wasting
Asymptomatic,
intervention not indicated
Mild, reversible and
manageable with
replacement
Irreversible, requiring
continued replacement
— —
ALSO CONSIDER: Acidosis (metabolic or respiratory); Bicarbonate, serum-low; Calcium, serum-low (hypocalcemia); Phosphate, serum-low (hypophosphatemia).
Urinary
frequency/urgency
Urinary frequency Increase in frequency or
nocturia up to 2 x normal;
enuresis
Increase >2 x normal but
<hourly
≥1 x/hr; urgency; catheter
indicated
— —
Urinary retention
(including neurogenic
bladder)
Urinary retention Hesitancy or dribbling, no
significant residual urine;
retention occurring during
the immediate
postoperative period
Hesitancy requiring
medication; or operative
bladder atony requiring
indwelling catheter
beyond immediate
postoperative period but
for <6 weeks
More than daily
catheterization indicated;
urological intervention
indicated (e.g., TURP,
suprapubic tube,
urethrotomy)
Life-threatening
consequences; organ
failure (e.g., bladder
rupture); operative
intervention requiring
organ resection indicated
Death
REMARK: The etiology of retention (if known) is graded as Obstruction, GU – Select; Stricture/stenosis (including anastomotic), GU – Select.
A
LSO CONSIDER: Obstruction, GU – Select; Stricture/stenosis (including anastomotic), GU – Select.
Urine color change Urine color change Present — — — —
REMARK: Urine color refers to change that is not related to other dietary or physiologic cause (e.g., bilirubin, concentrated urine, and hematuria).
Renal/Genitourinary –
Other (Specify, __)
Renal – Other (Specify) Mild Moderate Severe Life-threatening; disabling Death
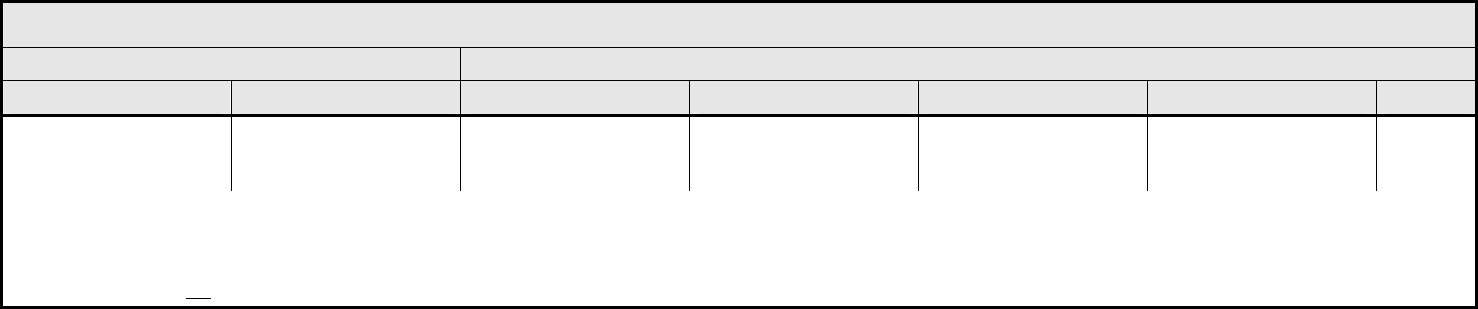
SECONDARY MALIGNANCY Page 1 of 1
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 63 - March 31, 2003, Publish Date: August 9, 2006
Secondary Malignancy –
possibly related to cancer
treatment (Specify, __)
Secondary Malignancy
(possibly related to
cancer treatment)
— — Non-life-threatening basal
or squamous cell
carcinoma of the skin
Solid tumor, leukemia or
lymphoma
Death
REMARK: Secondary malignancy excludes metastasis from initial primary. Any malignancy possibly related to cancer treatment (including AML/MDS) should be reported via the routine
reporting mechanisms outlined in each protocol. Important: Secondary Malignancy is an exception to NCI Expedited Adverse Event Reporting Guidelines. Secondary Malignancy is
“Grade 4, present” but NCI does not require AdEERS Expedited Reporting for any (related or unrelated to treatment) Secondary Malignancy. A diagnosis of AML/MDS following
treatment with an NCI-sponsored investigational agent is to be reported using the form available from the CTEP Web site at http://ctep.cancer.gov. Cancers not suspected of being
treatment-related are not to be reported here.
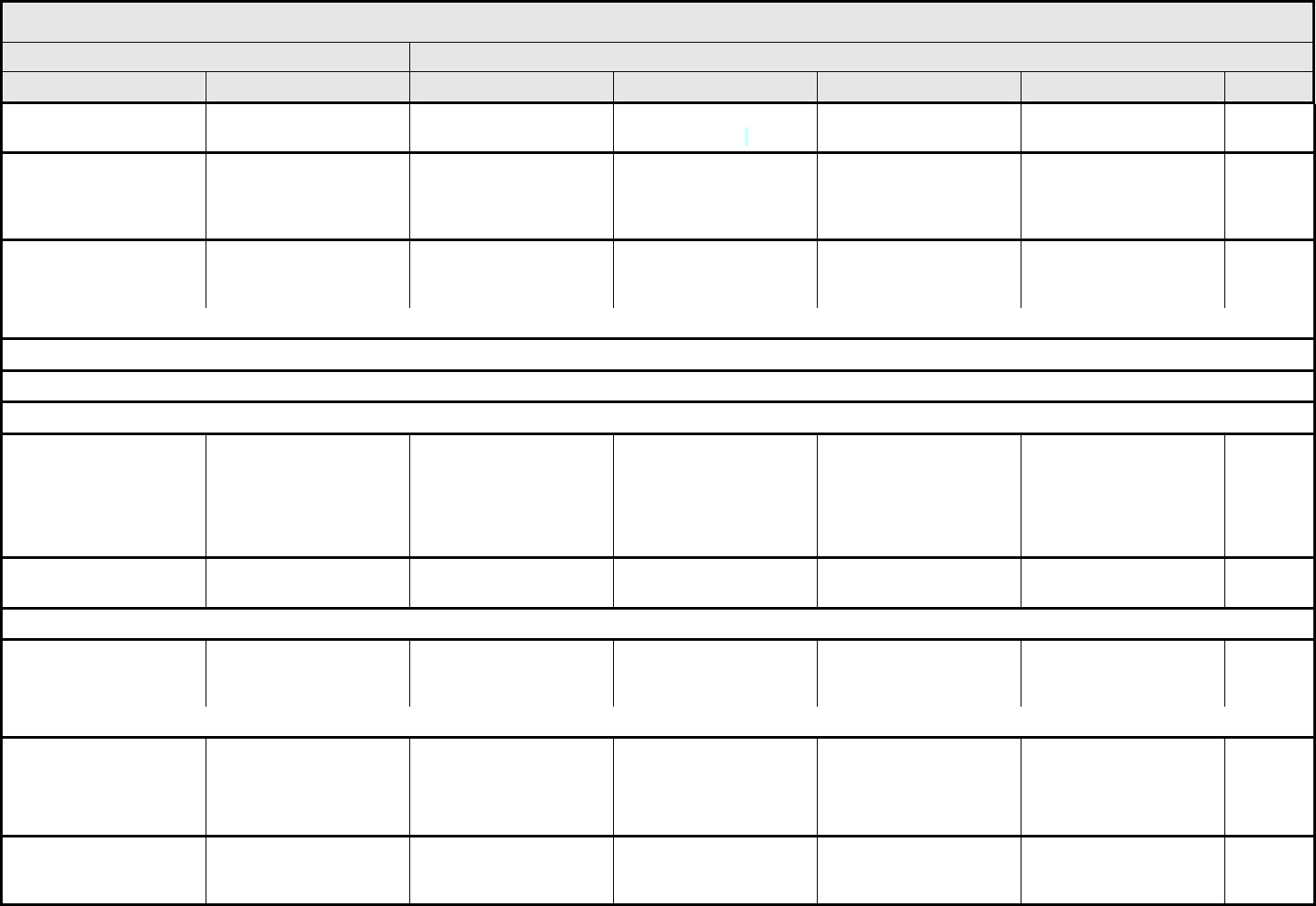
SEXUAL/REPRODUCTIVE FUNCTION Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 64 - March 31, 2003, Publish Date: August 9, 2006
Breast function/lactation Breast function Mammary abnormality,
not functionally significant
Mammary abnormality,
functionally significant
— — —
Breast nipple/areolar
deformity
Nipple/areolar Limited areolar
asymmetry with no
change in nipple/areolar
projection
Asymmetry of nipple
areolar complex with
slight deviation in nipple
projection
Marked deviation of
nipple projection
— —
Breast volume/hypoplasia Breast Minimal asymmetry;
minimal hypoplasia
Asymmetry exists, ≤1/3 of
the breast volume;
moderate hypoplasia
Asymmetry exists, >1/3 of
the breast volume; severe
hypoplasia
— —
REMARK: Breast volume is referenced with both arms straight overhead.
NAVIGATION NOTE: Dysmenorrhea is graded as Pain – Select in the PAIN CATEGORY.
NAVIGATION NOTE: Dyspareunia is graded as Pain – Select in the PAIN CATEGORY.
NAVIGATION NOTE: Dysuria (painful urination) is graded as Pain – Select in the PAIN CATEGORY.
Erectile dysfunction Erectile dysfunction Decrease in erectile
function
(frequency/rigidity of
erections) but erectile
aids not indicated
Decrease in erectile
function
(frequency/rigidity of
erections), erectile aids
indicated
Decrease in erectile
function
(frequency/rigidity of
erections) but erectile
aids not helpful; penile
prosthesis indicated
— —
Ejaculatory dysfunction Ejaculatory dysfunction Diminished ejaculation Anejaculation or
retrograde ejaculation
— — —
NAVIGATION NOTE: Feminization of male is graded in the ENDOCRINE CATEGORY.
Gynecomastia Gynecomastia — Asymptomatic breast
enlargement
Symptomatic breast
enlargement; intervention
indicated
— —
ALSO CONSIDER: Pain – Select.
Infertility/sterility Infertility/sterility — Male: oligospermia/low
sperm count
Female: diminished
fertility/ovulation
Male: sterile/azoospermia
Female: infertile/
anovulatory
— —
Irregular menses
(change from baseline)
Irregular menses 1 – 3 months without
menses
>3 – 6 months without
menses but continuing
menstrual cycles
Persistent amenorrhea
for >6 months
— —
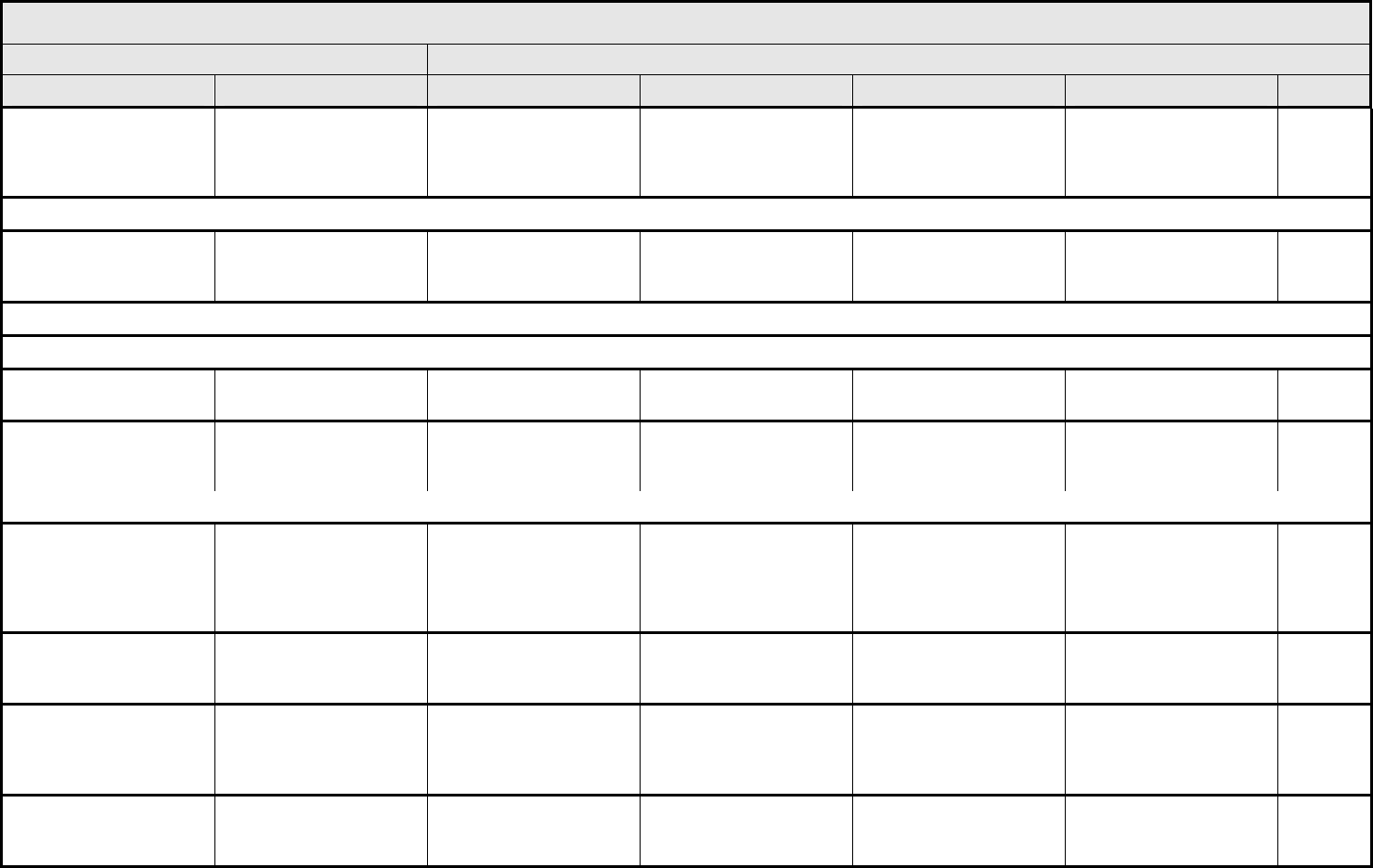
SEXUAL/REPRODUCTIVE FUNCTION Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 65 - March 31, 2003, Publish Date: August 9, 2006
Libido Libido Decrease in interest but
not affecting relationship;
intervention not indicated
Decrease in interest and
adversely affecting
relationship; intervention
indicated
— — —
NAVIGATION NOTE: Masculinization of female is graded in the ENDOCRINE CATEGORY.
Orgasmic dysfunction Orgasmic function Transient decrease Decrease in orgasmic
response requiring
intervention
Complete inability of
orgasmic response; not
responding to intervention
— —
NAVIGATION NOTE: Pelvic pain is graded as Pain – Select in the PAIN CATEGORY.
NAVIGATION NOTE: Ulcers of the labia or perineum are graded as Ulceration in DERMATOLOGY/SKIN CATEGORY.
Vaginal discharge
(non-infectious)
Vaginal discharge Mild Moderate to heavy; pad
use indicated
— — —
Vaginal dryness Vaginal dryness Mild Interfering with sexual
function; dyspareunia;
intervention indicated
— — —
ALSO CONSIDER: Pain – Select.
Vaginal mucositis Vaginal mucositis Erythema of the mucosa;
minimal symptoms
Patchy ulcerations;
moderate symptoms or
dyspareunia
Confluent ulcerations;
bleeding with trauma;
unable to tolerate vaginal
exam, sexual intercourse
or tampon placement
Tissue necrosis;
significant spontaneous
bleeding; life-threatening
consequences
—
Vaginal stenosis/length Vaginal stenosis Vaginal narrowing and/or
shortening not interfering
with function
Vaginal narrowing and/or
shortening interfering with
function
Complete obliteration; not
surgically correctable
— —
Vaginitis (not due to
infection)
Vaginitis Mild, intervention not
indicated
Moderate, intervention
indicated
Severe, not relieved with
treatment; ulceration, but
operative intervention not
indicated
Ulceration and operative
intervention indicated
—
Sexual/Reproductive
Function – Other
(Specify, __)
Sexual – Other (Specify) Mild Moderate Severe Disabling Death
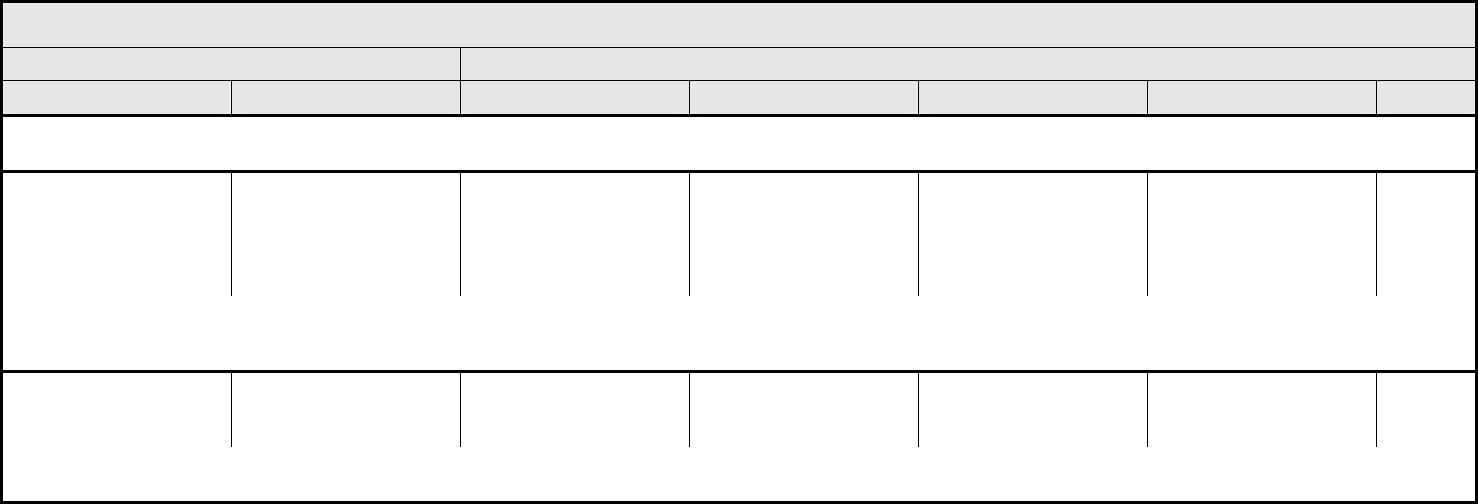
SURGERY/INTRA-OPERATIVE INJURY Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 66 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Intra-operative hemorrhage is graded as Hemorrhage/bleeding associated with surgery, intra-operative or postoperative in the HEMORRHAGE/BLEEDING
CATEGORY.
Intra-operative injury
– Select Organ or
Structure
‘Select’ AEs appear at the
end of the CATEGORY.
Intraop injury – Select Primary repair of injured
organ/structure indicated
Partial resection of
injured organ/structure
indicated
Complete resection or
reconstruction of injured
organ/structure indicated
Life threatening
consequences; disabling
—
REMARK: The ‘Select’ AEs are defined as significant, unanticipated injuries that are recognized at the time of surgery. These AEs do not refer to additional surgical procedures that
must be performed because of a change in the operative plan based on intra-operative findings. Any sequelae resulting from the intra-operative injury that result in an adverse
outcome for the patient must also be recorded and graded under the relevant CTCAE Term.
Intra-operative Injury –
Other (Specify, __)
Intraop Injury – Other
(Specify)
Primary repair of injured
organ/structure indicated
Partial resection of
injured organ/structure
indicated
Complete resection or
reconstruction of injured
organ/structure indicated
Life threatening
consequences; disabling
—
REMARK: Intra-operative Injury – Other (Specify, __) is to be used only to report an organ/structure not included in the ‘Select’ AEs found at the end of the CATEGORY. Any sequelae
resulting from the intra-operative injury that result in an adverse outcome for the patient must also be recorded and graded under the relevant CTCAE Term.

SURGERY/INTRA-OPERATIVE INJURY – SELECT Page 2 of 2
CTCAE v3.0 - 67 - March 31, 2003, Publish Date: August 9, 2006
AUDITORY/EAR
– Inner ear
– Middle ear
– Outer ear NOS
– Outer ear-Pinna
CARDIOVASCULAR
– Artery-aorta
– Artery-carotid
– Artery-cerebral
– Artery-extremity (lower)
– Artery-extremity (upper)
– Artery-hepatic
– Artery-major visceral artery
– Artery-pulmonary
– Artery NOS
– Heart
– Spleen
– Vein-extremity (lower)
– Vein-extremity (upper)
– Vein-hepatic
– Vein-inferior vena cava
– Vein-jugular
– Vein-major visceral vein
– Vein-portal vein
– Vein-pulmonary
– Vein-superior vena cava
– Vein NOS
DERMATOLOGY/SKIN
– Breast
– Nails
– Skin
ENDOCRINE
– Adrenal gland
– Parathyroid
– Pituitary
ENDOCRINE
(continued)
– Thyroid
HEAD AND NECK
– Gingiva
– Larynx
– Lip/perioral area
– Face NOS
– Nasal cavity
– Nasopharynx
– Neck NOS
– Nose
– Oral cavity NOS
– Parotid gland
– Pharynx
– Salivary duct
– Salivary gland
– Sinus
– Teeth
– Tongue
– Upper aerodigestive NOS
GASTROINTESTINAL
– Abdomen NOS
– Anal sphincter
– Anus
– Appendix
– Cecum
– Colon
– Duodenum
– Esophagus
– Ileum
– Jejunum
– Oral
– Peritoneal cavity
– Rectum
– Small bowel NOS
GASTROINTESTINAL
(continued)
– Stoma (GI)
– Stomach
HEPATOBILIARY/ PANCREAS
– Biliary tree-common bile
duct
– Biliary tree-common hepatic
duct
– Biliary tree-left hepatic duct
– Biliary tree-right hepatic duct
– Biliary tree NOS
– Gallbladder
– Liver
– Pancreas
– Pancreatic duct
MUSCULOSKELETAL
– Bone
– Cartilage
– Extremity-lower
– Extremity-upper
– Joint
– Ligament
– Muscle
– Soft tissue NOS
– Tendon
NEUROLOGY
– Brain
– Meninges
– Spinal cord
NERVES:
– Brachial plexus
– CN I (olfactory)
– CN II (optic)
– CN III (oculomotor)
– CN IV (trochlear)
NEUROLOGY
(continued)
NERVES:
– CN V (trigeminal) motor
– CN V (trigeminal) sensory
– CN VI (abducens)
– CN VII (facial) motor-face
– CN VII (facial) sensory-
taste
– CN VIII (vestibulocochlear)
– CN IX (glossopharyngeal)
motor pharynx
– CN IX (glossopharyngeal)
sensory ear-pharynx-
tongue
– CN X (vagus)
– CN XI (spinal accessory)
– CN XII (hypoglossal)
– Cranial nerve or branch
NOS
– Lingual
– Lung thoracic
– Peripheral motor NOS
– Peripheral sensory NOS
– Recurrent laryngeal
– Sacral plexus
– Sciatic
– Thoracodorsal
OCULAR
– Conjunctiva
– Cornea
– Eye NOS
– Lens
– Retina
PULMONARY/UPPER
RESPIRATORY
– Bronchus
– Lung
– Mediastinum
– Pleura
– Thoracic duct
– Trachea
– Upper airway NOS
RENAL/GENITOURINARY
– Bladder
– Cervix
– Fallopian tube
– Kidney
– Ovary
– Pelvis NOS
– Penis
– Prostate
– Scrotum
– Testis
– Ureter
– Urethra
– Urinary conduit
– Urinary tract NOS
– Uterus
– Vagina
– Vulva
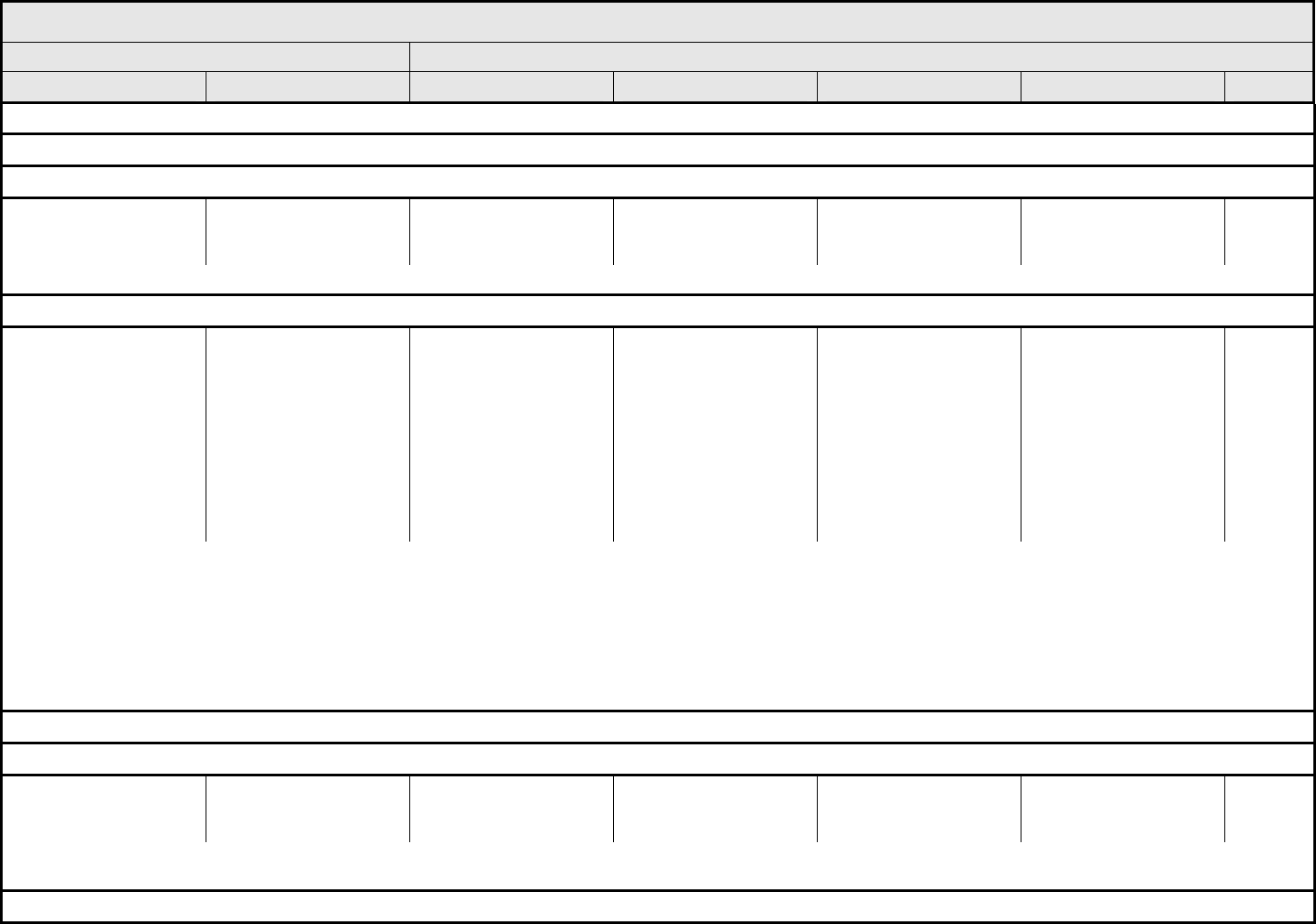
SYNDROMES Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 68 - March 31, 2003, Publish Date: August 9, 2006
NAVIGATION NOTE: Acute vascular leak syndrome is graded in the VASCULAR CATEGORY.
NAVIGATION NOTE: Adrenal insufficiency is graded in the ENDOCRINE CATEGORY.
NAVIGATION NOTE: Adult Respiratory Distress Syndrome (ARDS) is graded in the PULMONARY/UPPER RESPIRATORY CATEGORY.
Alcohol intolerance
syndrome (antabuse-like
syndrome)
Alcohol intolerance
syndrome
— — Present — Death
REMARK: An antabuse-like syndrome occurs with some new anti-androgens (e.g., nilutamide) when patient also consumes alcohol.
NAVIGATION NOTE: Autoimmune reaction is graded as Autoimmune reaction/hypersensitivity (including drug fever) in the ALLERGY/IMMUNOLOGY CATEGORY.
Cytokine release
syndrome/acute infusion
reaction
Cytokine release
syndrome
Mild reaction; infusion
interruption not indicated;
intervention not indicated
Requires therapy or
infusion interruption but
responds promptly to
symptomatic treatment
(e.g., antihistamines,
NSAIDS, narcotics, IV
fluids); prophylactic
medications indicated
for ≤24 hrs
Prolonged (i.e., not
rapidly responsive to
symptomatic medication
and/or brief interruption of
infusion); recurrence of
symptoms following initial
improvement;
hospitalization indicated
for other clinical sequelae
(e.g., renal impairment,
pulmonary infiltrates)
Life-threatening; pressor
or ventilatory support
indicated
Death
REMARK: Cytokine release syndromes/acute infusion reactions are different from Allergic/hypersensitive reactions, although some of the manifestations are common to both AEs. An
acute infusion reaction may occur with an agent that causes cytokine release (e.g., monoclonal antibodies or other biological agents). Signs and symptoms usually develop during or
shortly after drug infusion and generally resolve completely within 24 hrs of completion of infusion. Signs/symptoms may include: Allergic reaction/hypersensitivity (including drug
fever); Arthralgia (joint pain); Bronchospasm; Cough; Dizziness; Dyspnea (shortness of breath); Fatigue (asthenia, lethargy, malaise); Headache; Hypertension; Hypotension; Myalgia
(muscle pain); Nausea; Pruritis/itching; Rash/desquamation; Rigors/chills; Sweating (diaphoresis); Tachycardia; Tumor pain (onset or exacerbation of tumor pain due to treatment);
Urticaria (hives, welts, wheals); Vomiting.
A
LSO CONSIDER: Allergic reaction/hypersensitivity (including drug fever); Bronchospasm, wheezing; Dyspnea (shortness of breath); Hypertension; Hypotension; Hypoxia; Prolonged
QTc interval; Supraventricular and nodal arrhythmia – Select; Ventricular arrhythmia – Select.
NAVIGATION NOTE: Disseminated intravascular coagulation (DIC) is graded in the COAGULATION CATEGORY.
NAVIGATION NOTE: Fanconi’s syndrome is graded as Urinary electrolyte wasting (e.g., Fanconi’s syndrome, renal tubular acidosis) in the RENAL/GENITOURINARY CATEGORY.
Flu-like syndrome Flu-like syndrome Symptoms present but
not interfering with
function
Moderate or causing
difficulty performing some
ADL
Severe symptoms
interfering with ADL
Disabling Death
REMARK: Flu-like syndrome represents a constellation of symptoms which may include cough with catarrhal symptoms, fever, headache, malaise, myalgia, prostration, and is to be
used when the symptoms occur in a cluster consistent with one single pathophysiological process.
NAVIGATION NOTE: Renal tubular acidosis is graded as Urinary electrolyte wasting (e.g., Fanconi’s syndrome, renal tubular acidosis) in the RENAL/GENITOURINARY CATEGORY.
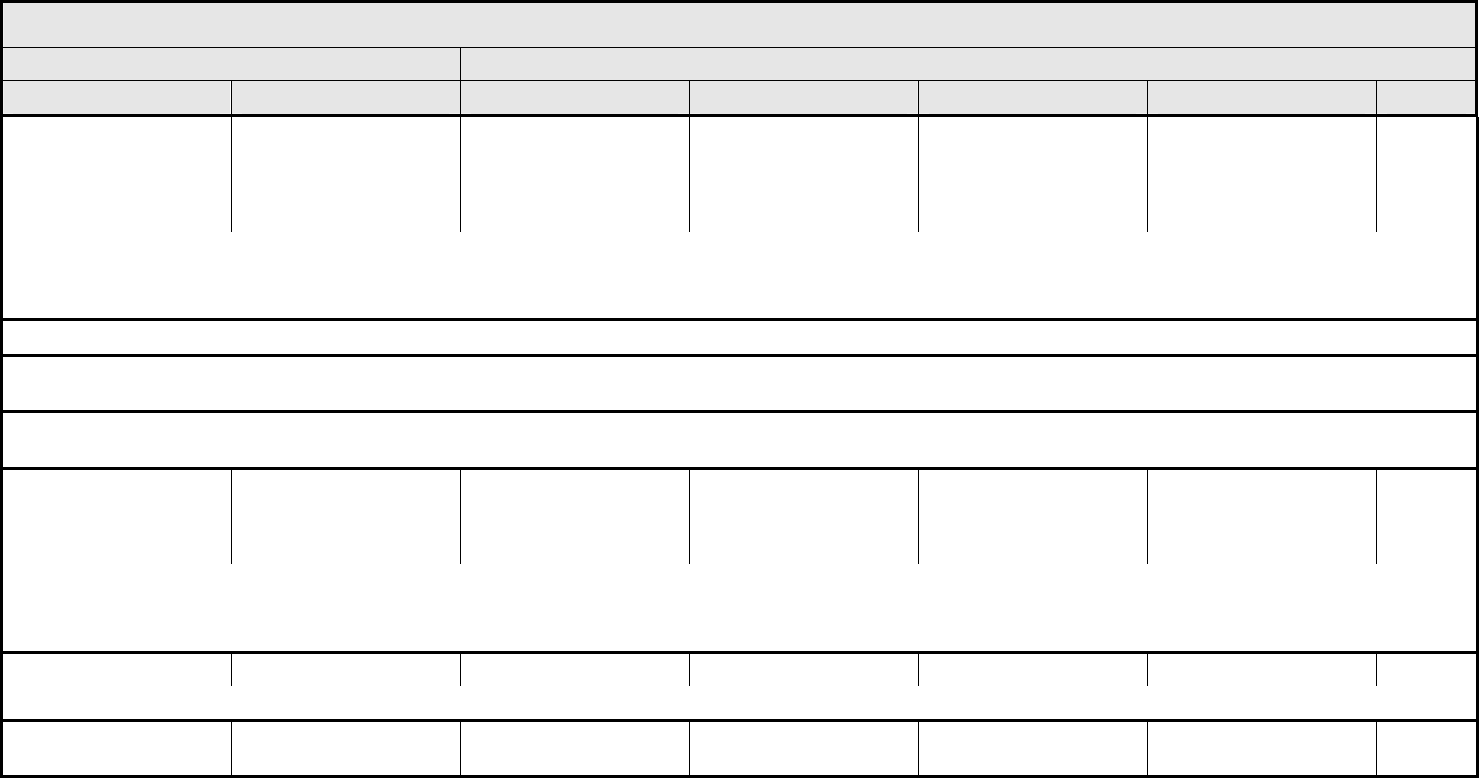
SYNDROMES Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 69 - March 31, 2003, Publish Date: August 9, 2006
“Retinoic acid syndrome” “Retinoic acid syndrome” Fluid retention; less than
3 kg of weight gain;
intervention with fluid
restriction and/or diuretics
indicated
Mild to moderate signs/
symptoms; steroids
indicated
Severe signs/symptoms;
hospitalization indicated
Life-threatening;
ventilatory support
indicated
Death
REMARK: Patients with acute promyelocytic leukemia may experience a syndrome similar to “retinoic acid syndrome” in association with other agents such as arsenic trioxide. The
syndrome is usually manifested by otherwise unexplained fever, weight gain, respiratory distress, pulmonary infiltrates and/or pleural effusion, with or without leukocytosis.
A
LSO CONSIDER: Acute vascular leak syndrome; Pleural effusion (non-malignant); Pneumonitis/pulmonary infiltrates.
NAVIGATION NOTE: SIADH is graded as Neuroendocrine: ADH secretion abnormality (e.g., SIADH or low ADH) in the ENDOCRINE CATEGORY.
NAVIGATION NOTE: Stevens-Johnson syndrome is graded as Rash: erythema multiforme (e.g., Stevens-Johnson syndrome, toxic epidermal necrolysis) in the DERMATOLOGY/SKIN
CATEGORY.
NAVIGATION NOTE: Thrombotic microangiopathy is graded as Thrombotic microangiopathy (e.g., thrombotic thrombocytopenic purpura [TTP] or hemolytic uremic syndrome [HUS]) in
the COAGULATION CATEGORY.
Tumor flare Tumor flare Mild pain not interfering
with function
Moderate pain; pain or
analgesics interfering with
function, but not
interfering with ADL
Severe pain; pain or
analgesics interfering with
function and interfering
with ADL
Disabling Death
REMARK: Tumor flare is characterized by a constellation of signs and symptoms in direct relation to initiation of therapy (e.g., anti-estrogens/androgens or additional hormones). The
symptoms/signs include tumor pain, inflammation of visible tumor, hypercalcemia, diffuse bone pain, and other electrolyte disturbances.
A
LSO CONSIDER: Calcium, serum-high (hypercalcemia).
Tumor lysis syndrome Tumor lysis syndrome — — Present — Death
ALSO CONSIDER: Creatinine; Potassium, serum-high (hyperkalemia).
Syndromes – Other
(Specify, __)
Syndromes – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
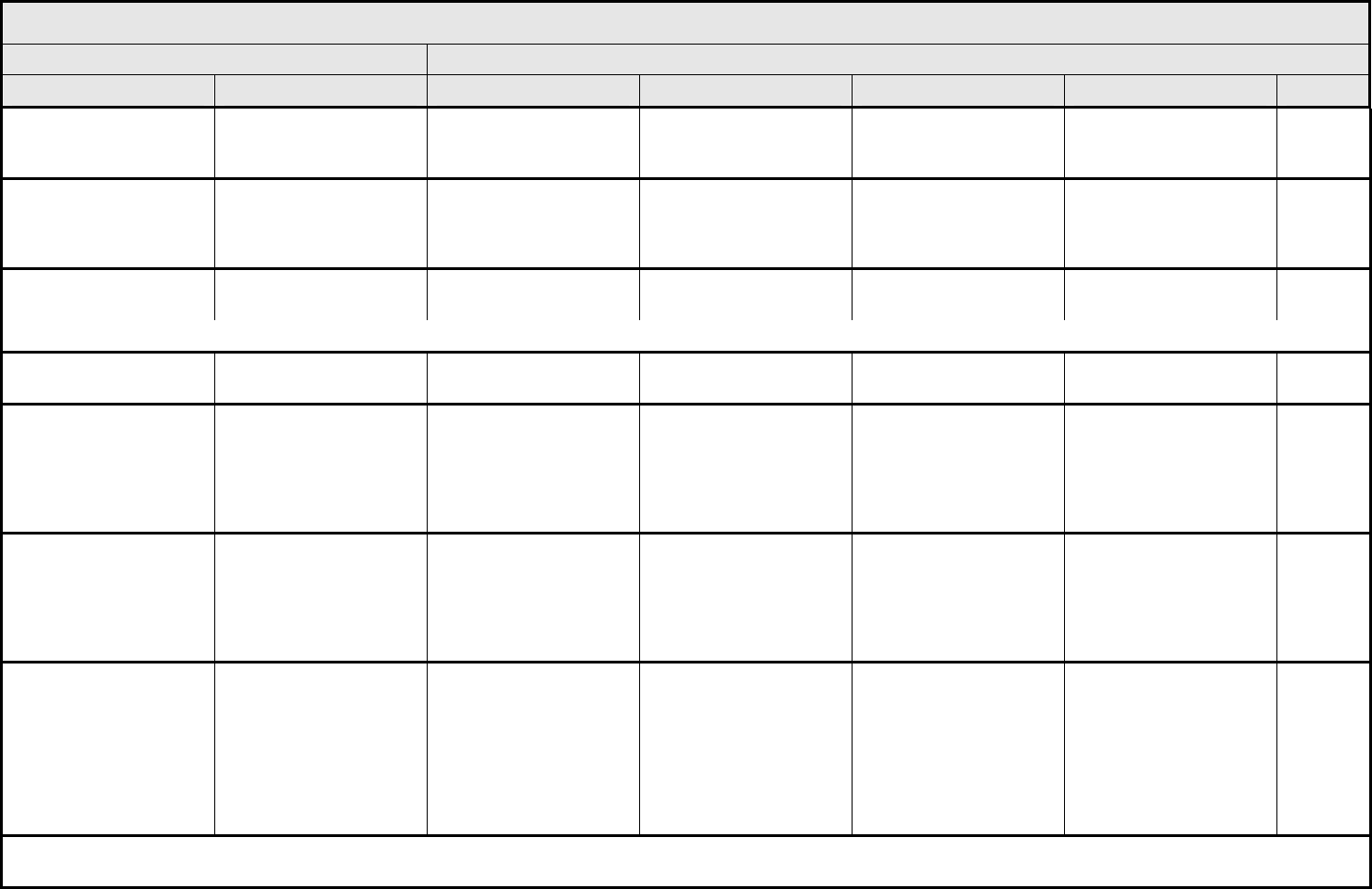
VASCULAR Page 1 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 70 - March 31, 2003, Publish Date: August 9, 2006
Acute vascular leak
syndrome
Acute vascular leak
syndrome
— Symptomatic, fluid
support not indicated
Respiratory compromise
or fluids indicated
Life-threatening; pressor
support or ventilatory
support indicated
Death
Peripheral arterial
ischemia
Peripheral arterial
ischemia
— Brief (<24 hrs) episode of
ischemia managed non-
surgically and without
permanent deficit
Recurring or prolonged
(≥24 hrs) and/or invasive
intervention indicated
Life-threatening, disabling
and/or associated with
end organ damage
(e.g., limb loss)
Death
Phlebitis (including
superficial thrombosis)
Phlebitis — Present — — —
ALSO CONSIDER: Injection site reaction/extravasation changes.
Portal vein flow Portal flow — Decreased portal vein
flow
Reversal/retrograde
portal vein flow
— —
Thrombosis/embolism
(vascular access-related)
Thrombosis/embolism
(vascular access)
— Deep vein thrombosis or
cardiac thrombosis;
intervention (e.g.,
anticoagulation, lysis,
filter, invasive procedure)
not indicated
Deep vein thrombosis or
cardiac thrombosis;
intervention (e.g.,
anticoagulation, lysis,
filter, invasive procedure)
indicated
Embolic event including
pulmonary embolism or
life-threatening thrombus
Death
Thrombosis/thrombus/
embolism
Thrombosis/thrombus/
embolism
— Deep vein thrombosis or
cardiac thrombosis;
intervention (e.g.,
anticoagulation, lysis,
filter, invasive procedure)
not indicated
Deep vein thrombosis or
cardiac thrombosis;
intervention (e.g.,
anticoagulation, lysis,
filter, invasive procedure)
indicated
Embolic event including
pulmonary embolism or
life-threatening thrombus
Death
Vessel injury-artery
– Select:
– Aorta
– Carotid
– Extremity-lower
– Extremity-upper
– Other NOS
– Visceral
Artery injury – Select Asymptomatic diagnostic
finding; intervention not
indicated
Symptomatic (e.g.,
claudication); not
interfering with ADL;
repair or revision not
indicated
Symptomatic interfering
with ADL; repair or
revision indicated
Life-threatening;
disabling; evidence of
end organ damage (e.g.,
stroke, MI, organ or limb
loss)
Death
NAVIGATION NOTE: Vessel injury to an artery intra-operatively is graded as Intra-operative injury – Select Organ or Structure in the SURGERY/INTRA-OPERATIVE INJURY
CATEGORY.
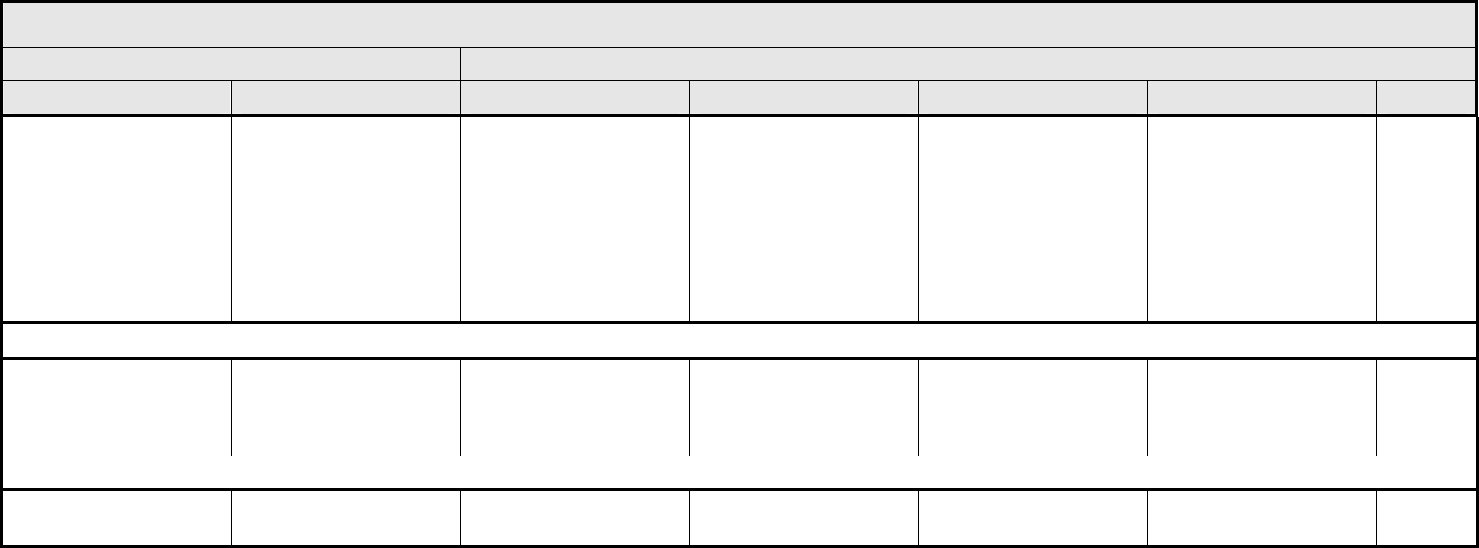
VASCULAR Page 2 of 2
Grade
Adverse Event Short Name 1 2 3 4 5
CTCAE v3.0 - 71 - March 31, 2003, Publish Date: August 9, 2006
Vessel injury-vein
– Select:
– Extremity-lower
– Extremity-upper
– IVC
– Jugular
– Other NOS
– SVC
– Viscera
Vein injury – Select Asymptomatic diagnostic
finding; intervention not
indicated
Symptomatic (e.g.,
claudication); not
interfering with ADL;
repair or revision not
indicated
Symptomatic interfering
with ADL; repair or
revision indicated
Life-threatening;
disabling; evidence of
end organ damage
Death
NAVIGATION NOTE: Vessel injury to a vein intra-operatively is graded as Intra-operative injury – Select Organ or Structure in the SURGERY/INTRA-OPERATIVE INJURY CATEGORY.
Visceral arterial ischemia
(non-myocardial)
Visceral arterial ischemia — Brief (<24 hrs) episode of
ischemia managed
medically and without
permanent deficit
Prolonged (≥24 hrs) or
recurring symptoms
and/or invasive
intervention indicated
Life-threatening;
disabling; evidence of
end organ damage
Death
ALSO CONSIDER: CNS cerebrovascular ischemia.
Vascular – Other
(Specify, __)
Vascular – Other
(Specify)
Mild Moderate Severe Life-threatening; disabling Death
