
NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 1 of 31
February 2023
National Healthcare Safety Network
Biovigilance Component
Hemovigilance Module
Surveillance Protocol
Division of Healthcare Quality Promotion
National Center for Emerging and Zoonotic Infectious Diseases
Centers for Disease Control and Prevention
Atlanta, GA, USA
NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 2 of 31
February 2023
Version History
Version
Release Date
Summary of Revisions
1.0
March 2009
First version publicly released.
1.1
June 2010
Revised background and text in main body of document.
Revised case definition criterion based on WG recommendations, pilot responses,
and CDC recommendations.
Updated FNHTR definition to allow reaction without documented fever.
Defined hypotension for infants and small children
Clarified TAGVHD probable and possible criteria.
1.2
July 2010
Corrected definition of hypoxemia in glossary of terms.
1.3
June 2011
Added version number and version history summary.
Summarized introduction and background sections for brevity.
Reorganized surveillance methods section for ease of use.
Clarified reporting of “approved deviation” incidents.
Clarified use of “other” in adverse reaction reporting.
Clarified use of “doubtful” or “ruled out” in adverse reaction reporting.
Added denominator summary options to list of available analysis reports.
Replaced < and > signs with appropriate text for.
Added “cessation of” to time frame requirements in case definitions.
NEW probable case definition category for allergic reaction reporting.
Updated adult hypotensive reaction case definition to align with updated ISBT
definition.
NEW possible imputability category for DHTR.
DELETED possible case definition category for hypotensive reaction.
NEW probable imputability category for PTP reaction.
Updated and clarified imputability categories for TAGVHD reaction.
DELETED possible case definition category for TRALI.
Simplified imputability criteria for TTI.
Clarified case definition and imputability criteria for all adverse reactions.
2.0
January 2013
Complete revision of organization and presentation of information
Major change in incident reporting requirements. With this release, only incidents
that relate to an adverse patient reaction are required for participation.
Major change in adverse reaction reporting requirements. With this release, minor
allergic reactions are no longer required for participation.
Combined the signs/symptoms with laboratory/radiology columns in case definition
tables for clarity. Listed criteria in alphabetical order where possible for consistency
and clarity. Moved general severity requirements from the appendix to the criteria
tables where they were previously missing.
Re-ordered adverse reaction tables to put respiratory reactions first.
Added Imputability criteria of Doubtful, Ruled Out, and Not Determined to the case
definition tables as OPTIONAL reporting categories. The reporting is not a change,
but including them in the table is new. They were added for clarity.
Added specific AHTR criteria to allow for reporting of non-immune mediated
reactions.
Added a separate case definition table for Other and Unknown reactions. These
categories are available for OPTONAL use.
Removed redundant and unnecessary appendices.
2.1
August 2013
Minor revisions to verbiage throughout for clarity.
Added definitions and illustration of surveillance key terms in Section 1.
Added clarification of surveillance vs. clinical definitions in Section 1.
Added less-specific case definition categories for OPTIONAL reporting of cases
that do not fully meet CDC case criteria for the following reactions: hypotension,
febrile non-hemolytic, acute hemolytic and delayed hemolytic.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 3 of 31
February 2023
Version
Release Date
Summary of Revisions
Added a possible case definition category for TTI for OPTIONAL reporting of
syndromic cases that are not laboratory confirmed.
2.1.1
September 2013
Updated diagram in Section 1 and added version history for v2.0 and v2.1.
2.1.2
January 2014
Updated the incident codes in Section 4 and included required reporting of discards
and total crossmatch procedures on the Monthly Reporting Denominators form in
Section 5.
2.1.3
August 2014
Added a suggested citation for the surveillance protocol in Section 1. Updated the
acute hemolytic case definition in Section 3 for clarity. Updated the reporting
requirements in Section 5 for clarity.
2.2
January 2016
Updated contact instructions for consistency in Section 1: User support
Updated version number in Section 1: Suggested Citation
Remove Root Cause Analysis Result from Section 4: Incident Glossary
Updated denominator report description to include Pathogen-reduced products in
Section 5: Required Reporting
2.3
June 2016
Updated denominator report description to include Table 3 description.
2.4
January 2017
Section 1: Setting – Added additional Annual Facility form for Non-Acute Care
Facilities to report.
Section 2: Annual Facility Survey – Added information about Non-Acute Care
Facility Annual Facility Survey, Added links to the Annual Facility Survey – Non-
Acute Care Facility form and table of instructions for clarity.
2.5
January 2018
Section 1: Training, User Support, Data Reporting – Minor language changes for
clarification
Section 3: Adverse Reaction Classification – Added information about module-
generated classification designations.
Adverse Reaction Glossary: Updated the definition of fever to be consistent with
FNHTR criteria.
2.5.2.
April 2018
Section 4: Incident codes - UT 06 – “incompatible” replaced with “unapproved”
2.6
March 2021
Section 3: Adverse Reaction Classification - Updated case definition criteria for
TACO reactions
2.7
October 2022
Section 4: Updated the incident codes in Section 4 to include under-transfusion with
the creation of a new process code: no blood (NB) and four incident codes
2.8
February 2023
Section 4: Optional Reporting: Clarification regarding analysis of optional reporting

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 4 of 31
February 2023
Table of Contents
Section 1. Hemovigilance Module Surveillance Overview .........................................5
Section 2. Hemovigilance Module Annual Facility Survey .........................................7
Section 3: Hemovigilance Module Adverse Reactions ...............................................8
Adverse Reaction Case Classification Criteria Tables .................................................... 9
Transfusion-associated circulatory overload (TACO) ....................................................................... 9
Transfusion-related acute lung injury (TRALI) .................................................................................10
Transfusion-associated dyspnea (TAD) ..........................................................................................11
Allergic reaction ..............................................................................................................................12
Hypotensive transfusion reaction ....................................................................................................13
Febrile non-hemolytic transfusion reaction (FNHTR) .......................................................................14
Acute hemolytic transfusion reaction (AHTR) ..................................................................................15
Delayed hemolytic transfusion reaction (DHTR) ..............................................................................16
Delayed serologic transfusion reaction (DSTR) ...............................................................................17
Transfusion-associated graft vs. host disease (TAGVHD) ...............................................................18
Post transfusion purpura (PTP) ......................................................................................................19
Transfusion-transmitted infection (TTI)............................................................................................20
Other or Unknown ..........................................................................................................................22
Adverse Reaction Glossary ........................................................................................... 23
Section 4. Hemovigilance Module Incidents ..............................................................24
Incident Codes ............................................................................................................... 25
Occupation Codes.......................................................................................................... 28
Incident Glossary ...........................................................................................................
30
Section 5. Hemovigilance Module Denominators ......................................................31

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 5 of 31
February 2023
Section 1. Hemovigilance Module Surveillance Overview
Purpose
The National Healthcare Safety Network (NHSN) Hemovigilance (HV) Module was created to implement
national surveillance of transfusion-associated adverse events aimed at improving patient safety,
minimizing morbidity and mortality of transfusion recipients, and identifying emerging complications and
pathogens associated with blood transfusion.
Settings
The Hemovigilance Module may be used by any U.S. healthcare facility where blood components and
manufactured blood products are transfused (e.g., adult or pediatric facilities, acute or non-acute care
facilities). Surveillance must be performed facility-wide, including patient care areas for emergency,
general medical, and surgical patients; obstetrics and gynecology; orthopedics, oncology, and other
chronic diseases; and any other facility location where transfusions are administered.
Methods
The NHSN Hemovigilance Module requires comprehensive surveillance of patients and blood
components throughout the transfusion process, from product receipt to administration to the patient.
Participation in the NHSN Hemovigilance Module requires reporting of all adverse transfusion reactions
and reaction-associated incidents that occur for patients transfused at or by your facility as well as a
monthly summary of components transfused or discarded and patient samples collected for type and
screen or crossmatch.
Data Collection
NHSN is a web-based application used by healthcare facilities to report surveillance data. Paper
versions of all forms are used to collect data prior to data entry in the NHSN Hemovigilance Module. The
paper forms are available on the NHSN Blood Safety Surveillance website.
A link to the appropriate
form(s) and their instructions is provided in the following sections for your convenience.
T
raining
Training presentations are available on the NHSN Blood Safety Surveillance website for self-pac
ed
training and must be reviewed prior to participating in the Hemovigilance Module. CDC also provides
webinar and in-person training opportunities for current NHSN participants. These opportunities are
communicated through the NHSN quarterly newsletter and emails from the Hemovigilance Team.
U
ser Support
CDC is available to answer your questions about the Surveillance Protocol and to help navigate the
NHSN web application. Please contact us at nhsn@cdc.gov. Type HE
MOVIGILANCE in the subject line
for quickest routing to the Hemovigilance Team.
S
uggested Citation for the Hemovigilance Module Surveillance Protocol
U.S. Centers for Disease Control and Prevention. The National Healthcare Safety Network
(NHSN) Manual: Biovigilance Component v2.5. Atlanta, GA: Division of Healthcare Quality
Promotion, National Center for Emerging and Zoonotic Infectious Diseases. Available at:
http://www.cdc.gov/nhsn/PDFs/Biovigilance/BV-HV-protocol-current.pdf. Accessed [enter date].
NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 6 of 31
February 2023
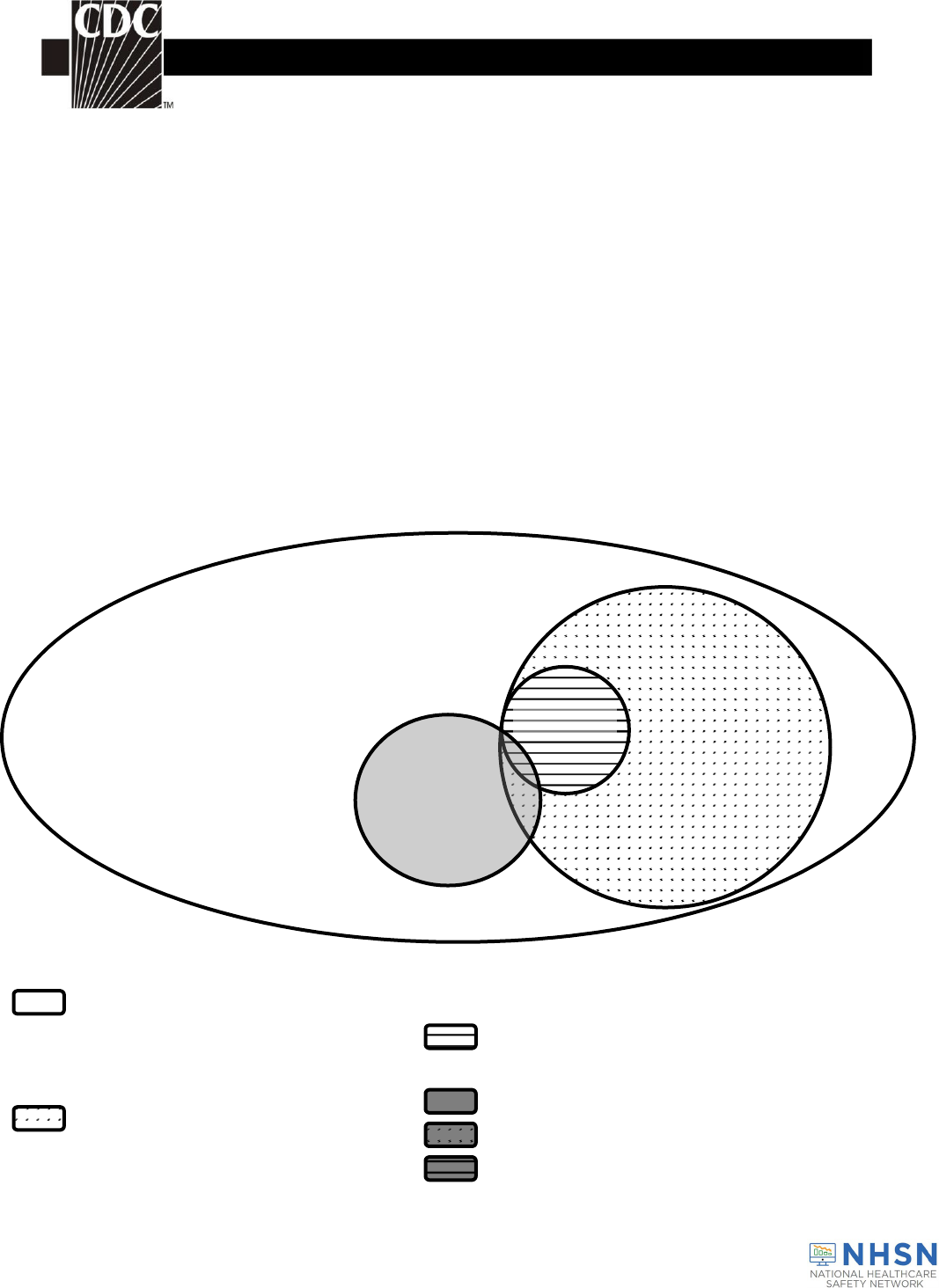
Key Terms (see Fig. 1)
• Adverse event: An unintended and undesirable occurrence before, during or after transfusion of
blood or blood components. Adverse events include both incidents and adverse reactions.
• Adverse reaction: An undesirable response or effect in a patient temporally associated with the
administration of blood or blood components. It may or may not be the result of an incident.
• Incident: Any error or accident that could affect the quality or efficacy of blood, blood components,
or patient transfusions. It may or may not result in an adverse reaction in a transfusion recipient.
• Near miss: A subset of incidents that are discovered before the start of a transfusion that could
have led to a wrongful transfusion or an adverse reaction in a transfusion recipient.
Data Reporting (See Fig. 1)
• An annual facility demographic and practice survey for each calendar year of participation
• ALL adverse reactions defined in this protocol that follow transfusion at or by your facility
• ALL incidents (i.e., errors or accidents) associated with an adverse reaction
• The number of blood components transfused or discarded and patient samples collected for type
and screen or crossmatch each month
Figure 1. Venn diagram of NHSN Hemovigilance Module surveillance terms.
Transfusion-Related Activities
• Patient Sample Collection
• Sample Handling and Testing
• Inventory Management
• Patient Monitoring
Transfusion
• Number of Components
• Number of Patients
Adverse Events
Reactions
Incidents
Near Miss Incidents
Incidents Related to Transfusion (No Adverse Reaction)
Incidents Related to Transfusion and Adverse Reaction
Transfusion-Related
Activities
Transfusions
Incidents
Reactions

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 7 of 31
February 2023
Section 2. Hemovigilance Module Annual Facility Survey
Required Reporting
Participating facilities must enter the Hemovigilance Module Annual Facility Survey at the time that they
enroll or activate the Biovigilance Component and at the beginning of each calendar year thereafter. The
survey is used by CDC to classify facilities for appropriate comparisons in aggregate data analyses and
to learn more about common practices among transfusion services. The data collected in the survey
covers the previous calendar year. For example, if the facility is enrolling in NHSN for the first time in
October of 2013, report information for January 2012-December 2012 on the first Hemovigilance Module
Annual Facility Survey. In January 2014, complete a new survey with data from January 2013-December
2013. CDC recommends collecting all survey information on a paper form before attempting to enter
data into the web application.
As of January 2017, non-acute care facilities are able to report hemovigilance data to NHSN. Non-acute
care facilities should complete Annual Facility Survey for Non-acute care facility 57.306. This form
contains questions tailored to non-acute care facilities. Users may refer to the Non-Acute Care Facility
Table of Instructions form 57.306 for detailed instruction about data collection.
Form
CDC 57.300 Hemovigilance Module Annual Facility Survey - Acute Care Facility
CDC 57.306 Hemovigilance Module Annual Facility Survey - Non-Acute Care Facility
Form Instructions
CDC 57.300 Hemovigilance Module Annual Facility Survey - Acute Care Facility Table of Instructions
CDC 57.306 Hemovigilance Module Annual Facility Survey - Non-Acute Care Facility Table of
Instructions

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 8 of 31
February 2023
Section 3: Hemovigilance Module Adverse Reactions
Required Reporting
All CDC-defined transfusion-associated adverse reactions that are possibly, probably, or definitely
related to a transfusion performed by the participating facility must be reported to NHSN. If a patient
experiences more than one adverse reaction during or following the same transfusion episode, complete
a separate form for each reaction. Adverse reaction reports should be entered into NHSN after an
investigation of the reaction has been completed and imputability has been determined to the extent
possible. Reports should be entered within 30 days of the month that the reaction occurred or when the
investigation is completed.
Optional Reporting
Reporting suspected adverse reactions where imputability is determined to be doubtful or ruled out is not
required. A facility may report reactions determined to be doubtful or ruled out in order to use NHSN to
document transfusion reaction investigations each month. Adverse reactions that are not defined in the
surveillance protocol may also be reported using the ‘Other’ and ‘Unknown’ adverse reaction categories;
standard severity and imputability criteria are provided for that purpose.
Adverse Reaction Classification
Each CDC-defined transfusion-associated adverse reaction must be classified according to the reaction-
specific case definition, severity, and imputability criteria printed in the protocol. It is imperative that
every facility classify adverse reactions according to protocol definitions. Accurate classification will
usually require a detailed review of the patient record.
To assist in classification, the Module will generate and assign designations for case definition, severity,
and imputability based on signs, symptoms, and lab results entered in the investigation results section of
the adverse reaction form.
Surveillance definitions are distinctly different from clinical definitions. Surveillance definitions are
designed to capture data consistently and reliably in order to identify trends and inform quality
improvement practices. The surveillance definitions are not intended as clinical diagnostic criteria or to
provide treatment guidance.
Defined Adverse Reactions
• Transfusion-associated circulatory overload (TACO)
• Transfusion-related acute lung injury (TRALI)
• Transfusion-associated dyspnea (TAD)
• Allergic reaction (where severity is severe, life threatening, or death)
• Hypotensive transfusion reaction
• Febrile non-hemolytic transfusion reaction (FNHTR)
• Acute hemolytic transfusion reaction (AHTR)
• Delayed hemolytic transfusion reaction (DHTR)
• Delayed serologic transfusion reaction (DSTR)
• Transfusion-associated graft vs. host disease (TAGVHD)
• Post-transfusion purpura (PTP)
• Transfusion-transmitted infection (TTI)
Form
Adverse reaction forms are available at the NHSN Blood Safety Surveillance website.
Form Instructions
Adverse Reaction forms’ Table of Instructions are available at the NHSN Blood Safety Surveillance
website.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 9 of 31
February 2023
Adverse Reaction Case Classification Criteria Tables
Transfusion-associated circulatory overload (TACO)
Case Definition
Severity
Imputability
Definitive:
New onset or exacerbation
of 3 or more of the
following within 12 hours
of cessation of transfusion:
(At least 1 of the
following:)
•Evidence of acute or
worsening respiratory
distress (dyspnea,
tachypnoea, cyanosis and
decreased oxygen
saturation values in the
absence of other specific
causes) and/or
•Radiographic or clinical
evidence of acute or
worsening pulmonary
edema (crackles on lung
auscultation, orthopnea,
cough, a third heart sound
and pinkish frothy sputum
in severe cases); or both
AND
•Elevated brain natriuretic
peptide (BNP) or NT-pro
BNP relevant biomarker
•Evidence of
cardiovascular system
changes not explained by
underlying medical
condition (Elevated central
venous pressure, evidence
of left heart failure
including development of
tachycardia, hypertension,
widened pulse pressure,
jugular venous distension,
enlarged cardiac silhouette
and/or peripheral edema)
•Evidence of fluid overload
Probable:
N/A
Possible:
N/A
Non-severe:
Medical intervention (e.g. symptomatic
treatment) is required but lack of such would
not result in permanent damage or
impairment of a bodily function.
Severe:
Inpatient hospitalization or prolongation of
hospitalization is directly attributable to the
adverse reaction, persistent or significant
disability or incapacity of the patient occurs
as a result of the reaction, or a medical or
surgical intervention is necessary to
preclude permanent damage or impairment
of a body function.
Life-threatening:
Major intervention required following the
transfusion (e.g. vasopressors, intubation,
transfer to intensive care) to prevent death.
Death:
The recipient died as a result of the
adverse transfusion reaction. Death
should be used if death is possibly,
probably or definitely related to
transfusion. If the patient died of a cause
other than the transfusion, the severity of the
reaction should be graded as appropriate
given the clinical circumstances related to
the reaction.
Not Determined:
The severity of the adverse reaction is
unknown or not stated.
Definite:
No other explanations for circulatory
overload are possible.
Probable:
Transfusion is a likely contributor to
circulatory overload
AND EITHER
The patient received other fluids as
well
OR
The patient has a history of cardiac
insufficiency that could explain the
circulatory overload, but
transfusion is just as likely to have
caused the circulatory overload.
Possible:
The patient has a history of pre-
existing cardiac insufficiency that
most likely explains circulatory
overload.
OPTIONAL
Doubtful:
Evidence is clearly in favor of a
cause other than the transfusion, but
transfusion cannot be excluded.
Ruled Out:
There is conclusive evidence beyond
reasonable doubt of a cause other
than the transfusion.
Not Determined:
The relationship between the
adverse reaction and the transfusion
is unknown or not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 10 of 31
February 2023
Transfusion-related acute lung injury (TRALI)
Case Definition
Severity
Imputability
Definitive:
NO evidence of acute lung
injury (ALI) prior to
transfusion
AND
ALI onset during or within
6 hours of cessation of
transfusion
AND
Hypoxemia defined by any
of these methods:
• PaO2/FiO2 less than
or equal to 300 mm
Hg
• Oxygen saturation
less than 90% on
room air
• Other clinical
evidence
AND
Radiographic evidence of
bilateral infiltrates
AND
No evidence of left atrial
hypertension (i.e.,
circulatory overload)
Probable:
N/A
Possible:
N/A
Non-severe:
Medical intervention (e.g.
symptomatic treatment) is required
but lack of such would not result in
permanent damage or impairment of
a bodily function.
Severe:
Inpatient hospitalization or
prolongation of hospitalization is
directly attributable to the adverse
reaction, persistent or significant
disability or incapacity of the patient
occurs as a result of the reaction, or a
medical or surgical intervention is
necessary to preclude permanent
damage or impairment of a body
function.
Life-threatening:
Major intervention required following
the transfusion (e.g. vasopressors,
intubation, transfer to intensive care)
to prevent death.
Death:
The recipient died as a result of the
adverse transfusion reaction.
Death should be used if death is
possibly, probably or definitely
related to transfusion. If the patient
died of a cause other than the
transfusion, the severity of the
reaction should be graded as
appropriate given the clinical
circumstances related to the reaction.
Definite:
There are no alternative risk factors for ALI
present.
Probable:
N/A
Possible:
There is evidence of other causes for acute
lung injury such as:
Direct Lung Injury
• Aspiration
• Pneumonia
• Toxic inhalation
• Lung contusion
• Near drowning
Indirect Lung Injury
• Severe sepsis
• Shock
• Multiple trauma
• Burn injury
• Acute pancreatitis
• Cardiopulmonary bypass
• Drug overdose
OPTIONAL
Doubtful:
Evidence is clearly in favor of a cause other
than the transfusion, but transfusion cannot
be excluded.
Ruled Out:
There is conclusive evidence beyond
reasonable doubt of a cause other than the
transfusion.
Not Determined:
The severity of the adverse reaction
is unknown or not stated.
Not Determined:
The relationship between the adverse
reaction and the transfusion is unknown or
not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 11 of 31
February 2023
Transfusion-associated dyspnea (TAD)
Case Definition
Severity
Imputability
Definitive:
Acute respiratory
distress occurring within
24 hours of cessation of
transfusion
AND
Allergic reaction, TACO,
and TRALI definitions
are not applicable.
Probable:
N/A
Possible:
N/A
Non-severe:
Medical intervention (e.g. symptomatic treatment)
is required but lack of such would not result in
permanent damage or impairment of a bodily
function.
Severe:
Inpatient hospitalization or prolongation of
hospitalization is directly attributable to the
adverse reaction, persistent or significant disability
or incapacity of the patient occurs as a result of
the reaction, or a medical or surgical intervention
is necessary to preclude permanent damage or
impairment of a body function.
Life-threatening:
Major intervention required following the
transfusion (e.g. vasopressors, intubation, transfer
to intensive care) to prevent death.
Death:
The recipient died as a result of the adverse
transfusion reaction. Death should be used if
death is possibly, probably or definitely related
to transfusion. If the patient died of a cause other
than the transfusion, the severity of the reaction
should be graded as appropriate given the clinical
circumstances related to the reaction.
Not Determined:
The severity of the adverse reaction is unknown
or not stated.
Definite:
Patient has no other conditions
that could explain symptoms.
Probable:
There are other potential causes
that could explain symptoms, but
transfusion is the most likely
cause.
Possible:
Other present causes are most
likely, but transfusion cannot be
ruled out.
OPTIONAL
Doubtful:
Evidence is clearly in favor of a
cause other than the transfusion,
but transfusion cannot be
excluded.
Ruled Out:
There is conclusive evidence
beyond reasonable doubt of a
cause other than the transfusion.
Not Determined:
The relationship between the
adverse reaction and the
transfusion is unknown or not
stated.
NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 12 of 31
February 2023
Allergic reaction
Note: Minor allergic reactions (Non-severe) do not have to be reported to NHSN.
Case Definition
Severity
Imputability
Definitive:
2 or more of the following
occurring during or within 4
hours of cessation of
transfusion:
• Conjunctival edema
• Edema of lips, tongue and
uvula
• Erythema and edema of
the periorbital area
• Generalized flushing
• Hypotension
• Localized angioedema
• Maculopapular rash
• Pruritus (itching)
• Respiratory distress;
bronchospasm
• Urticaria (hives)
Probable:
ANY 1 of the following occurring
during or within 4 hours of
cessation of transfusion:
• Conjunctival edema
• Edema of lips, tongue and
uvula
• Erythema and edema of
the periorbital area
• Localized angioedema
• Maculopapular rash
• Pruritus (itching)
• Urticaria (hives)
Severe, Life-threatening, Death:
Involves respiratory and/or cardiovascular
systems and presents like an anaphylactic
reaction. There is anaphylaxis when, in
addition to mucocutaneous symptoms,
there are airway symptoms, hypotension,
or associated symptoms like hypotonia
and syncope. The respiratory signs and
symptoms may be laryngeal (tightness in
the throat, dysphagia, dysphonia,
hoarseness, stridor) or pulmonary
(dyspnea, cough, wheezing,
bronchospasm, hypoxemia). Such a
reaction usually occurs during or shortly
after cessation of transfusion.
Death should be used if death is
possibly, probably or definitely related
to transfusion. If the patient died of a
cause other than the transfusion, the
severity of the reaction should be graded
as appropriate given the clinical
circumstances related to the reaction.
Not Determined:
The severity of the adverse reaction is
unknown or not stated.
Definite:
Occurs during or within 2 hours of
cessation of transfusion
AND
No other evidence of
environmental, drug or dietary
risks.
Probable:
Occurs during or within 2 hours of
cessation of transfusion
AND
There are other potential causes
present that could explain
symptoms, but transfusion is the
most likely cause.
Possible:
Occurs 2 - 4 hours after cessation
of transfusion
OR
Other present causes are most
likely, but transfusion cannot be
ruled out.
OPTIONAL
OPTIONAL
OPTIONAL
Possible:
N/A
Non-severe:
There is no immediate risk to the life of
the patient, and the patient responds
quickly to symptomatic treatment.
Doubtful:
Evidence is clearly in favor of a
cause other than the transfusion,
but transfusion cannot be
excluded.
Ruled Out:
There is conclusive evidence
beyond reasonable doubt of a
cause other than the transfusion.
Not Determined:
The relationship between the
adverse reaction and the
transfusion is unknown or not
stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 13 of 31
February 2023
Hypotensive transfusion reaction
Case Definition
Severity
Imputability
Definitive:
All other adverse reactions
presenting with hypotension are
excluded
AND
Hypotension occurs during or
within 1 hour after cessation of
transfusion.
Non-severe:
The recipient required no
more than discontinuation of
transfusion and symptom
management and no long-
term morbidity resulted from
the reaction.
Severe:
Inpatient hospitalization or
prolongation of
hospitalization is directly
attributable to hypotension,
or hypotension led directly to
long-term morbidity (e.g.,
brain damage)
AND
Vasopressors were not
required.
Life-threatening:
The recipient required
vasopressors.
Death:
The recipient died as a
result of the adverse
transfusion reaction.
Death should be used if
death is possibly, probably
or definitely related to
transfusion. If the patient
died of a cause other than
the transfusion, the severity
of the reaction should be
graded as appropriate given
the clinical circumstances
related to the reaction.
Not Determined:
The severity of the adverse
reaction is unknown or not
stated.
Definite:
Occurs less than 15 minutes after the start of
the transfusion
AND
Responds rapidly (i.e., within 10 minutes) to
cessation of transfusion and supportive
treatment
AND
The patient has no other conditions that could
explain hypotension.
Probable:
Onset is between 15 minutes after start and 1
hour after cessation of transfusion
OR
The patient does not respond rapidly to
cessation of transfusion and supportive
treatment
OR
There are other potential causes present that
could explain hypotension, but transfusion is
the most likely cause.
Possible:
Other conditions that could readily explain
hypotension are present.
• Adults (18 years and
older):
Drop in systolic BP of
greater than or equal to 30
mmHg and systolic BP
less than or equal to 80
mmHg.
• Infants, children and
adolescents (1 year to
less than 18 years old):
Greater than 25% drop in
systolic BP from baseline
(e.g., drop in systolic BP of
120mmHg to below
90mmHg).
• Neonates and small
infants (less than 1 year
old OR any age and less
than 12 kg body weight):
Greater than 25% drop in
baseline value using
whichever measurement is
being recorded (e.g., mean
BP).
Probable:
N/A
OPTIONAL
OPTIONAL
Possible:
Hypotension occurs, does not
meet the criteria above. Other,
more specific reaction definitions
do not apply.
Doubtful:
Evidence is clearly in favor of a cause other
than the transfusion, but transfusion cannot be
excluded.
Ruled Out:
There is conclusive evidence beyond
reasonable doubt of a cause other than the
transfusion.
Not Determined:
The relationship between the adverse reaction
and the transfusion is unknown or not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 14 of 31
February 2023
Febrile non-hemolytic transfusion reaction (FNHTR)
Note: Reactions may be classified as FNHTRs in the absence of fever if chills or rigors occur.
Case Definition
Severity
Imputability
Definitive:
Occurs during or within 4
hours of cessation of
transfusion
AND EITHER
Fever (greater than or
equal to 38°C/100.4°F
oral and a change of at
least 1°C/1.8°F) from pre-
transfusion value
OR
Chills/rigors are present.
Probable:
N/A
Non-severe:
Medical intervention (e.g. symptomatic
treatment) is required but lack of such would
not result in permanent damage or impairment
of a bodily function.
Severe:
Inpatient hospitalization or prolongation of
hospitalization is directly attributable to the
adverse reaction, persistent or significant
disability or incapacity of the patient occurs as
a result of the reaction, or a medical or surgical
intervention is necessary to preclude
permanent damage or impairment of a body
function.
Life-threatening:
Major intervention required following the
transfusion (e.g. vasopressors, intubation,
transfer to intensive care) to prevent death.
Death:
The recipient died as a result of the adverse
transfusion reaction. Death should be used if
death is possibly, probably or definitely
related to transfusion. If the patient died of a
cause other than the transfusion, the severity
of the reaction should be graded as
appropriate given the clinical circumstances
related to the reaction.
Not Determined:
The severity of the adverse reaction is
unknown or not stated.
Definite:
Patient has no other conditions
that could explain
signs/symptoms.
Probable:
There are other potential causes
present that could explain
signs/symptoms, but transfusion
is the most likely cause.
Possible:
Other present causes are most
likely, but transfusion cannot be
ruled out.
OPTIONAL
OPTIONAL
Possible:
FNHTR is suspected, but
reported symptoms and/or
available information are
not sufficient to meet the
criteria defined above.
Other, more specific
adverse reaction definitions
do not apply.
Doubtful:
Evidence is clearly in favor of a
cause other than the transfusion,
but transfusion cannot be
excluded.
Ruled Out:
There is conclusive evidence
beyond reasonable doubt of a
cause other than the transfusion.
Not Determined:
The relationship between the
adverse reaction and the
transfusion is unknown or not
stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 15 of 31
February 2023
Acute hemolytic transfusion reaction (AHTR)
Note: Report hemolytic reactions resulting from immune or non-immune causes, including when the
recipient is intentionally transfused with incompatible blood components.
Case Definition
Severity
Imputability
Definitive:
Occurs during, or within 24 hours of cessation of
transfusion with new onset of ANY of the following
signs/symptoms:
• Back/flank pain
• Chills/rigors
• Disseminated intravascular coagulation (DIC)
• Epistaxis
• Fever
• Hematuria (gross visual hemolysis)
• Hypotension
• Oliguria/anuria
• Pain and/or oozing at IV site
• Renal failure
AND
2 or more of the following:
• Decreased fibrinogen
• Decreased haptoglobin
• Elevated bilirubin
• Elevated LDH
• Hemoglobinemia
• Hemoglobinuria
• Plasma discoloration c/w hemolysis
• Spherocytes on blood film
AND EITHER
(IMMUNE-MEDIATED)
Positive direct antiglobulin test (DAT) for anti-IgG or
anti-C3
AND
Positive elution test with alloantibody present on the
transfused red blood cells
OR
(NON-IMMUNE MEDIATED)
Serologic testing is negative, and physical cause (e.g.,
thermal, osmotic, mechanical, chemical) is confirmed.
Probable:
Meets signs and symptoms criteria for acute hemolysis
AND EITHER
(IMMUNE MEDIATED)
Physical cause is excluded but serologic evidence is not
sufficient to meet definitive criteria
OR
(NON-IMMUNE MEDIATED)
Physical cause is suspected and serologic testing is
negative.
Non-severe:
Medical intervention (e.g.
symptomatic treatment) is
required but lack of such would
not result in permanent
damage or impairment of a
bodily function.
Severe:
Inpatient hospitalization or
prolongation of hospitalization
is directly attributable to the
adverse reaction, persistent or
significant disability or
incapacity of the patient occurs
as a result of the reaction, or a
medical or surgical intervention
is necessary to preclude
permanent damage or
impairment of a body function.
Life-threatening:
Major intervention required
following the transfusion (e.g.
vasopressors, intubation,
transfer to intensive care) to
prevent death.
Death:
The recipient died as a result
of the adverse transfusion
reaction. Death should be
used if death is possibly,
probably or definitely related
to transfusion. If the patient
died of a cause other than the
transfusion, the severity of the
reaction should be graded as
appropriate given the clinical
circumstances related to the
reaction.
Not Determined:
The severity of the adverse
reaction is unknown or not
stated.
Definite:
ABO or other allotypic
RBC antigen
incompatibility is known
OR
Only transfusion-related
(i.e., immune or non-
immune) cause of acute
hemolysis is present.
Probable:
There are other
potential causes
present that could
explain acute
hemolysis, but
transfusion is the most
likely cause.
Possible:
Other causes of acute
hemolysis are more
likely, but transfusion
cannot be ruled out.
OPTIONAL
Doubtful:
Evidence is clearly in
favor of a cause other
than the transfusion, but
transfusion cannot be
excluded.
Ruled Out:
There is conclusive
evidence beyond
reasonable doubt of a
cause other than the
transfusion.
Not Determined:
The relationship
between the adverse
reaction and the
transfusion is unknown
or not stated.
OPTIONAL
Possible:
AHTR is suspected within 24 hours of cessation of
transfusion, but symptoms, test results, and/or information
are not sufficient to meet the criteria defined above. Other,
more specific adverse definitions do not apply.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 16 of 31
February 2023
Delayed hemolytic transfusion reaction (DHTR)
Note: Report all hemolytic reactions, including when the recipient is intentionally transfused with
incompatible blood components.
Case Definition
Severity
Imputability
Definitive:
Positive direct antiglobulin test (DAT)
for antibodies developed between 24
hours and 28 days after cessation of
transfusion
AND EITHER
Positive elution test with
alloantibody present on the
transfused red blood cells
OR
Newly-identified red blood cell
alloantibody in recipient serum
AND EITHER
Inadequate rise of post-transfusion
hemoglobin level or rapid fall in
hemoglobin back to pre-transfusion
levels
OR
Otherwise unexplained appearance
of spherocytes.
Probable:
Newly-identified red blood cell
alloantibody demonstrated between
24 hours and 28 days after cessation
of transfusion
BUT
Incomplete laboratory evidence to
meet definitive case definition criteria.
NOTE: Patient may be asymptomatic
or have symptoms that are similar to
but milder than AHTR; symptoms are
not required to meet case definition
criteria.
Non-severe:
Medical intervention (e.g.
symptomatic treatment) is
required but lack of such would
not result in permanent damage
or impairment of a bodily
function.
Severe:
Inpatient hospitalization or
prolongation of hospitalization is
directly attributable to the
adverse reaction, persistent or
significant disability or incapacity
of the patient occurs as a result
of the reaction, or a medical or
surgical intervention is necessary
to preclude permanent damage
or impairment of a body function.
Life-threatening:
Major intervention required
following the transfusion (e.g.
vasopressors, intubation, transfer
to intensive care) to prevent
death.
Death:
The recipient died as a result of
the adverse transfusion
reaction. Death should be used
if death is possibly, probably or
definitely related to transfusion.
If the patient died of a cause
other than the transfusion, the
severity of the reaction should be
graded as appropriate given the
clinical circumstances related to
the reaction.
Not Determined:
The severity of the adverse
reaction is unknown or not
stated.
Definite:
No other explanation for symptoms
or newly-identified antibody is
present.
Probable:
An alternate explanation for
symptoms or newly-identified
antibody is present, but transfusion is
the most likely cause.
Possible:
Other explanations for symptoms or
newly-identified antibody are more
likely, but transfusion cannot be ruled
out.
OPTIONAL
OPTIONAL
Possible:
DHTR is suspected, but reported
symptoms, test results, and/or
available information are not sufficient
to meet the criteria defined above.
Other, more specific adverse reaction
definitions do not apply.
Doubtful:
Evidence is clearly in favor of a
cause other than the transfusion, but
transfusion cannot be excluded.
Ruled Out:
There is conclusive evidence beyond
reasonable doubt of a cause other
than the transfusion.
Not Determined:
The relationship between the
adverse reaction and the transfusion
is unknown or not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 17 of 31
February 2023
Delayed serologic transfusion reaction (DSTR)
Note: Delayed serologic reactions should only be reported for patients transfused by your facility.
Case Definition
Severity
Imputability
Definitive:
Absence of clinical signs
of hemolysis
AND
Demonstration of new,
clinically-significant
antibodies against red
blood cells
BY EITHER
Positive direct
antiglobulin test (DAT)
OR
Positive antibody
screen with newly
identified RBC
alloantibody.
Not Determined:
Since this is by definition a
reaction with no clinical
symptoms, severity of the
reaction cannot be
graded.
Definite:
New alloantibody is identified between 24 hours and 28
days after cessation of transfusion
AND
Transfusion performed by your facility is the only
possible cause for seroconversion.
Probable:
New alloantibody is identified between 24 hours and 28
days after cessation of transfusion
AND
The patient has other exposures (e.g. transfusion by
another facility or pregnancy) that could explain
seroconversion, but transfusion by your facility is the
most likely cause.
Probable:
N/A
Possible:
N/A
Possible:
New alloantibody is identified between 24 hours and 28
days after cessation of transfusion
AND
The patient was transfused by your facility, but other
exposures are present that most likely explain
seroconversion.
OPTIONAL
Doubtful:
Evidence is clearly in favor of a cause other than the
transfusion, but transfusion cannot be excluded.
Ruled Out:
There is conclusive evidence beyond reasonable doubt
of a cause other than the transfusion.
Not Determined:
The relationship between the adverse reaction and the
transfusion is unknown or not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 18 of 31
February 2023
Transfusion-associated graft vs. host disease (TAGVHD)
Case Definition
Severity
Imputability
Definitive:
A clinical syndrome occurring from
2 days to 6 weeks after cessation of
transfusion characterized by:
• Characteristic rash:
erythematous, maculopapular
eruption centrally that spreads
to extremities and may, in
severe cases, progress to
generalized erythroderma and
hemorrhagic bullous
formation.
• Diarrhea
• Fever
• Hepatomegaly
• Liver dysfunction (i.e.,
elevated ALT, AST, Alkaline
phosphatase, and bilirubin)
• Marrow aplasia
• Pancytopenia
AND
Characteristic histological
appearance of skin or liver biopsy.
Probable:
Meets definitive criteria
EXCEPT
Biopsy negative or not done.
Non-severe:
N/A
Severe:
Patient had marked
symptoms and responded to
treatment.
Life-threatening:
Patient had severe symptoms
and required life-saving
treatment (e.g.,
immunosuppression).
Death:
The recipient died as a result
of the adverse transfusion
reaction. Death should be
used if death is possibly,
probably or definitely
related to transfusion. If the
patient died of a cause other
than the transfusion, the
severity of the reaction should
be graded as appropriate
given the clinical
circumstances related to the
reaction.
Definite:
WBC chimerism present in the absence of
alternative diagnoses.
Probable:
WBC chimerism present
BUT
Other potential causes are present (e.g.,
stem cell transplantation).
Possible:
WBC chimerism not present or not done
OR
Alternative explanations are more likely
(e.g., solid organ transplantation).
OPTIONAL
Doubtful:
Evidence is clearly in favor of a cause
other than the transfusion, but transfusion
cannot be excluded.
Ruled Out:
There is conclusive evidence beyond
reasonable doubt of a cause other than
the transfusion.
Possible:
N/A
Not Determined:
The severity of the adverse
reaction is unknown or not
stated.
Not Determined:
The relationship between the adverse
reaction and the transfusion is unknown or
not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 19 of 31
February 2023
Post transfusion purpura (PTP)
Case Definition
Severity
Imputability
Definitive:
Alloantibodies in the patient
directed against HPA or other
platelet specific antigen detected
at or after development of
thrombocytopenia
AND
Thrombocytopenia (i.e., decrease
in platelets to less than 20% of
pre-transfusion count).
Probable:
Alloantibodies in the patient
directed against HPA or other
platelet specific antigen detected
at or after development of
thrombocytopenia.
AND
Decrease in platelets to levels
between 20% and 80% of pre-
transfusion count.
Non-severe:
Medical intervention (e.g. symptomatic
treatment) is required but lack of such
would not result in permanent damage
or impairment of a bodily function.
Severe:
Inpatient hospitalization or
prolongation of hospitalization is
directly attributable to the adverse
reaction, persistent or significant
disability or incapacity of the patient
occurs as a result of the reaction, or a
medical or surgical intervention is
necessary to preclude permanent
damage or impairment of a body
function.
Life-threatening:
Major intervention required following
the transfusion (e.g. vasopressors,
intubation, transfer to intensive care) to
prevent death.
Death:
The recipient died as a result of the
adverse transfusion reaction. Death
should be used if death is possibly,
probably or definitely related to
transfusion. If the patient died of a
cause other than the transfusion, the
severity of the reaction should be
graded as appropriate given the
clinical circumstances related to the
reaction.
Not Determined:
The severity of the adverse reaction is
unknown or not stated.
Definite:
Occurs 5-12 days post-transfusion
AND
Patient has no other conditions to
explain thrombocytopenia.
Probable:
Occurs less than 5 or more than
12 days post-transfusion
OR
There are other potential causes
present that could explain
thrombocytopenia, but transfusion
is the most likely cause.
Possible:
Alternate explanations for
thrombocytopenia are more likely,
but transfusion cannot be ruled
out.
OPTIONAL
OPTIONAL
Possible:
PTP is suspected, but laboratory
findings and/or information are not
sufficient to meet defined criteria
above. For example, the patient
has a drop in platelet count to less
than 80% of pre-transfusion count
but HPA antibodies were not
tested or were negative. Other,
more specific adverse reaction
definitions do not apply.
Doubtful:
Evidence is clearly in favor of a
cause other than the transfusion,
but transfusion cannot be
excluded.
Ruled Out:
There is conclusive evidence
beyond reasonable doubt of a
cause other than the transfusion.
Not Determined:
The relationship between the
adverse reaction and the
transfusion is unknown or not
stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 20 of 31
February 2023
Transfusion-transmitted infection (TTI)
Case Definition
Severity
Imputability
Definitive:
Laboratory
evidence of a
pathogen in the
transfusion
recipient.
Probable:
N/A
Non-severe:
Medical intervention
(e.g. symptomatic
treatment) is required
but lack of such
would not result in
permanent damage
or impairment of a
bodily function.
Severe:
Inpatient
hospitalization or
prolongation of
hospitalization is
directly attributable to
the adverse reaction,
persistent or
significant disability or
incapacity of the
patient occurs as a
result of the reaction,
or a medical or
surgical intervention
is necessary to
preclude permanent
damage or
impairment of a body
function.
Definite:
ONE or more of the following:
• Evidence of the pathogen in the transfused component
• Evidence of the pathogen in the donor at the time of donation
• Evidence of the pathogen in an additional component from the same donation
• Evidence of the pathogen in an additional recipient of a component from the
same donation
AND
No other potential exposures to the pathogen could be identified in the recipient.
AND EITHER
Evidence that the recipient was not infected with the pathogen prior to transfusion
OR
Evidence that the identified pathogen strains are related by molecular or extended
phenotypic comparison testing with statistical confidence (p<0.05).
Probable:
ONE or more of the following:
• Evidence of the pathogen in the transfused component
• Evidence of the pathogen in the donor at the time of donation
• Evidence of the pathogen in an additional component from the same donation
• Evidence of the pathogen in an additional recipient of a component from the
same donation.
AND EITHER:
Evidence that the recipient was not infected with this pathogen prior to transfusion
OR
No other potential exposures to the pathogen could be identified in the recipient.
Possible:
Case fails to meet definite, probable, doubtful, or ruled out imputability criteria.
OPTIONAL
OPTIONAL
Possible:
Temporally
associated
unexplained
clinical illness
consistent with
infection, but no
pathogen is
detected in the
recipient. Other,
more specific
adverse reactions
are ruled out.
Note: Possible
cases cannot meet
the definite or
probable
imputability criteria.
Life-threatening:
Major intervention
required following the
transfusion (e.g.
vasopressors,
intubation, transfer to
intensive care) to
prevent death.
Death:
The recipient died as
a result of the
adverse transfusion
reaction.
Not Determined:
The severity of the
adverse reaction is
unknown or not
stated.
Doubtful:
Laboratory evidence that the recipient was infected with this pathogen prior to
transfusion
OR
Evidence is clearly in favor of a cause other than transfusion, but transfusion cannot
be excluded.
Ruled Out:
ALL of the following (where applicable):
• Evidence that the transfused component was negative for this pathogen at the
time of transfusion
• Evidence that the donor was negative for this pathogen at the time of donation
• Evidence that additional components from the same donation were negative
for this pathogen
OR
There is conclusive evidence beyond reasonable doubt of a cause other than the
transfusion.
Not Determined:
The relationship between the adverse reaction and the transfusion is unknown or
not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 21 of 31
February 2023
Transfusion-transmitted infection (TTI)
(continued)
Pathogens of well-documented importance in blood safety.
These pathogens have public health significance for hemovigilance, are well-documented blood stream
pathogens, and/or are routinely screened for in blood donors. A full list of potentially infectious organisms is
available in the drop-down pathogen list in NHSN.
Bacterial
Viral
Parasitic
Other
Enterobacter cloacae
Escherichia coli
Klebsiella oxytoca
Klebsiella pneumoniae
Pseudomonas aeruginosa
Serratia marcescens
Staphylococcus aureus
Staphylococcus
epidermidis
Staphylococcus
lugdunensis
Syphilis (Treponema
pallidum)
Yersinia enterocolitica
Cytomegalovirus (CMV)
Enterovirus spp.
Epstein Barr (EBV)
Hepatitis A
Hepatitis B
Hepatitis C
Human Immunodeficiency Virus 1
(HIV-1)
Human Immunodeficiency Virus 2
(HIV-2)
Human Parvovirus B-19
Human T-Cell Lymphotropic
Virus-1 (HTLV-1)
Human T-Cell Lymphotropic
Virus-2 (HTLV-2)
West Nile Virus (WNV)
Zika Virus (ZIKAVI)
Babesiosis (Babesia spp.)
Chagas disease
(Trypanosoma cruzi)
Malaria (Plasmodium spp.)
Creutzfeldt-
Jakob Disease,
Variant (vCJD)
Investigation triggers for potential transfusion-transmitted infections:
1. Identification by testing (e.g., gram stain, other smear/staining, culture, or other method) of a
bacterial, mycobacterial, or fungal pathogen in a recipient within the time period from exposure
(i.e., transfusion) to onset of infection appropriate for the suspected pathogen.
2. Identification of an unexpected virus in the transfusion recipient by testing (e.g., culture, direct
fluorescent antibody, or polymerase chain reaction) within the time period from exposure (i.e.,
transfusion) to onset of infection appropriate for the suspected virus.
3. Identification of an unexpected parasite in the recipient by testing (e.g., blood smear,
histopathology, serologic testing, or polymerase chain reaction) within the time period from
exposure (i.e., transfusion) to onset of infection appropriate for the suspected parasite.
4. Any of the above laboratory findings in the recipient unit upon residual testing.
5. Unexplained clinical events occurring after transfusion that are consistent with transfusion-
transmitted infection, such as:
a. Encephalitis, meningitis, or other unexplained central nervous system abnormalities.
b. Sepsis with or without multi-organ system dysfunction.
c. Hemolytic anemia and/or fever (e.g., in cases of transfusion-associated babesiosis or malaria).
d. Recipient death.
6. For pathogens routinely screened in the blood donor, any infection in the recipient occurring within
6 months after transfusion if:
a. The index donation testing was negative but
b. The donor was subsequently found to be infected, and
c. The recipient had no pre-transfusion history of the same infection.
NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 22 of 31
February 2023
Other or Unknown
Other: Use this option if the recipient experienced an adverse reaction that is not defined in the Hemovigilance
Module surveillance protocol (e.g., transfusion-associated acute gut injury (TRAGI), transfusion-associated
immunomodulation (TRIM), iron overload, microchimerism, hyperkalemia, thrombosis).
Unknown: Use this category if the patient experienced transfusion-related symptoms, but the medical event that
caused those symptoms could not be classified.
Note: Reporting ‘Other’ and ‘Unknown’ reactions is not required by CDC.
REPORTING OPTIONAL
Case Definition
Severity
Imputability
Not Applicable:
CDC does not
specifically define the
‘Other’ or ‘Unknown’
adverse reaction
categories, therefore
the case definition
criteria may only be
reported as N/A.
Non-severe:
Medical intervention (e.g. symptomatic treatment)
is required but lack of such would not result in
permanent damage or impairment of a bodily
function.
Severe:
Inpatient hospitalization or prolongation of
hospitalization is directly attributable to the
adverse reaction, persistent or significant disability
or incapacity of the patient occurs as a result of
the reaction, or a medical or surgical intervention
is necessary to preclude permanent damage or
impairment of a body function.
Definite:
Conclusive evidence exists that the
adverse reaction can be attributed to
the transfusion.
Probable:
Evidence is clearly in favor of
attributing the adverse reaction to
the transfusion.
Possible:
Evidence is indeterminate for
attributing the adverse reaction to
the transfusion or an alternate
cause.
Life-threatening:
Major intervention required following the
transfusion (e.g. vasopressors, intubation, transfer
to intensive care) to prevent death.
Doubtful:
Evidence is clearly in favor of a
cause other than the transfusion, but
transfusion cannot be excluded.
Death:
The recipient died as a result of the adverse
transfusion reaction. Death should be used if
death is possibly, probably or definitely related
to transfusion. If the patient died of a cause other
than the transfusion, the severity of the reaction
should be graded as appropriate given the clinical
circumstances related to the reaction.
Ruled Out:
There is conclusive evidence
beyond reasonable doubt of a cause
other than the transfusion.
Not Determined:
The severity of the adverse reaction is unknown
or not stated.
Not Determined:
The relationship between the
adverse reaction and the transfusion
is unknown or not stated.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 23 of 31
February 2023
Adverse Reaction Glossary
Antibodies often associated with AHTR, DHTR, DSTR:
Anti-A
Anti-B
Anti-A,B
Anti-C
Anti-c
Anti-D
Anti-E
Anti-e
Anti-Fy
a
Anti-Fy
b
Anti-Jk
a
Anti-Jk
b
Anti-K
Anti-k
Anti-M
Anti-S
Other
Bronchospasm (wheezing): A contraction of smooth muscle in the walls of the bronchi and
bronchioles, causing acute narrowing and obstruction of the respiratory airway. This constriction can
result in a rasp or whistling sound while breathing.
Chills/rigors: A feeling of cold with shivering or shaking and pallor.
Disseminated intravascular coagulation (DIC): Bleeding disorder characterized by reduction in the
factors involved in blood clotting due to their use in widespread clotting within the vessels. The
intravascular clotting ultimately produces hemorrhage because of rapid consumption of clotting factors.
Edema: Swelling of soft tissues as a result of excessive fluid accumulation.
Epistaxis: Bleeding from the nose.
Fever: For the purposes of hemovigilance, greater than or equal to 38°C/100.4°F oral and a change of
at least 1°C/1.8°F from pre-transfusion value.
Hematuria: Presence of blood or red blood cells in the urine.
Hemoglobinemia: The presence of free hemoglobin in the blood plasma.
Hemoglobinuria: Presence of free hemoglobin in the urine.
Hypoxemia: Abnormal deficiency in the concentration of oxygen in arterial blood. PaO2 / FiO2 less
than or equal to 300 mm Hg OR oxygen saturation is less than 90% on room air.
Jaundice: New onset or worsening of yellow discoloration (icterus) of the skin or sclera (scleral icterus)
secondary to an increased level of bilirubin.
Oliguria: New onset of decreased urinary output (less than 500cc output per 24 hours).
Other rash: Non-urticarial skin rash.
Pruritus: Itching.
Shock: A drop in blood pressure accompanied by a drop in cardiac output including rapid heart rate
(increase to 100 beats per minute or more), rapid breathing, cutaneous vasoconstriction, pallor,
sweating, decreased or scanty urine production, agitation and/or loss of consciousness that required
fluid resuscitation, with or without inotropic support.
Shortness of breath (dyspnea): New onset or significant worsening of shortness of breath; or a
significant increase in respiratory rate (with or without hypoxemia).
Urticaria (hives): Raised wheals on the skin.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 24 of 31
February 2023
Section 4. Hemovigilance Module Incidents
Required Reporting
All incidents (i.e., accidents or errors) that are associated with a reported adverse reaction must be
reported to NHSN using a detailed Incident form (CDC 57.305). If multiple incidents occur in association
with an adverse reaction then report all. Incidents may occur before (e.g., wrong product released) or
after (e.g., failure to report adverse reaction to blood bank) an adverse reaction. Each reaction must be
reported using the detailed incident form; the incident result must be coded as ‘Product transfused,
reaction’ to enter the associated patient identifier on the form. After the incident record is entered, the
adverse reaction record must be linked to the incident record in the NHSN web application.
Incident Classification
Use the incident codes provided at the end of this section to classify incidents. If there is uncertainty then
please contact NHSN User Support.
Optional Reporting
Any incident may be optionally reported to NHSN using the detailed Incident form (57.305) or the
Monthly Incident Summary form (57.302). Approved deviations from standard operating procedure are
not considered incidents because they did not occur by accident or in error. However, approved
deviations may be optionally reported for a facility’s use.
Form
CDC 57.305 Hemovigilance Module Incident
Form Instructions
CDC 57.305 Hemovigilance Module Incident Table of Instructions
Summary Form (Optional)
CDC 57.302 Hemovigilance Module Monthly Incident Summary
Summary Form Instructions (Optional)
CDC 57.302 Hemovigilance Module Monthly Incident Summary Table of Instructions

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 25 of 31
February 2023
Incident Codes
Note: Incident codes are based on MERS TM (US) and TESS (Canada) incident classification schemes.
Product Check-In
(Transfusion Service)
Events that occur during the shipment and receipt of
products into the transfusion service from the
supplier, another hospital site, satellite storage, or
clinical area.
PC 00 Detail not specified
PC 01 Data entry incomplete/incorrect/not performed
PC 02 Shipment incomplete/incorrect
PC 03 Products and paperwork do not match
PC 04 Shipped/transported under inappropriate
conditions
PC 05 Inappropriate return to inventory
PC 06 Product confirmation incorrect/not performed
PC 07 Administrative check not incorrect/not
performed (record review/audit)
PC 08 Product label incorrect/missing
Product Storage
(Transfusion Service)
Events that occur during product storage by the
transfusion service.
US 00 Detail not specified
US 01 Incorrect storage conditions
US 03 Inappropriate monitoring of storage device
US 04 Unit stored on incorrect shelf (e.g.,
ABO/autologous s/directed)
US 05 Incorrect storage location
Inventory Management
(Transfusion Service)
Events that involve quality management of the blood
product inventory.
IM 00 Detail not specified
IM 01 Inventory audit incorrect/not performed
IM 02 Product status incorrectly/not updated online
(e.g., available/discarded)
IM 03 Supplier recall/traceback not appropriately
addressed/not performed
IM 04 Product order incorrectly/not submitted to
supplier
IM 05 Outdated product in available inventory
IM 06 Recalled/quarantined product in available
inventory
Product/Test Request
(Clinical Service)
Events that occur when the clinical service orders
patient tests or blood products for transfusion.
PR 00 Detail not specified
PR 01 Order for wrong patient
PR 02 Order incompletely/incorrectly ordered (online
order entry)
PR 03 Special processing needs not indicated (e.g.,
CMV negative, autologous)
PR 04 Order not done
PR 05 Inappropriate/unnecessary (intended) test
ordered
PR 06 Inappropriate/unnecessary (intended) blood
product ordered
PR 07 Incorrect (unintended) test ordered
PR 08 Incorrect (unintended) blood product ordered
Product/Test Order Entry
(Transfusion Service)
Events that occur when the transfusion service
receives a patient order. This process may be
excluded if clinical service uses online ordering.
OE 00 Detail not specified
OE 01 Order entered for wrong patient
OE 02 Order incompletely/incorrectly entered
online OE 03 Special processing needs not entered
(e.g.,
CMV-, autologous)
OE 04 Order entry not done
OE 05 Inappropriate/unnecessary (intended) test
order entered
OE 06 Inappropriate/unnecessary (intended) blood
product order entered
OE 07 Incorrect (unintended) test ordered
OE 08 Incorrect (unintended) blood product ordered
Sample Collection
(Service collecting the samples)
Events that occur during patient sample collection.
SC 00 Detail not specified
SC 01 Sample labeled with incorrect patient name
SC 02 Not labeled
SC 03 Wrong patient collected
SC 04 Collected in wrong tube type
SC 05 Sample QNS
SC 06 Sample hemolyzed
SC 07 Label incomplete/illegible/incorrect (other than
patient name)
SC 08 Sample collected in error
SC 09 Requisition arrived without samples
SC 10 Wristband incorrect/not available
SC 11 Sample contaminated

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 26 of 31
February 2023
Incident Codes
(continued)
Note: Incident codes are based on MERS TM (US) and TESS (Canada) incident classification schemes.
Sample Handling
(Service collecting the samples)
Events that occur when a patient sample is sent for
testing.
SH 00 Detail not specified
SH 01 Sample sent without requisition
SH 02 Requisition and sample label don’t match
SH 03 Patient ID incomplete/illegible on requisition
SH 04 No Patient ID on requisition
SH 05 No phlebotomist/witness identification
SH 06 Sample sent with incorrect requisition type
SH 07 Patient information (other than ID)
missing/incorrect on requisition
SH 08 Requisition sent without sample
SH 09 Data entry incorrect/incomplete/not performed
SH 10 Sample transport issue (e.g., sample
broken/inappropriate conditions)
SH 11 Duplicate sample sent in error
Sample Receipt
(Transfusion Service)
Events that occur when a sample is received by the
transfusion service.
SR 00 Detail not specified
SR 01 Sample accepted in error
SR 02 Historical review incorrect/not performed
SR 03 Demographic review/ data entry incorrect/not
performed
SR 04 Sample incorrectly accessioned
Sample Testing
(Transfusion Service)
Events that occur during patient sample testing by
the transfusion service.
ST 00 Detail not specified
ST 01 Data entry incomplete/incorrect/not performed
ST 02 Appropriate sample checks
incomplete/incorrect/not performed
ST 03 Computer warning overridden in error or
outside SOP
ST 05 Sample test tube incorrectly accessioned
ST 07 Sample test tubes mixed up
ST 09 Sample test tube mislabeled (wrong patient
identifiers)
ST 10 Equipment problem/failure/not properly QC’d
ST 12 Sample testing not performed
ST 13 Incorrect sample testing method chosen
ST 14 Sample testing performed incorrectly
ST 15 Sample test result misinterpreted
Sample Testing (continued)
ST 16 Reagents used were
incorrect/inappropriate/expired/not properly
QC’d
ST 17 ABO/Rh error caught on final check
ST 18 Current/historical ABO/Rh mismatch
ST 19 Additional testing not performed
ST 20 Confirmatory check incorrect/not performed (at
time work performed)
ST 21 Administrative check incorrect/not performed
(record review/audit)
ST 22 Sample storage incorrect/inappropriate
Product Manipulation/Processing/Testing
(Transfusion Service)
Events that occur while testing, manipulating (e.g.,
pooling, washing, aliquoting, irradiating), processing,
or labeling blood products.
UM 00 Detail not specified
UM 01 Data entry incomplete/incorrect/not performed
UM 02 Record review incomplete/incorrect/not
performed
UM 03 Incorrect product (type) selected
UM 04 Incorrect product (patient) selected
UM 05 Product labeled incorrectly (new/updated)
UM 06 Computer warning overridden in error or
outside SOP
UM 07 Special processing needs not checked
UM 08 Special processing needs misunderstood or
misinterpreted
UM 09 Special processing needs performed
incorrectly
UM 10 Special processing needs not performed
UM 11 Equipment problem/failure/not properly QC’d
UM 12 Reagents used were
incorrect/inappropriate/expired/not properly
QC’d
UM 13 Confirmatory check incorrect/not performed (at
time work performed)
UM 14 Administrative check incorrect/not performed
(record review/audit)
No Blood
NB 01 Inventory less than usual par level due to
supplier unable to meet usual steady demand
NB 02 Demand for blood product exceeding usual par
inventory level
NB 03 I
ncompatible/inappropriate units issued due to
inventory constraints when demand for blood
product exceeds usual par inventory levels.
NB 04 Suboptimal dose (less than optimal quantity)
transfusion or no transfusion due to inventory
constraints when demand for blood product
exceeds usual par inventory levels.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 27 of 31
February 2023
Incident Codes
(continued)
Note: Incident codes are based on MERS TM (US) and TESS (Canada) incident classification schemes.
Request for Pick-up
(Clinical Service)
Events that occur when the clinical service requests
pick-up of a blood product from the transfusion
service.
RP 00 Detail not specified
RP 01 Request for pick-up on wrong patient
RP 02 Incorrect product requested for pick-up
RP 03 Product requested prior to obtaining consent
RP 04 Product requested for pick-up, but patient not
available
RP 05 Product requested for pick-up, but IV not ready
RP 06 Request for pick-up incomplete (e.g., patient
ID/product type missing)
RP 07 Pick-up slip did not match patient information
on product
Product Issue
(Transfusion Service)
Events that occur when the transfusion service
issues blood product to the clinical service.
UI 00 Detail not specified
UI 01 Data entry incomplete/incorrect/not performed
UI 02 Record review incomplete/incorrect/not
performed
UI 03 Product issued for wrong patient
UI 04 Product issued out of order
UI 05 Product issue delayed
UI 06 LIS warning overridden in error or outside SOP
UI 07 Computer issue not completed
UI 08 Issued visibly defective product (e.g.,
clots/aggregates/particulate matter)
UI 09 Not/incorrect checking of unit and/or patient
information
UI 10 Product transport issues (e.g., delayed) by
transfusion service
UI 11 Unit delivered to incorrect location by
transfusion service
UI 12 Product transport issue (from transfusion
service to clinical area)
UI 18 Wrong product issued for intended patient (e.g.,
incompatible)
UI 19 Inappropriate product issued for patient (e.g.,
not irradiated, CMV+)
UI 20 Confirmatory check incorrect/not performed (at
time work performed)
UI 21 Administrative check incorrect/not performed
(record review/audit)
UI 22 Issue approval not obtained/documented
UI 23 Receipt verification not performed (pneumatic
tube issue)
Satellite Storage
(Clinical Service)
Events that occur while product is stored and
handled by the clinical service.
CS 00 Detail not specified
CS 01 Incorrect storage conditions of product in
clinical area
CS 02 Incorrect storage location in the clinical area
CS 03 Labeling issue (by clinical staff)
CS 04 Floor/clinic did not check for existing products
in their area
CS 05 Product transport issues (to or between clinical
areas)
CS 06 Monitoring of satellite storage
incorrect/incomplete/not performed
CS 07 Storage tracking/documentation
incorrect/incomplete/not performed
Product Administration
(Clinical Service)
Events that occur during the administration of blood
products.
UT 00 Detail not specified
UT 01 Administered intended product to wrong patient
UT 02 Administered wrong product to intended patient
UT 03 Transfusion not performed in error
UT 05 Bedside check (patient ID confirmation)
incomplete/not performed
UT 06 Transfused product with unapproved IV fluid
UT 07 Transfusion delayed beyond pre-approved
timeframe
UT 09 Transfused unsuitable product (e.g.,
outdated/inappropriately stored)
UT 10 Administered components in wrong order
UT 11 Appropriate monitoring of patient not
performed
UT 14 Transfusion volume too low (per order or SOP)
UT 15 Transfusion volume too high (per order or
SOP)
UT 16 Transfusion rate too slow (per order or SOP)
UT 17 Transfusion rate too fast (per order or SOP)
UT 18 Inappropriate preparation of product
UT 19 Transfusion protocol not followed (not
otherwise specified)
UT 22 Order/consent check incorrect/not performed
UT 23 Transfusion documentation
incorrect/incomplete/not performed
UT 24 Transfusion documentation not returned to
transfusion service
UT 26 Transfusion reaction protocol not followed
Other
MS 99 Other

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 28 of 31
February 2023
Occupation Codes
Laboratory
IVT
IVT Team Staff
MLT
Medical Laboratory Technician
MTE
Medical Technologist
PHL
Phlebotomist/IV Team
Nursing
LPN
Licensed Practical Nurse
CNA
Nurse Anesthetist
CNM
Certified Nurse Midwife
NUA
Nursing Assistant
NUP
Nurse Practitioner
RNU
Registered Nurse
Physician
FEL
Fellow
MST
Medical Student
PHY
Attending/Staff Physician
RES
Intern/Resident
Technicians
EMT
EMT/Paramedic
HEM
Hemodialysis Technician
ORS
OR/Surgery Technician
PCT
Patient Care Technician
Other Personnel
CLA
Clerical/Administrative
TRA
Transport/Messenger/Porter

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 29 of 31
February 2023
Occupation Codes
(continued)
Additional Occupation Types
ATT
Attendant/Orderly
CSS
Central Supply
CSW
Counselor/Social Worker
DIT
Dietician
DNA
Dental Assistant/Technician
DNH
Dental Hygienist
DNO
Other Dental Worker
DNT
Dentist
DST
Dental Student
FOS
Food Service
HSK
Housekeeper
ICP
Infection Control Professional
LAU
Laundry Staff
MNT
Maintenance/Engineering
MOR
Morgue Technician
OAS
Other Ancillary Staff
OFR
Other First Responder
OH
Occupational Health Professional
OMS
Other Medical Staff
OTH
Other
OTT
Other Technician/Therapist
PAS
Physician Assistant
PHA
Pharmacist
PHW
Public Health Worker
PLT
Physical Therapist
PSY
Psychiatric Technician
RCH
Researcher
RDT
Radiologic Technologist
RTT
Respiratory Therapist/Technician
STU
Other Student
VOL
Volunteer

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 30 of 31
February 2023
Incident Glossary
Incident Result
Product transfused; reaction (No recovery; harm):
A product related to this incident was transfused; the patient experienced an adverse reaction.
Product transfused; no reaction (No recovery; no harm):
A product related to this incident was transfused; the patient did not experience an adverse reaction.
No product transfused; unplanned recovery (Near miss; unplanned recovery):
No product related to this incident was transfused; the incident was discovered ad hoc, by accident, by
human lucky catch, etc.
No product transfused; planned recovery (Near miss; planned recovery):
No product related to this incident was transfused; the incident was discovered through a standardized
process or barrier designed to prevent errors.

NHSN Biovigilance Component
Hemovigilance Module Surveillance Protocol v2.8
Page 31 of 31
February 2023
Section 5. Hemovigilance Module Denominators
Required Reporting
Facilities must report the total number of units and aliquots of specified blood components transfused
and total number of discards each month. When reporting aliquots, the units from which they are made
should NOT be counted as a transfused unit. The components transfused count should include
autologous units. The total number of patient samples collected and total crossmatch procedures must
also be reported. This form must be completed each month that surveillance is conducted and data can
only be entered once the calendar month is over. For instance, February data must be entered after
March 1
st
. Additionally, data cannot be entered for upcoming months.
Pathogen Reduced Blood Products
The total number of transfused units of blood components which are produced with pathogen-reduction
technology (PRT) should be reported each month, if applicable. These PRT units are reported in Table 2
and are a subset of total number of units and aliquots transfused that are reported in Table 1. Table 3
relates to pathogen reduced apheresis platelets, if reported in table 2. For more guidance please refer to
the Denominator QuickLearn on the NHSN Blood Safety Surveillance website.
Electronic Reporting
In January 2017, the NHSN Hemovigilance Module can accept electronically reported denominator data
via clinical documentation architecture (CDA). Compared to manual reporting, electronic reporting will
decrease the time required for data collection and reporting, reduce data entry errors, and increase data
granularity. In order to electronically report data, facilities’ software system must have CDA functionality.
For more information about electronic reporting and CDA, review CDA Frequently Asked Questions on
the NHSN Blood Safety Surveillance website.
Form
CDC 57.303 Hemovigilance Module Monthly Reporting Denominators
Form Instructions
CDC 57.303 Hemovigilance Module Monthly Reporting Denominators Tables of Instructions
